Scalable Production of 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene for Advanced Polyimide Applications
Scalable Production of 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene for Advanced Polyimide Applications
The development of advanced functional materials for the aerospace and electronic industries relies heavily on the availability of high-performance monomers with precise structural integrity. Patent CN101234986A introduces a robust and industrially viable preparation method for 1,4-bis(2,4-dinitrophenoxy)naphthalene, a pivotal intermediate in the synthesis of highly branched aromatic polyimides. This specific chemical architecture is renowned for imparting exceptional thermal stability, chemical resistance, and superior mechanical properties to the final polymer matrix, making it indispensable for liquid crystal displays (LCDs) and microelectronic components. The disclosed methodology addresses the historical lack of published literature regarding the efficient synthesis of this complex aromatic ether, offering a pathway that balances high yield with operational simplicity. By leveraging a nucleophilic substitution strategy under controlled reflux conditions, the process ensures the production of material with purity levels exceeding 99%, which is critical for downstream polymerization performance. For R&D teams and procurement specialists alike, understanding this synthetic route provides a strategic advantage in securing a reliable electronic chemical supplier capable of meeting the rigorous demands of next-generation material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic ethers containing multiple nitro groups has been plagued by significant challenges related to reaction control and purification efficiency. Traditional approaches often suffer from incomplete conversion due to the steric hindrance presented by the naphthalene core and the electron-withdrawing nature of the nitro substituents, which can deactivate the nucleophile if not properly managed. Furthermore, many legacy processes require harsh reaction conditions, such as elevated pressures or the use of expensive phase-transfer catalysts that are difficult to remove from the final product, leading to contamination issues in sensitive electronic applications. The inability to effectively separate unreacted starting materials and by-products often results in a broad impurity profile that compromises the thermal and electrical properties of the resulting polyimide. Additionally, conventional solvent systems frequently lack the capacity for efficient recovery, generating substantial volumes of hazardous waste that increase both disposal costs and environmental liability. These factors collectively create a bottleneck in the supply chain, limiting the availability of high-purity precursors necessary for high-end manufacturing.
The Novel Approach
The innovative method detailed in the patent data overcomes these obstacles through a carefully optimized etherification protocol that utilizes a dual-solvent system and specific salt-forming agents. By employing a mixture of a water-insoluble organic solvent, such as toluene or xylene, alongside a highly polar aprotic solvent like N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), the reaction medium achieves an ideal balance of solubility for both the organic substrates and the inorganic base. This unique solvent combination facilitates the formation of the reactive phenoxide species while maintaining the solubility of the growing oligomeric species, preventing premature precipitation that could halt the reaction. The process operates under atmospheric pressure with heating and reflux water separation, which drives the equilibrium forward by continuously removing the water by-product generated during the salt formation and substitution steps. This approach not only simplifies the equipment requirements, eliminating the need for costly high-pressure reactors, but also ensures a cleaner reaction profile with minimal side reactions. The result is a streamlined workflow that delivers consistent quality and supports the commercial scale-up of complex aromatic ethers required for advanced material applications.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), where the phenoxide anion derived from 1,4-naphthalenediol attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene. The presence of two strongly electron-withdrawing nitro groups at the ortho and para positions relative to the halogen leaving group significantly activates the benzene ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. This stabilization is crucial for lowering the activation energy of the rate-determining step, allowing the reaction to proceed efficiently at moderate temperatures ranging from 80°C to 200°C. The choice of the halogen leaving group (fluorine, chlorine, bromine, or iodine) offers flexibility in tuning the reaction kinetics, with fluorine often providing superior rates due to its high electronegativity enhancing the electrophilicity of the ipso-carbon, despite being a poorer leaving group in aliphatic systems. The base, typically an alkali metal carbonate or hydroxide, serves the dual purpose of deprotonating the naphthalenediol to generate the nucleophile and neutralizing the hydrogen halide acid produced during the substitution. This mechanistic pathway ensures high regioselectivity and minimizes the formation of isomeric by-products, which is essential for maintaining the linearity and crystallinity of the final polyimide structure.
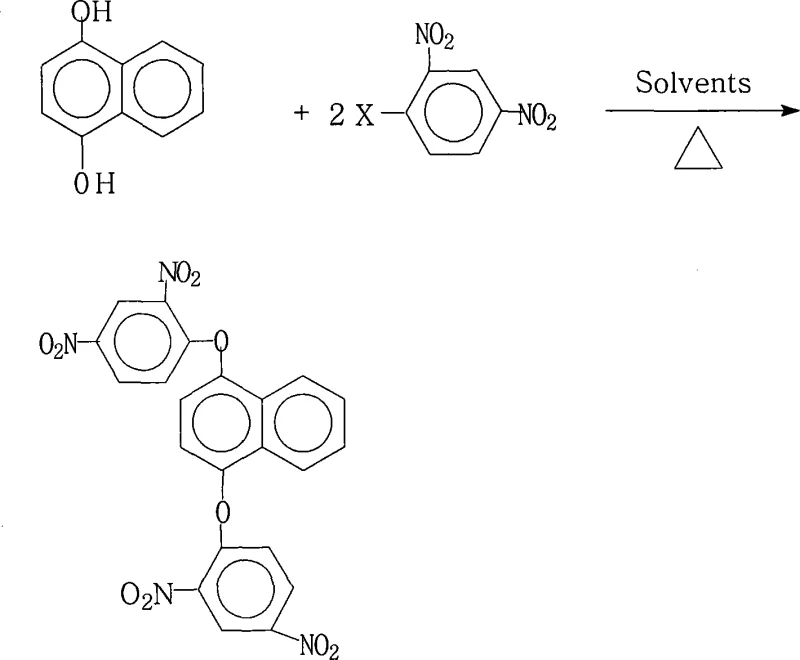
Controlling the impurity profile in this reaction is paramount, particularly for applications in the semiconductor and display sectors where trace metal ions or organic contaminants can degrade device performance. The use of a mixed solvent system plays a critical role in impurity management by keeping the desired product in solution during the reaction while allowing inorganic salts to remain suspended or be easily separated during the workup phase. Upon completion, the addition of water to the concentrated reaction mixture induces precipitation of the product, leveraging the differential solubility to exclude soluble organic impurities and residual inorganic bases. The washing steps with hot water further purify the crystal lattice by removing adsorbed salts and polar by-products, resulting in a final product with purity specifications often exceeding 99%. This rigorous control over the reaction environment and workup procedure ensures that the resulting 1,4-bis(2,4-dinitrophenoxy)naphthalene meets the stringent quality standards required for high-purity polyimide precursors, thereby reducing the risk of failure in downstream polymerization processes.
How to Synthesize 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene Efficiently
The synthesis of this high-value intermediate requires precise control over stoichiometry and thermal conditions to maximize yield and minimize waste generation. The patented process outlines a straightforward procedure where 1,4-naphthalenediol and 2,4-dinitrohalobenzene are combined in a molar ratio of approximately 1.0:2.0 to 2.2 in the presence of a base and the optimized solvent mixture. The reaction is heated to reflux with continuous water separation for a duration of 3 to 18 hours, depending on the specific reactivity of the halogenated substrate and the boiling point of the solvent system employed. Following the reaction, the mixture is concentrated to recover the valuable organic solvents for reuse, cooled, and treated with water to precipitate the solid product, which is then filtered, washed, and dried to obtain the final crystalline material. For a comprehensive breakdown of the exact reagent quantities, temperature profiles, and safety protocols, please refer to the standardized synthesis guide below.
- Charge the reactor with 1,4-naphthalenediol, 2,4-dinitrohalobenzene, a salt-forming agent like potassium carbonate, and a mixed solvent system.
- Heat the mixture to reflux (80°C to 200°C) with continuous water separation for 3 to 18 hours to drive the etherification reaction to completion.
- Concentrate the reaction solution, cool the system, precipitate the product with water, and filter to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthesis method offers profound benefits for organizations seeking cost reduction in display material manufacturing and enhanced supply chain resilience. The process is designed with industrial scalability in mind, utilizing common, commercially available raw materials such as 1,4-naphthalenediol and various dinitrohalobenzenes, which mitigates the risk of supply disruptions associated with exotic or proprietary reagents. Furthermore, the operational simplicity of running the reaction under atmospheric pressure significantly lowers the capital expenditure (CAPEX) required for plant infrastructure, as it eliminates the need for specialized high-pressure vessels and complex safety systems. This accessibility allows for a more distributed manufacturing network, reducing lead times for high-purity intermediates and enabling faster response to market fluctuations in the electronics sector. The ability to recycle solvents repeatedly not only aligns with green chemistry principles but also translates into substantial operational cost savings by reducing the volume of fresh solvent purchases and waste disposal fees.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive transition metal catalysts and the use of inexpensive inorganic bases like potassium carbonate or sodium hydroxide. By avoiding the need for costly noble metals, the process removes the requirement for extensive and expensive metal scavenging steps during purification, which traditionally adds significant time and cost to the production cycle. Additionally, the high atom economy of the substitution reaction ensures that the majority of the starting mass is incorporated into the final product, minimizing raw material waste. The efficient recovery and reuse of the organic solvent fraction further compound these savings, creating a lean manufacturing model that maximizes resource utilization and minimizes variable production costs without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for both substrates and reagents ensures a stable and predictable supply chain, shielding manufacturers from the volatility often seen with specialized fine chemical ingredients. Since the reaction does not generate corrosive by-products or require extreme conditions, the equipment maintenance intervals are extended, and the risk of unplanned downtime due to reactor corrosion or failure is drastically reduced. This operational robustness guarantees a consistent output of material, allowing procurement managers to plan inventory levels with greater confidence and reduce the need for excessive safety stock. The simplified logistics of handling non-hazardous solid bases and standard organic solvents also streamline the inbound logistics process, facilitating smoother integration into existing chemical supply networks.
- Scalability and Environmental Compliance: The process generates minimal three-waste (wastewater, waste gas, and solid waste), primarily consisting of aqueous salt solutions and recovered solvents, which are easier to treat and manage compared to heavy metal-laden effluents. The absence of toxic catalysts simplifies the regulatory compliance landscape, reducing the administrative burden and potential liabilities associated with environmental reporting and permitting. As global regulations on chemical manufacturing tighten, having a process that inherently produces less waste and consumes fewer resources provides a competitive advantage in markets with strict environmental standards. This sustainability profile not only future-proofs the supply chain against regulatory changes but also enhances the brand reputation of the end-product manufacturers who prioritize eco-friendly sourcing in their value chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,4-bis(2,4-dinitrophenoxy)naphthalene, based on the detailed specifications provided in the patent literature. Understanding these nuances helps stakeholders make informed decisions regarding process adoption and material qualification. The answers reflect the specific advantages of the described nucleophilic substitution route over alternative synthetic strategies.
Q: What is the primary application of 1,4-bis(2,4-dinitrophenoxy)naphthalene?
A: It serves as a critical intermediate for synthesizing highly branched aromatic polyimides, which are essential for high-performance applications in aerospace, microelectronics, and LCD displays due to their thermal stability and mechanical strength.
Q: How does this synthesis method improve environmental compliance?
A: The process utilizes a solvent system that allows for convenient recovery and repeated recycling, significantly reducing waste generation and minimizing the environmental footprint compared to traditional single-use solvent protocols.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction operates under atmospheric pressure without requiring specialized high-pressure equipment, and uses readily available raw materials, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(2,4-Dinitrophenoxy)Naphthalene Supplier
As the demand for high-performance polyimides continues to surge in the aerospace and flexible electronics sectors, securing a partner with deep technical expertise in aromatic ether synthesis is critical for maintaining product quality and market competitiveness. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-volume supply. Our state-of-the-art facilities are equipped to handle the specific solvent recovery and purification requirements of this synthesis, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that the performance of your final polyimide material is directly dependent on the quality of the monomer, which is why we prioritize process control and impurity profiling at every stage of manufacturing.
We invite you to collaborate with us to optimize your supply chain and achieve significant efficiencies in your material sourcing strategy. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project needs. Let us help you accelerate your development timeline with a reliable supply of this critical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
