Advanced Synthesis of Tetramethylbiphenyl Diamine for High-Performance Polyimide Manufacturing
Advanced Synthesis of Tetramethylbiphenyl Diamine for High-Performance Polyimide Manufacturing
The rapid evolution of the flexible electronics industry has placed unprecedented demands on the thermal and mechanical properties of substrate materials, specifically driving the need for advanced aromatic polyimides. Patent CN101186579A introduces a robust and industrially viable preparation method for 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl, a critical diamine monomer used in the synthesis of thermoplastic polyimides for Flexible Copper Clad Laminates (FCCL). This technology addresses the longstanding challenges of cost, purity, and environmental impact associated with traditional synthetic routes. By leveraging a unique combination of nucleophilic substitution and iron-catalyzed reduction, the process achieves product purity levels exceeding 99% while utilizing readily available reagents. The strategic implementation of this synthesis route offers a compelling value proposition for manufacturers seeking to optimize their supply chains for high-performance electronic chemicals without compromising on material specifications.
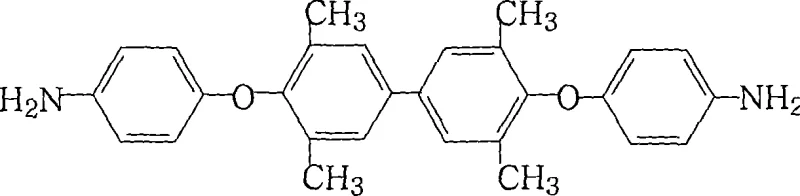
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines for polyimide applications has relied heavily on noble metal catalysis and harsh reaction conditions that pose significant operational and economic burdens. Prior art methods, such as those disclosed in Japanese Patent JP H03-101644, typically employ palladium on carbon (Pd/C) catalysts under high-pressure hydrogen atmospheres to reduce nitro intermediates. This approach not only inflates production costs due to the volatility of precious metal prices but also introduces severe safety risks associated with handling high-pressure hydrogen gas on an industrial scale. Furthermore, conventional workup procedures often involve pouring reaction mixtures into large volumes of methanol and water to precipitate products, a technique that generates substantial quantities of hazardous wastewater and complicates solvent recovery efforts. The difficulty in completely removing trace palladium residues from the final product is another critical drawback, as metal contamination can detrimentally affect the dielectric properties and long-term reliability of electronic grade polyimide films.
The Novel Approach
The methodology outlined in patent CN101186579A represents a paradigm shift towards safer and more sustainable chemical manufacturing by replacing noble metals with abundant base metals and optimizing solvent systems for recyclability. Instead of high-pressure hydrogenation, this novel approach utilizes a ferric chloride and hydrazine hydrate reduction system operating at atmospheric pressure and moderate temperatures between 50°C and 90°C. This modification drastically reduces the capital expenditure required for pressure-rated reactors and eliminates the safety hazards inherent to hydrogen gas usage. Additionally, the first step of the synthesis employs a biphasic solvent system comprising a polar aprotic solvent and a water-insoluble organic solvent like toluene or xylene, facilitating azeotropic water removal during reflux. This design allows for the efficient recovery and repeated recycling of organic solvents, significantly minimizing waste generation and aligning the process with stringent modern environmental regulations while maintaining high reaction yields.
Mechanistic Insights into FeCl3-Catalyzed Hydrazine Reduction
The core innovation of this synthesis lies in the second step, where the dinitro intermediate is converted to the target diamine through a highly efficient catalytic transfer hydrogenation mechanism mediated by iron species. In this system, ferric chloride acts as a catalyst precursor that interacts with hydrazine hydrate to generate active iron-hydride species or nascent hydrogen in situ, which subsequently reduces the nitro groups to amino groups. The presence of activated carbon in the reaction mixture plays a dual role: it serves as a support to disperse the catalytic iron species effectively and acts as a decolorizing agent to adsorb organic impurities and tarry byproducts formed during the reduction. This synergistic effect ensures that the reduction proceeds smoothly at relatively low temperatures, preventing thermal degradation of the sensitive biphenyl backbone. The reaction kinetics are carefully balanced by controlling the addition rate of hydrazine hydrate, ensuring that the exothermic nature of the reduction is managed safely while driving the conversion to completion over a period of 8 to 20 hours.
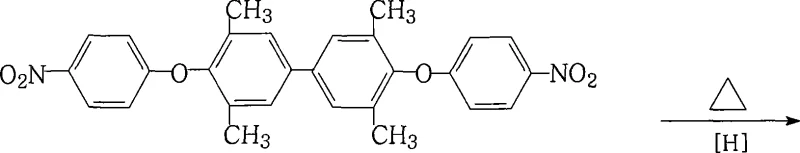
Impurity control is rigorously maintained throughout the synthetic pathway, particularly during the initial etherification stage where the stoichiometry of the base and halonitrobenzene is critical. The process utilizes a molar ratio of 3,3',5,5'-tetramethyl-4,4'-biphenyldiol to 4-halonitrobenzene of approximately 1:2.00 to 2.20, ensuring that the diol is fully consumed to prevent the formation of mono-substituted byproducts which are difficult to separate. The use of specific salt-forming agents like potassium carbonate or bicarbonate in a mixed solvent system promotes the formation of the phenoxide anion while minimizing side reactions such as hydrolysis of the nitro group. Following the reaction, the product is isolated by concentrating the solution and adding water, a method that leverages the solubility differences between the desired product and inorganic salts, resulting in a crude intermediate with purity levels already approaching 99% before the final reduction step even begins.
How to Synthesize 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl Efficiently
The synthesis protocol described in the patent provides a clear roadmap for scaling this chemistry from laboratory benchtop to commercial production vessels. The procedure begins with the precise charging of 3,3',5,5'-tetramethyl-4,4'-biphenyldiol and 4-chloronitrobenzene into a reactor equipped with a Dean-Stark trap for water separation. After the etherification is complete and the intermediate is isolated, the reduction phase requires careful temperature ramping from 50°C to roughly 77°C while maintaining vigorous stirring to ensure proper contact between the solid activated carbon, the liquid phase, and the evolving gas. Detailed standardized operating procedures regarding the specific addition rates of hydrazine and the filtration protocols for removing the spent carbon catalyst are essential for reproducibility.
- Perform nucleophilic substitution of 3,3',5,5'-tetramethyl-4,4'-biphenyldiol with 4-halonitrobenzene using a carbonate base in a mixed solvent system with azeotropic water removal.
- Isolate the intermediate 4,4'-bis(4-nitrophenoxy)-3,3',5,5'-tetramethylbiphenyl via concentration and precipitation.
- Reduce the dinitro intermediate to the final diamine using ferric chloride and hydrazine hydrate in an alcoholic solvent with activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple unit cost calculations. By eliminating the dependency on palladium catalysts, manufacturers can insulate their production costs from the extreme volatility of the precious metals market, leading to more predictable budgeting and pricing stability for downstream customers. The ability to recycle solvents repeatedly without significant loss of efficiency translates directly into reduced raw material consumption and lower waste disposal fees, contributing to a leaner and more cost-effective manufacturing operation. Furthermore, the simplified equipment requirements, which do not necessitate high-pressure hydrogenation infrastructure, lower the barrier to entry for production and reduce maintenance overheads, thereby enhancing the overall resilience and flexibility of the supply chain.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive ferric chloride results in a drastic reduction in direct material costs per kilogram of product. Additionally, the elimination of complex heavy metal scavenging steps, which are mandatory when using Pd/C to meet electronic grade specifications, simplifies the downstream purification process and reduces the consumption of specialized filtering media and reagents. This streamlined workflow minimizes labor hours and utility consumption, compounding the savings achieved through raw material optimization and creating a highly competitive cost structure for the final polyimide monomer.
- Enhanced Supply Chain Reliability: Sourcing ferric chloride and hydrazine hydrate is significantly more reliable than securing high-purity palladium on carbon, as these base chemicals are produced in vast quantities by a wide range of global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source supplier failures or geopolitical constraints on precious metal exports. Moreover, the robustness of the reaction conditions, which tolerate slight variations in temperature and pressure better than high-pressure hydrogenation, ensures consistent batch-to-batch quality and reliable delivery schedules for customers in the fast-paced electronics sector.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard atmospheric pressure reactors and common organic solvents that are easily handled in large-scale chemical plants. The environmental profile is markedly improved by the reduction of hazardous waste streams, particularly the avoidance of heavy metal contaminated sludge and the minimization of aqueous waste through azeotropic drying techniques. This alignment with green chemistry principles facilitates easier permitting for new production facilities and ensures long-term compliance with increasingly strict global environmental regulations, safeguarding the continuity of supply against regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this monomer into their polymer formulations and for procurement specialists assessing the total cost of ownership.
Q: What are the advantages of using FeCl3 over Pd/C for this reduction?
A: Using ferric chloride eliminates the need for expensive noble metal catalysts like palladium on carbon, significantly reducing raw material costs and removing the complex purification steps required to remove trace heavy metals from the final electronic-grade product.
Q: How does this process improve environmental compliance?
A: The process utilizes azeotropic distillation for water removal in the first step, allowing for efficient solvent recovery and recycling, while avoiding the generation of large volumes of wastewater associated with traditional methanol precipitation methods.
Q: What purity levels can be achieved with this method?
A: The patented method consistently yields products with purity exceeding 99%, evidenced by sharp melting points around 229°C, which is critical for ensuring the thermal stability of the resulting polyimide films.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101186579A can be realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl meets the exacting standards required for high-performance polyimide applications in the electronics industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific product needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of this critical electronic chemical intermediate.
