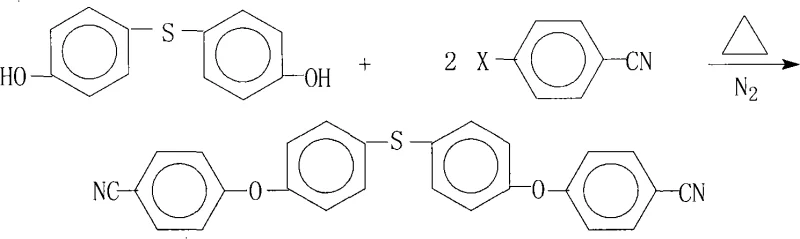
Scalable Synthesis of 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide for High-Performance Electronic Polymers
The rapid advancement of flexible electronics and high-temperature resistant materials has created an urgent demand for specialized monomers capable of withstanding extreme thermal and mechanical stress. Patent CN101328142A introduces a groundbreaking preparation method for 4,4'-bis(4-cyanophenoxy)diphenyl sulfide, a critical intermediate used in the synthesis of advanced aromatic polyamide-imide resins. This compound serves as a foundational building block for Flexible Copper Clad Laminates (FCCL), liquid crystal alignment agents, and high-performance composite matrix resins. The disclosed technology addresses significant historical challenges in synthesizing this complex ether-sulfide structure, offering a pathway that combines high yield with exceptional purity. By leveraging a specific nucleophilic aromatic substitution strategy under controlled thermal conditions, this method ensures the production of electronic-grade intermediates that meet the rigorous standards of the semiconductor and aerospace industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of bis-ether compounds containing nitrile groups has been plagued by inefficient reaction kinetics and difficult purification processes. Conventional methods often suffer from incomplete conversion due to the poor solubility of phenolic precursors in standard organic solvents, leading to heterogeneous reaction mixtures that stall before reaching completion. Furthermore, the removal of inorganic salt byproducts, such as potassium halides generated during the substitution, frequently requires extensive aqueous workups that can hydrolyze sensitive nitrile groups or lead to significant product loss. Many older protocols also rely on stoichiometric excesses of expensive reagents without effective mechanisms to drive the equilibrium forward, resulting in yields that are commercially unviable for large-scale electronic material manufacturing. The presence of trace impurities in these traditional routes often necessitates multiple recrystallization steps, drastically increasing production time and solvent consumption while failing to guarantee the ultra-high purity required for dielectric applications.
The Novel Approach
The methodology outlined in CN101328142A represents a significant technological leap by optimizing the reaction environment through a sophisticated solvent system and precise stoichiometric control. This novel approach utilizes a combination of aprotic polar solvents, such as N-methylpyrrolidone (NMP) or dimethylformamide (DMF), paired with an entrainer like toluene or xylene to facilitate azeotropic dehydration. This dual-solvent strategy not only enhances the solubility of the reactants but also actively removes water generated during the salt formation phase, pushing the reaction equilibrium towards the desired product. By operating at elevated temperatures ranging from 120°C to 200°C under a nitrogen atmosphere, the process ensures rapid kinetics while preventing oxidative degradation of the sensitive sulfide linkage. The result is a streamlined one-pot synthesis that achieves conversion rates and yields far superior to legacy methods, producing a crude product that is already of high quality prior to the final crystallization step.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr) reaction, where the phenoxide anion acts as a potent nucleophile attacking the electron-deficient aromatic ring of the 4-halobenzonitrile. The presence of the strong electron-withdrawing cyano group at the para-position significantly activates the aromatic ring towards nucleophilic attack, facilitating the displacement of the halogen leaving group (fluorine, chlorine, or bromine). The reaction mechanism initiates with the deprotonation of the 4,4'-dihydroxydiphenyl sulfide by a base such as potassium carbonate, generating the reactive phenoxide species in situ. This anionic intermediate then attacks the carbon atom bearing the halogen on the benzonitrile molecule, forming a transient Meisenheimer complex before expelling the halide ion to restore aromaticity. The use of polar aprotic solvents is crucial here, as they effectively solvate the cationic counter-ions (like K+) while leaving the phenoxide anion relatively naked and highly reactive, thereby accelerating the rate of substitution.
Impurity control in this mechanism is achieved through careful management of reaction temperature and stoichiometry. By maintaining a slight molar excess of the 4-halobenzonitrile (ratio of 1.0:2.0 to 2.4), the process ensures that both hydroxyl groups on the diphenyl sulfide core are fully substituted, minimizing the formation of mono-substituted byproducts which are difficult to separate. Additionally, the hot filtration step described in the patent is mechanistically vital; it removes the insoluble inorganic salts (e.g., KCl, KBr) while the product remains dissolved in the hot mother liquor. This physical separation prevents the entrapment of salt crystals within the final product lattice during crystallization, which is a common source of ash content and ionic contamination in electronic chemicals. The subsequent cooling and crystallization allow the target molecule to precipitate in a highly ordered lattice, excluding remaining soluble impurities and ensuring the final purity exceeds 99%.
How to Synthesize 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide Efficiently
Executing this synthesis requires strict adherence to the patented parameters regarding reagent ratios and thermal profiles to ensure reproducibility and safety. The process begins by charging the reactor with the dihydroxy precursor and the halobenzonitrile derivative under an inert nitrogen blanket to prevent oxidation. A salt-forming agent is added along with the solvent mixture, and the system is heated gradually to initiate the formation of the phenoxide salt and the subsequent substitution reaction. The temperature is carefully ramped up to the optimal range of 120°C to 200°C and maintained for a duration of 10 to 30 hours, depending on the specific halogen reactivity and solvent choice.
- Charge 4,4'-dihydroxydiphenyl sulfide and 4-halobenzonitrile (molar ratio 1: 2.0-2.4) with a salt-forming agent and solvents into a reactor under nitrogen.
- Heat the mixture to 120°C-200°C for 10-30 hours, utilizing an entrainer to remove water azeotropically and drive the reaction to completion.
- Filter the hot reaction mixture to remove salts, concentrate the filtrate under reduced pressure, and crystallize the product to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages in terms of cost stability and operational efficiency. The ability to utilize common, commercially available starting materials like 4-chlorobenzonitrile and 4,4'-dihydroxydiphenyl sulfide mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the robustness of the reaction conditions allows for significant flexibility in sourcing; the patent indicates that various halogens (F, Cl, Br) and different bases (carbonates, hydroxides) can be used interchangeably without compromising the outcome. This flexibility empowers purchasing teams to negotiate better pricing by switching between equivalent reagents based on market availability, ensuring continuous production even when specific commodity prices fluctuate wildly.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the elimination of complex purification stages. By achieving high conversion rates directly in the reactor, the need for extensive chromatographic purification or multiple recrystallizations is removed, which drastically cuts down on solvent usage and labor costs. The recovery and recycling of the aprotic polar solvents and entrainers further contribute to significant operational expenditure savings, as these solvents represent a major cost component in fine chemical synthesis. Additionally, the high yield reported in the examples reduces the cost per kilogram of the final active ingredient, allowing for more competitive pricing in the downstream polymer market.
- Enhanced Supply Chain Reliability: The simplicity of the workup procedure, involving basic filtration and crystallization, means that the manufacturing cycle time is significantly shorter compared to multi-step synthetic routes. This efficiency translates directly into improved lead times for customers, enabling faster response to market demands for FCCL and polyimide materials. The process is also inherently safer and more environmentally friendly due to the closed-loop nature of the solvent recovery and the minimization of aqueous waste streams. This compliance with environmental regulations reduces the risk of production shutdowns due to regulatory scrutiny, ensuring a steady and reliable supply of this critical electronic intermediate.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction conditions are well-suited for large-scale industrial reactors, as they do not require exotic catalysts or extreme pressures. The use of standard heating and stirring equipment facilitates a smooth transition from pilot plant to commercial production volumes. Moreover, the reduction in three wastes (waste gas, waste water, and solid waste) aligns with modern green chemistry principles, making the facility more sustainable. The ability to recycle solvents not only lowers costs but also minimizes the environmental footprint, which is increasingly becoming a key criterion for selection by top-tier electronics manufacturers who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-bis(4-cyanophenoxy)diphenyl sulfide. These answers are derived directly from the technical specifications and beneficial effects detailed in the patent literature to provide clarity for potential partners. Understanding these nuances is essential for R&D teams evaluating this material for new formulations and for procurement officers assessing supplier capabilities.
Q: What are the critical reaction parameters for maximizing yield in this synthesis?
A: According to patent CN101328142A, maintaining a precise molar ratio of 4-halobenzonitrile to dihydroxy precursor (2.0-2.4:1) and utilizing a dual-solvent system with an entrainer like toluene or xylene at temperatures between 120°C and 200°C are critical for achieving yields exceeding 90%.
Q: How does this method ensure high purity suitable for electronic applications?
A: The process employs a hot filtration step to remove inorganic salt byproducts immediately after reaction, followed by recrystallization. This effectively eliminates impurities, resulting in a final product purity of over 99%, which is essential for high-performance polyimide and FCCL applications.
Q: Can the solvents used in this process be recycled?
A: Yes, the patent explicitly highlights that the aprotic polar solvents (such as DMF or NMP) and entrainers are easy to recover and recycle. This feature significantly reduces waste generation and lowers the overall environmental footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final electronic materials depends entirely on the quality of the monomers you source. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that every gram of 4,4'-bis(4-cyanophenoxy)diphenyl sulfide we deliver is backed by comprehensive data packages that validate its suitability for high-performance polyimide and FCCL applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your next generation of electronic materials. Let us be your partner in innovation, delivering the reliability and quality your projects demand.
