Scalable Production of 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide for Advanced Electronic Polymers
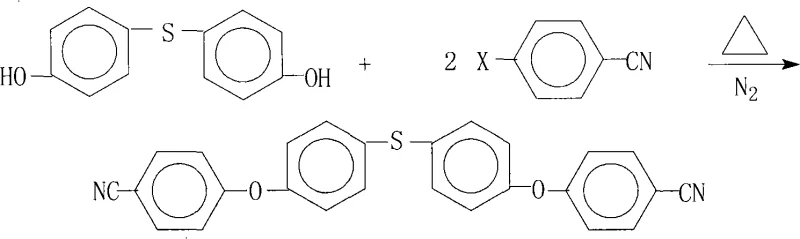
The rapid evolution of flexible electronics and high-performance composite materials has intensified the demand for specialized aromatic monomers capable of withstanding extreme thermal and mechanical stress. Patent CN101328142A introduces a groundbreaking preparation method for 4,4'-bis(4-cyanophenoxy)diphenyl sulfide, a critical intermediate for synthesizing high-temperature resistant aromatic polyamide-imide resins. This molecule serves as a foundational building block for advanced applications such as Flexible Copper Clad Laminates (FCCL), liquid crystal alignment agents, and passivation coatings for VLSI. The disclosed technology addresses the historical lack of efficient synthetic routes for this specific bis-nitrile compound, offering a pathway that combines operational simplicity with exceptional product quality. By leveraging a nucleophilic aromatic substitution mechanism in a carefully optimized solvent system, this process achieves yields exceeding 90% and purities greater than 99%, making it an ideal candidate for reliable electronic chemical supplier networks seeking to secure high-grade raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN101328142A, the synthesis of complex aromatic ethers containing nitrile functionalities often suffered from significant inefficiencies that hindered large-scale adoption. Traditional approaches frequently relied on multi-step sequences involving harsh conditions or expensive transition metal catalysts, which introduced difficult-to-remove impurities detrimental to electronic performance. Furthermore, many conventional methods struggled with poor solubility of the phenolic precursors, leading to incomplete conversions and heterogeneous reaction mixtures that were challenging to control on an industrial scale. The absence of effective water removal strategies in older protocols often resulted in hydrolysis of the nitrile groups or reversal of the etherification, drastically lowering the overall yield. These technical bottlenecks not only inflated production costs but also created substantial environmental burdens due to the generation of complex waste streams that required extensive treatment before disposal.
The Novel Approach
The methodology presented in the patent revolutionizes the production landscape by employing a direct, one-pot nucleophilic substitution reaction that elegantly bypasses the complexities of previous techniques. By reacting 4,4'-dihydroxydiphenyl sulfide with 4-halobenzonitrile in the presence of a specific salt-forming agent and an entrainer, the process ensures complete conversion while maintaining the integrity of the sensitive cyano groups. The strategic use of an azeotropic entrainer, such as toluene or xylene, facilitates the continuous removal of water generated during the salt formation, driving the equilibrium towards the desired product without requiring excessive temperatures that could degrade the polymer precursors. This approach not only simplifies the operational workflow by consolidating multiple steps into a single reactor but also significantly enhances the safety profile by eliminating the need for pyrophoric reagents or sensitive catalytic systems. The result is a robust, scalable process that delivers consistent quality suitable for the rigorous standards of the semiconductor and aerospace industries.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SNAr), where the phenoxide anion acts as a potent nucleophile attacking the electron-deficient aromatic ring of the halobenzonitrile. The presence of the electron-withdrawing cyano group at the para-position significantly activates the aromatic ring towards nucleophilic attack by stabilizing the Meisenheimer complex intermediate through resonance. In this system, the salt-forming agent, typically an alkali metal carbonate like potassium carbonate, deprotonates the hydroxyl groups of the diphenyl sulfide derivative, generating the reactive phenoxide species in situ. The choice of solvent is paramount; aprotic polar solvents such as N,N-dimethylformamide (DMF) or N-methylpyrrolidone (NMP) are utilized because they effectively solvate the cation without hydrogen bonding to the nucleophile, thereby increasing its reactivity and ensuring a homogeneous reaction phase. This precise control over the reaction environment allows for the selective formation of the ether linkage while preserving the nitrile functionality, which is crucial for subsequent polymerization steps.
Impurity control in this process is meticulously managed through the optimization of stoichiometry and temperature profiles to minimize side reactions such as hydrolysis or over-alkylation. The patent specifies a molar ratio of dihydroxy compound to halobenzonitrile between 1.0:2.0 and 1.0:2.4, ensuring a slight excess of the electrophile to drive the reaction to completion without leaving significant amounts of mono-substituted intermediates. Furthermore, the temperature range of 120°C to 200°C is carefully selected to provide sufficient activation energy for the substitution while avoiding thermal decomposition of the reactants or products. The subsequent workup procedure, involving hot filtration to remove inorganic salts followed by crystallization, acts as a powerful purification barrier. This physical separation technique effectively isolates the target bis-nitrile compound from unreacted starting materials and minor byproducts, ensuring that the final material meets the stringent purity specifications required for high-performance polyamide-imide resin synthesis.
How to Synthesize 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for manufacturing this high-value intermediate with maximum efficiency and minimal environmental impact. The process begins with the careful charging of reactants under an inert nitrogen atmosphere to prevent oxidation, followed by a controlled heating ramp that utilizes azeotropic distillation to manage water content. This specific sequence of operations is designed to maximize yield while maintaining safety and reproducibility across different batch sizes. For detailed standard operating procedures and specific parameter optimizations tailored to your facility, please refer to the comprehensive guide below.
- Charge 4,4'-dihydroxydiphenyl sulfide and 4-halobenzonitrile (molar ratio 1: 2.0-2.4) into a reactor with salt-forming agent, entrainer, and aprotic polar solvent under nitrogen.
- Heat the mixture to 120°C-200°C and maintain reaction for 10-30 hours, utilizing the entrainer to remove water azeotropically.
- Filter hot to remove salts, concentrate the mother liquor under reduced pressure, cool to crystallize, then filter and dry the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for electronic chemical manufacturing. The elimination of expensive transition metal catalysts removes a significant cost center associated with both raw material acquisition and the downstream purification processes required to meet low-metal specifications. Additionally, the ability to recover and recycle the bulk solvents used in the reaction dramatically reduces the volume of hazardous waste generated, leading to substantial cost savings in waste disposal and compliance management. This closed-loop solvent system not only lowers the variable cost per kilogram of product but also insulates the supply chain from volatility in solvent pricing, providing greater financial predictability for long-term contracts.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing readily available commodity chemicals such as potassium carbonate and common halobenzonitriles as starting materials, avoiding the need for bespoke or proprietary reagents. The high atom economy of the nucleophilic substitution reaction ensures that the majority of the input mass is converted into the desired product, minimizing raw material waste. Furthermore, the simplified workup procedure, which relies on standard filtration and crystallization rather than complex chromatography or extraction, reduces labor hours and energy consumption per batch. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of this chemical process translates directly into supply chain resilience, as the reaction tolerances are wide enough to accommodate minor variations in raw material quality without affecting the final outcome. The use of stable, non-hazardous reagents simplifies logistics and storage requirements, reducing the risk of supply disruptions caused by regulatory changes or transportation restrictions on dangerous goods. Moreover, the scalability of the method means that production volumes can be rapidly increased to meet surging demand from the electronics sector without the need for extensive capital investment in new reactor types or specialized equipment. This flexibility ensures a continuous and reliable flow of materials to downstream polymer manufacturers.
- Scalability and Environmental Compliance: The design of this synthesis inherently supports green chemistry principles by minimizing the generation of three wastes (waste gas, waste water, and solid waste) through efficient solvent recovery and high conversion rates. The absence of heavy metals in the reaction mixture simplifies the environmental permitting process and reduces the liability associated with soil or water contamination. As global regulations on industrial emissions become increasingly stringent, adopting a process that naturally aligns with sustainability goals provides a strategic advantage in maintaining operational licenses and corporate social responsibility standings. This forward-looking approach ensures long-term viability in a regulatory landscape that favors cleaner production technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-bis(4-cyanophenoxy)diphenyl sulfide. These answers are derived directly from the technical specifications and experimental data provided in the source patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific polymer formulation or electronic material supply chain.
Q: What is the primary advantage of this synthesis route for electronic grade intermediates?
A: The process utilizes a simple one-pot nucleophilic substitution that avoids complex transition metal catalysts, ensuring low metal residue critical for electronic applications like FCCL.
Q: How is high purity (>99%) achieved in this manufacturing process?
A: High purity is secured through a specific hot filtration step to remove inorganic salts followed by controlled crystallization from the concentrated mother liquor, effectively eliminating unreacted starting materials.
Q: Can the solvents used in this reaction be recycled for cost efficiency?
A: Yes, the patent explicitly highlights that the aprotic polar solvents and entrainers are easy to recover and recycle, significantly reducing waste generation and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(4-Cyanophenoxy)Diphenyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of next-generation electronic materials and high-performance polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of 4,4'-bis(4-cyanophenoxy)diphenyl sulfide meets the exacting standards necessary for FCCL and polyamide-imide applications. Our commitment to technical excellence allows us to support your R&D efforts with materials that perform reliably in the most demanding environments.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis capabilities can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing process can reduce your total cost of ownership. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition from laboratory scale to full commercial implementation.
