Advanced Clopidogrel Manufacturing: Overcoming Prior Art Limitations for Commercial Scale-Up
The pharmaceutical landscape for antiplatelet agents continues to evolve, driven by the relentless demand for high-purity, cost-effective active pharmaceutical ingredients (APIs). Patent CN101787032A, published in July 2010, introduces a transformative approach to the synthesis of clopidogrel and its salts, specifically targeting the bottlenecks inherent in legacy manufacturing processes. This intellectual property outlines a novel integrated synthesis method that leverages D-camphorsulfonic acid for optical resolution, bypassing the toxic and expensive reagents typical of earlier generations. For R&D directors and procurement strategists, this technology represents a pivotal shift towards greener, more economically viable production pathways. By initiating the chiral separation at the amino acid stage rather than the ester stage, the process fundamentally alters the impurity profile and yield trajectory. The methodology described ensures that raw materials are not only readily available but also inexpensive, addressing the critical need for supply chain resilience in the generic pharmaceutical sector. Furthermore, the mild reaction conditions and simplified operational steps suggest a robust platform for commercial scale-up, minimizing the technical risks associated with complex multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of clopidogrel has been plagued by significant technical and economic hurdles, primarily stemming from the choice of starting materials and the timing of chiral resolution. One prevalent legacy route involves the use of 4,5,6,7-tetramethylenethieno[3,2-c]pyridine as a key precursor. As illustrated in the reaction scheme below, this pathway suffers from the inherent difficulty in purifying this specific heterocyclic starting material, which drastically impacts the yield and purity of downstream products. The complexity of synthesizing and isolating this tetramethylene-fused system creates a bottleneck that limits throughput and increases production costs.
![Prior art synthesis route using 4,5,6,7-tetramethylenethieno[3,2-c]pyridine showing purification difficulties](/insights/img/clopidogrel-synthesis-d-camphorsulfonic-pharma-supplier-20260309132324-01.png)
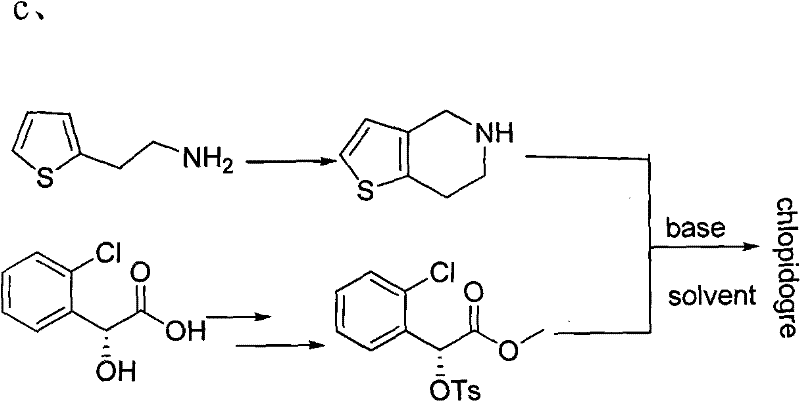
Another common historical approach relies on the condensation of o-chlorobenzaldehyde with 2-thiophene ethylamine and sodium cyanide. While chemically feasible, this route is increasingly untenable for modern industrial standards due to the reliance on sodium cyanide, a highly toxic reagent that necessitates rigorous safety protocols and expensive waste management systems. Moreover, in this sequence, the optical resolution step occurs relatively late in the synthesis, meaning that any loss of material during the resolution of the final ester significantly depresses the overall productive rate. The third major conventional pathway utilizes (R)-o-chloromandelic acid as a chiral pool starter. Although this avoids early resolution, the starting material itself is prohibitively expensive, rendering the process economically uncompetitive for large-scale generic manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101787032A proposes a streamlined sequence that begins with the resolution of DL-o-chlorobenzene glycine. This strategic pivot allows for the use of D-camphorsulfonic acid as a resolving agent in an aqueous medium, a process that is both safe and highly efficient. By securing the chiral integrity at the amino acid stage, the subsequent chemical transformations proceed with higher fidelity. The novel approach eliminates the need for toxic cyanides and expensive chiral pool acids, replacing them with commodity chemicals like 2-thiophene ethanol derivatives. This shift not only reduces the direct cost of goods sold (COGS) but also simplifies the regulatory burden associated with handling hazardous materials. The integration of a recyclable resolving agent further enhances the economic profile, as the mother liquor can be processed to recover both the unwanted enantiomer for racemization and the valuable camphorsulfonic acid for reuse.
Mechanistic Insights into Chiral Resolution and Mannich Cyclization
The core of this synthetic innovation lies in the precise control of stereochemistry during the initial salification step. The process involves reacting DL-o-chlorobenzene glycine with D-camphorsulfonic acid in water at temperatures ranging from 40°C to 70°C. This specific thermal window is critical for maximizing the solubility differential between the diastereomeric salts, allowing the desired (S)-(+)-enantiomer to crystallize out with high optical purity. Following filtration, the free base is liberated using alkaline purification, yielding (S)-(+)-o-chlorobenzene glycine with an enantiomeric excess (e.e.) that can reach 99% after recrystallization. Crucially, the mother liquor containing the (R)-enantiomer is not discarded; instead, it is subjected to racemization under strongly basic conditions at elevated temperatures (85°C). This closed-loop system ensures that no chiral material is wasted, theoretically allowing for a 100% conversion of the racemic starting material into the desired product over multiple cycles.
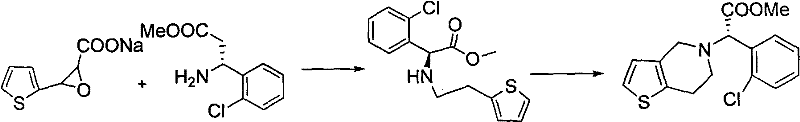
Following resolution, the mechanism proceeds through a nucleophilic substitution where the chiral amino acid attacks a 2-thiophene ethanol derivative, specifically the p-toluenesulfonate ester. This reaction is conducted in anhydrous acetonitrile in the presence of an alkali base, facilitating the formation of the C-N bond. A key mechanistic advantage here is the avoidance of ester hydrolysis, a common side reaction in prior art methods where resolution was performed at the ester stage. The resulting amino acid intermediate is then converted to its hydrochloride salt by feeding hydrogen chloride gas, which stabilizes the molecule and improves isolation yields. The final ring closure is achieved via a Mannich-type cyclization using formaldehyde or paraformaldehyde in an acidic or aqueous medium. This intramolecular cyclization constructs the tetrahydrothienopyridine core efficiently, preserving the stereochemical integrity established in the first step and delivering the clopidogrel base with high purity.
How to Synthesize Clopidogrel Efficiently
The synthesis of clopidogrel via this novel route requires strict adherence to temperature controls and stoichiometric ratios to ensure optimal yield and purity. The process begins with the preparation of the chiral amino acid, followed by coupling with the thiophene moiety, esterification, and final cyclization. Each step has been optimized to minimize byproduct formation and simplify workup procedures, such as using gas-phase salification to precipitate intermediates directly from solution. For detailed operational parameters, including specific solvent volumes, reaction times, and crystallization temperatures, please refer to the standardized protocol outlined below.
- Resolve DL-o-chlorobenzene glycine using D-camphorsulfonic acid in water at 40-70°C to obtain (S)-(+)-o-chlorobenzene glycine.
- Perform condensation with 2-thiophene ethanol derivative (tosylate) in acetonitrile with alkali, followed by salification with HCl gas to form the amino acid hydrochloride intermediate.
- Esterify the intermediate with thionyl chloride in methanol, then conduct Mannich cyclization in aqueous or acidic solvent to form the clopidogrel base, finally converting to bisulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic advantages beyond mere technical feasibility. The primary value driver is the substantial reduction in raw material costs achieved by substituting expensive chiral starters with inexpensive racemic glycine derivatives. By eliminating the dependency on specialized, high-cost building blocks like (R)-o-chloromandelic acid, manufacturers can significantly lower their exposure to volatile raw material markets. Furthermore, the removal of sodium cyanide from the process flow removes a major logistical and compliance hurdle, simplifying the sourcing of reagents and reducing the overhead associated with hazardous material storage and disposal. This translates directly into a more resilient supply chain capable of sustaining continuous production without the interruptions often caused by regulatory inspections or reagent shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the recyclability of the resolving agent and the high atom economy of the substitution reactions. By recovering D-camphorsulfonic acid from the mother liquor and recycling the unwanted (R)-enantiomer back into the process via racemization, the effective consumption of chiral auxiliaries is minimized. Additionally, the avoidance of complex purification steps required for tetramethylene-fused precursors reduces solvent usage and energy consumption. These factors combine to create a manufacturing cost structure that is significantly leaner than traditional methods, allowing for competitive pricing in the generic drug market without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as o-chlorobenzene glycine, thiophene ethanol, and formaldehyde ensures a stable and diversified supply base. Unlike proprietary intermediates that may be sourced from a single vendor, these raw materials are widely available from multiple global suppliers, mitigating the risk of supply disruption. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic catalysts or extreme pressures, further enhances reliability. This stability allows for long-term production planning and secure inventory management, crucial for meeting the demands of large-scale pharmaceutical contracts.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The absence of cyanide and the use of water as a solvent for the resolution step align with green chemistry principles, reducing the facility's environmental footprint. The simplified workup procedures, which often involve straightforward filtration and crystallization rather than complex chromatographic separations, facilitate easier scale-up from pilot plant to commercial tonnage. This scalability ensures that production can be ramped up quickly to meet market surges, while the cleaner process profile simplifies waste treatment and regulatory reporting, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clopidogrel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, purity profiles, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: Why is D-camphorsulfonic acid preferred for resolving o-chlorobenzene glycine in this process?
A: D-camphorsulfonic acid allows for efficient optical resolution in water with recyclable mother liquors. Unlike ester-based resolution methods, resolving the amino acid directly avoids the hydrolysis issues encountered when racemizing invalid enantiomers at the ester stage, significantly improving overall transformation efficiency.
Q: How does this method improve upon traditional sodium cyanide-based routes?
A: Traditional routes often utilize toxic sodium cyanide for Strecker synthesis, which poses severe safety and environmental challenges for industrial production. This novel method eliminates the need for cyanide entirely, utilizing a safer nucleophilic substitution with 2-thiophene ethanol derivatives, thereby enhancing operational safety and reducing waste treatment costs.
Q: What are the purity specifications achievable with this synthetic route?
A: The patent data indicates that through careful crystallization and purification steps, particularly during the resolution and final salt formation, the process yields intermediates with purity exceeding 99% and enantiomeric excess (e.e.) values reaching 99%, ensuring the final clopidogrel bisulfate meets stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101787032A can be realized in a GMP-compliant environment. We maintain stringent purity specifications across all our lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify enantiomeric excess and impurity profiles at every stage. Our commitment to quality ensures that every batch of clopidogrel intermediate meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security in your pharmaceutical manufacturing operations.
