Advanced Synthesis of 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene for High-Performance Polyimide Manufacturing
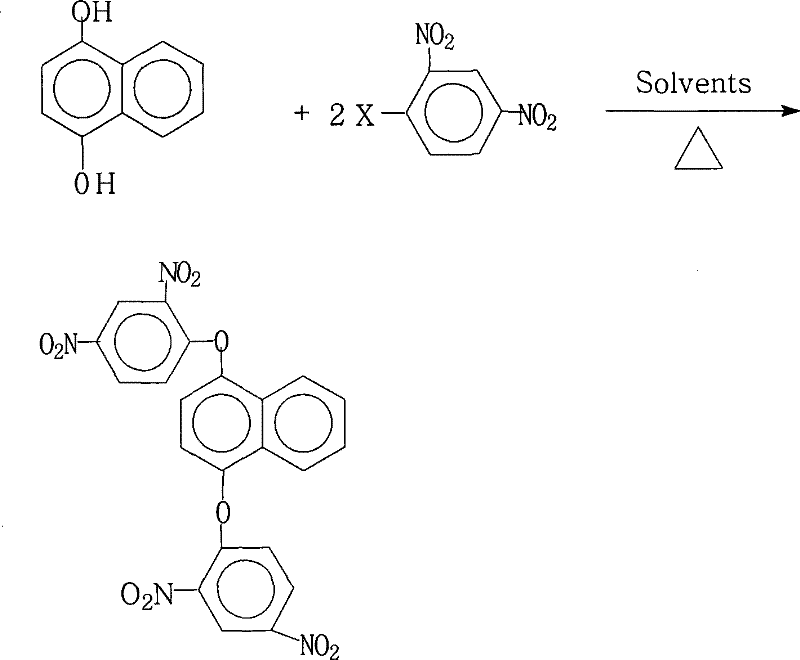
The development of advanced functional materials for the electronics and aerospace sectors relies heavily on the availability of high-purity monomers capable of withstanding extreme thermal and mechanical stress. Patent CN101234986B introduces a robust and scalable methodology for the preparation of 1,4-bis(2,4-dinitrophenoxy)naphthalene, a pivotal intermediate in the synthesis of highly branched aromatic polyimides. This specific chemical architecture is renowned for imparting exceptional thermostability, chemical resistance, and superior electrical insulation properties to the final polymer matrix. The disclosed process addresses critical gaps in prior art by offering a streamlined pathway that operates under atmospheric pressure, thereby eliminating the need for complex high-pressure reactors while maintaining exceptional product quality. For R&D directors and procurement specialists seeking reliable electronic chemical suppliers, this technology represents a significant leap forward in manufacturing efficiency and material performance consistency.
Furthermore, the strategic importance of this synthesis route lies in its adaptability to industrial-scale production without compromising on purity standards. The patent explicitly details a protocol that achieves yields exceeding 95% in optimized embodiments, coupled with product purities reaching 99.0% or higher. Such high fidelity in synthesis is paramount for downstream applications in liquid-crystal displays (LCD) and microelectronics, where trace impurities can catastrophically affect device longevity and performance. By leveraging a nucleophilic aromatic substitution mechanism between 1,4-naphthalenediol and various 2,4-dinitrohalobenzenes, the process ensures a clean reaction profile. This technical breakthrough not only secures the supply chain for high-performance polyimide precursors but also aligns with modern green chemistry principles through efficient solvent management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic ethers required for high-performance polyimides has been plagued by significant operational challenges and economic inefficiencies. Traditional routes often necessitated the use of harsh reaction conditions, including elevated pressures and the employment of expensive or hazardous catalysts that were difficult to remove from the final product matrix. These legacy methods frequently resulted in broad impurity profiles, requiring extensive and costly purification steps such as column chromatography or multiple recrystallizations, which drastically reduced overall throughput. Moreover, the reliance on stoichiometric amounts of strong bases in non-recyclable solvent systems generated substantial volumes of hazardous waste, creating a heavy burden on environmental compliance and waste treatment infrastructure. For supply chain heads, these factors translated into unpredictable lead times and volatile pricing structures due to the complexity of the manufacturing process and the disposal costs associated with toxic byproducts.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101234986B revolutionizes the production landscape by utilizing a simple yet highly effective reflux system that operates entirely at atmospheric pressure. This novel approach employs a dual-solvent strategy, combining water-insoluble organic solvents like toluene or xylene with strong polar aprotic solvents such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). This specific solvent combination facilitates the formation of the reactive phenoxide species while simultaneously enabling the azeotropic removal of water, driving the equilibrium towards product formation without the need for exotic dehydrating agents. The result is a process that is not only operationally simpler but also inherently safer, as it avoids the generation of corrosive intermediates and minimizes equipment investment requirements. This shift towards a more benign and efficient synthetic route directly supports cost reduction in electronic chemical manufacturing by streamlining the workflow and enhancing the reliability of the supply chain.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr) reaction, facilitated by the powerful electron-withdrawing effects of the nitro groups on the benzene ring. In this mechanism, the hydroxyl groups of 1,4-naphthalenediol are first deprotonated by a salt-forming agent, such as potassium carbonate or sodium hydroxide, to generate highly nucleophilic phenoxide anions. These anions then attack the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene at the position ortho or para to the nitro groups, where the electron density is lowest. The presence of two nitro groups significantly activates the ring towards nucleophilic attack, stabilizing the negatively charged Meisenheimer complex intermediate that forms during the transition state. This stabilization lowers the activation energy of the reaction, allowing the substitution of the halogen atom (fluorine, chlorine, bromine, or iodine) to proceed smoothly at moderate temperatures ranging from 80°C to 200°C.
From an impurity control perspective, the choice of the halogen leaving group and the precise control of the molar ratio between the diol and the halobenzene are critical parameters for ensuring high purity. The patent specifies a molar ratio of 1,4-naphthalenediol to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, which ensures that both hydroxyl groups are fully substituted, minimizing the formation of mono-substituted byproducts that could act as chain terminators in subsequent polymerization steps. Furthermore, the use of a mixed solvent system helps to solubilize both the organic reactants and the inorganic salts formed during the reaction, preventing the precipitation of salts that could occlude product or catalyze side reactions. The final workup involves concentrating the reaction mixture and adding water, which exploits the low solubility of the highly nitrated product in aqueous media to induce crystallization, effectively washing away soluble inorganic salts and residual solvents to yield a product with purity specifications suitable for high-end electronic applications.
How to Synthesize 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene Efficiently
To achieve the high yields and purity levels described in the patent, operators must strictly adhere to the specified protocol regarding solvent ratios and temperature control during the reflux period. The process begins with the careful charging of the reactor with the diol, the dinitrohalobenzene, and the chosen base in the mixed solvent system, followed by a prolonged heating phase to ensure complete conversion. Detailed standard operating procedures for this synthesis, including specific quantities and timing for each step, are essential for reproducibility and safety in a commercial setting.
- Charge a reactor with 1,4-naphthalenediol and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0 to 2.2, along with a salt-forming agent and a mixed organic solvent system.
- Heat the mixture to reflux temperatures between 80°C and 200°C for 3 to 18 hours to facilitate water separation and reaction completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain the final crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented synthesis route offers profound advantages in terms of cost structure and operational reliability. The elimination of high-pressure equipment requirements significantly reduces capital expenditure (CapEx) for new production lines, while the ability to run the reaction under atmospheric pressure enhances overall plant safety and reduces maintenance overheads. Additionally, the process is designed with solvent recovery in mind; the organic solvents used, such as toluene and DMF, can be conveniently separated and recycled repeatedly, leading to substantial cost savings in raw material consumption over the lifecycle of the production campaign. This focus on resource efficiency not only lowers the direct cost of goods sold (COGS) but also mitigates the financial risks associated with volatile solvent markets and stringent environmental regulations regarding waste disposal.
- Cost Reduction in Manufacturing: The economic viability of this process is largely driven by the use of readily available and inexpensive starting materials, such as 1,4-naphthalenediol and various dinitrochlorobenzenes, which are commodity chemicals with stable supply chains. By avoiding the use of precious metal catalysts or specialized reagents, the manufacturing cost is significantly reduced, allowing for more competitive pricing in the global market for electronic intermediates. Furthermore, the high yield reported in the patent examples means that less raw material is wasted per unit of product, maximizing the return on investment for every batch produced. The simplicity of the workup procedure, which relies on basic filtration and drying rather than complex purification techniques, further contributes to lower labor and utility costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures a consistent and reliable supply of high-purity 1,4-bis(2,4-dinitrophenoxy)naphthalene, which is critical for manufacturers of polyimides who cannot afford interruptions in their raw material flow. Since the reaction tolerates a range of halogenated reactants (fluoro, chloro, bromo, iodo), supply chain managers have the flexibility to switch between different grades of 2,4-dinitrohalobenzene based on availability and price without needing to requalify the entire process. This flexibility acts as a buffer against supply shocks in the upstream chemical market, ensuring continuity of operations even when specific raw materials face temporary shortages. The scalability of the process from laboratory to industrial scale further guarantees that demand surges can be met without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions continue to tighten globally, the environmental profile of a chemical process becomes a key determinant of its long-term viability. This method generates minimal three-waste discharge due to the efficient recycling of solvents and the absence of corrosive byproducts, making it easier to obtain and maintain environmental permits for large-scale production facilities. The ability to scale up the reaction without encountering the heat transfer or mixing limitations often associated with high-pressure or exothermic processes allows for the construction of larger batch reactors, increasing total production capacity. Consequently, this aligns perfectly with the goals of sustainable manufacturing, reducing the carbon footprint of the supply chain while ensuring compliance with international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate, based on the detailed disclosures within the patent literature. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this material into their existing product portfolios or supply networks.
Q: What is the primary application of 1,4-bis(2,4-dinitrophenoxy)naphthalene?
A: This compound serves as a crucial synthetic monomer source material for highly branched aromatic polyimides, which are essential for high-temperature resistant materials in aerospace, aviation, and LCD display technologies.
Q: What are the optimal reaction conditions described in the patent?
A: The process utilizes a mixed solvent system of water-insoluble organics (like toluene) and polar aprotic solvents (like DMF), reacting at 80°C to 200°C under atmospheric pressure with a salt-forming agent.
Q: How does this method improve environmental compliance?
A: The method allows for convenient solvent recovery and repeated recycling, significantly reducing three-waste discharge and eliminating the need for corrosive reagents or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of next-generation electronic materials and polyimide systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients with consistency and precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our dedication to quality assurance ensures that the 1,4-bis(2,4-dinitrophenoxy)naphthalene we supply is perfectly suited for the synthesis of high-cladodification polyimides required in aerospace and microelectronics applications.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient synthesis route can lower your overall material costs while enhancing supply security. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your organization.
