Optimizing HIV Protease Inhibitor Intermediate Production for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex antiretroviral agents, particularly those targeting Human Immunodeficiency Virus (HIV) protease enzymes. Patent CN1066723C introduces a groundbreaking methodology for preparing critical intermediates used in the synthesis of HIV protease inhibitors, specifically compounds like L-735,524. This intellectual property outlines a novel chemical strategy that reacts glycidol or its activated derivatives with specific amide enolates to generate epoxide intermediates with high efficiency. For R&D Directors and Procurement Managers overseeing the supply of high-purity pharmaceutical intermediates, understanding this technological shift is vital. The disclosed process addresses long-standing challenges in stereoselectivity and reagent toxicity, offering a more viable route for commercial manufacturing. By leveraging this advanced chemistry, manufacturers can secure a more stable supply chain for essential antiretroviral components while adhering to stringent environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of building blocks for HIV protease inhibitors has been plagued by excessive complexity and reliance on hazardous materials. Traditional routes often necessitate up to twelve distinct reaction steps, creating a logistical nightmare for supply chain heads concerned with yield loss and timeline extension. A significant bottleneck in prior art involves the use of osmium tetroxide, a highly toxic oxidizing agent that requires specialized handling and extensive waste remediation protocols. Furthermore, conventional methods frequently depend on expensive coupling reagents such as BOC-ON and EDC, which drastically inflate the cost of goods sold. The cumulative effect of these factors is a fragile production process where minor deviations can lead to substantial batch failures. For procurement teams, this translates to volatile pricing and potential supply disruptions, making the sourcing of these critical pharmaceutical intermediates a high-risk endeavor that demands urgent optimization.
The Novel Approach
In stark contrast, the patented methodology streamlines the synthetic route by directly introducing the necessary three-carbon unit through a highly selective alkylation process. This innovative approach bypasses the need for multiple protection and deprotection cycles, effectively collapsing the reaction sequence into fewer, more manageable steps. By utilizing activated glycidyl derivatives, such as glycidyl tosylate, the process achieves direct coupling with amide enolates under controlled conditions. This not only eliminates the requirement for toxic osmium reagents but also removes the dependency on costly coupling agents. The result is a cleaner reaction profile with improved overall throughput. For stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing, this represents a paradigm shift towards sustainability and economic efficiency. The simplified workflow ensures that commercial scale-up of complex polymer additives and drug precursors becomes more predictable and financially viable.
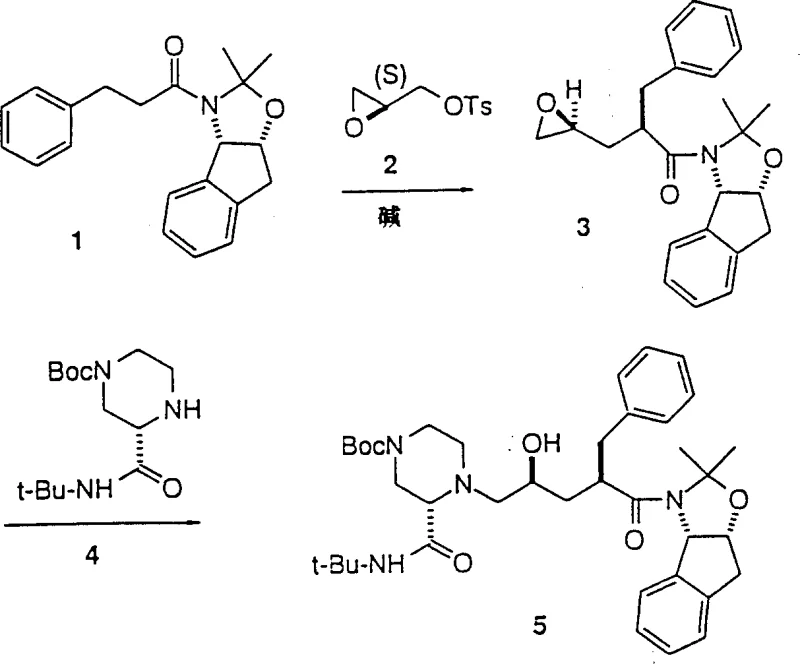
Mechanistic Insights into Amide Enolate Alkylation
The core of this technological advancement lies in the precise generation and reaction of stabilized amide enolates. The process begins with the metalation of a specific amide substrate using a strong base, such as lithium diisopropylamide (LDA) or lithium bis(trimethylsilyl)amide, at cryogenic temperatures ranging from -50°C to -45°C. This low-temperature environment is crucial for maintaining the integrity of the enolate and preventing unwanted side reactions. Once formed, the nucleophilic enolate attacks the terminal position of the activated glycidyl derivative, leading to the opening of the epoxide ring and the formation of a new carbon-carbon bond. This step creates a new asymmetric center with high stereochemical fidelity, which is paramount for the biological activity of the final HIV inhibitor. The ability to control this stereocenter without resorting to complex resolution techniques post-reaction is a significant mechanistic advantage that ensures high optical purity from the outset.
Furthermore, the mechanism inherently suppresses the formation of impurities that typically arise from double addition or polymerization of the epoxide species. In many conventional scenarios, nucleophilic attack on epoxides can lead to oligomerization, reducing yield and complicating purification. However, the specific electronic properties of the amide enolate, combined with the activation of the glycidyl leaving group, favor a single, clean substitution event. This selectivity minimizes the burden on downstream purification processes, such as chromatography or crystallization, thereby enhancing the overall mass balance of the operation. For quality assurance teams, this means a more consistent impurity profile and reduced risk of genotoxic impurities carrying over into the final active pharmaceutical ingredient. The robustness of this mechanistic pathway provides a solid foundation for reliable high-purity pharmaceutical intermediate production.
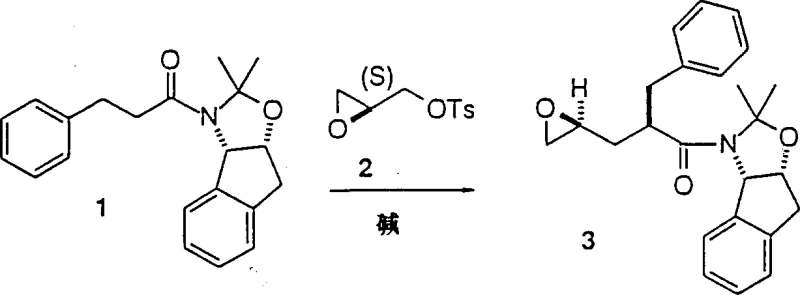
How to Synthesize L-735,524 Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction conditions and reagent quality to maximize the benefits of the patented route. The process is designed to be scalable, moving seamlessly from laboratory benchtop to industrial reactors without losing efficiency. Operators must ensure strict temperature control during the enolate formation phase to prevent decomposition. Following the coupling reaction, standard workup procedures involving aqueous quenching and organic extraction are sufficient to isolate the crude product, which can then be purified via crystallization. This operational simplicity is a key driver for adoption in contract development and manufacturing organizations. Detailed standardized synthesis steps see the guide below.
- Prepare the amide substrate by reacting cis-1-amino-2,3-indanol with 3-phenylpropionyl chloride followed by ketal protection.
- Generate the amide enolate using a strong base like LDA or LiN(TMS)2 at low temperatures between -50°C to -45°C in THF.
- React the enolate with activated glycidyl derivatives such as glycidyl tosylate to form the epoxide intermediate with high diastereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis offers profound benefits for procurement managers and supply chain directors aiming to optimize their vendor networks. The primary advantage stems from the drastic simplification of the chemical process, which directly correlates to reduced operational expenditures. By eliminating multiple reaction steps and removing the need for exotic or hazardous reagents, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on quality. Additionally, the reduced complexity lowers the barrier for entry for qualified manufacturers, increasing the pool of potential suppliers and enhancing supply chain resilience against geopolitical or logistical disruptions.
- Cost Reduction in Manufacturing: The elimination of toxic osmium tetroxide and expensive coupling reagents like EDC removes significant cost centers from the production budget. Without the need for specialized containment systems for highly toxic materials, capital expenditure on facility infrastructure is also lowered. Furthermore, the higher yield associated with fewer reaction steps means less raw material is wasted per kilogram of final product. These factors combine to create a leaner manufacturing model that drives down the unit cost of the intermediate. This economic efficiency is critical for maintaining margins in the competitive generic pharmaceutical market while ensuring long-term viability for the production line.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as simple amides and glycidyl derivatives, mitigates the risk of raw material shortages. Unlike specialized chiral catalysts that may have limited global suppliers, the reagents used in this process are commodity chemicals with robust supply networks. This availability ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates. For supply chain heads, this reliability translates to better inventory management and the ability to respond quickly to fluctuations in market demand. It fosters a more stable partnership between the chemical manufacturer and the downstream pharmaceutical client.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing common solvents like tetrahydrofuran and ethyl acetate which are easily recovered and recycled. The absence of heavy metal contaminants simplifies the environmental compliance landscape, reducing the regulatory burden associated with effluent treatment. This green chemistry aspect is increasingly important for multinational corporations aiming to meet sustainability goals. Easier scale-up means that transitioning from pilot plant to commercial tonnage production can be achieved with minimal technical risk. This scalability ensures that the supply can grow in tandem with the clinical and commercial success of the final HIV protease inhibitor drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in the patent documentation. Understanding these nuances helps stakeholders make informed decisions about integrating this intermediate into their supply chains. The answers reflect the consensus on best practices for handling these chemical transformations safely and efficiently.
Q: How does this new method improve upon conventional HIV inhibitor synthesis?
A: The patented method significantly reduces the number of reaction steps compared to traditional twelve-step processes, eliminating the need for toxic osmium tetroxide and expensive coupling reagents.
Q: What are the key advantages for large-scale manufacturing of these intermediates?
A: The process utilizes readily available starting materials and avoids hazardous reagents, which simplifies waste treatment and enhances operational safety during commercial scale-up.
Q: Is the stereochemical control reliable for pharmaceutical grade production?
A: Yes, the method employs specific chiral amide enolates and activated glycidyl derivatives to achieve high diastereoselectivity, ensuring consistent purity profiles required for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-735,524 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for antiretroviral therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at implementing complex chemistries like the amide enolate alkylation described in CN1066723C, ensuring that every batch meets stringent purity specifications. We operate rigorous QC labs equipped to verify stereochemical integrity and impurity profiles, guaranteeing that our clients receive materials ready for immediate downstream processing. Our commitment to excellence makes us a trusted ally in the global fight against HIV.
We invite pharmaceutical partners to collaborate with us to leverage these technological advancements for their product pipelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain security. Let us help you optimize your production of high-purity pharmaceutical intermediates with confidence and precision.
