Advanced Regiospecific Synthesis of cis-1-Amino-2-Indanol for Commercial HIV Protease Inhibitor Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving as the backbone for antiretroviral therapies. Patent CN1083425C introduces a groundbreaking regiospecific process for the synthesis of cis-1-amino-2-indanol, a pivotal chiral building block used in the manufacture of HIV protease inhibitors such as Compound J. This innovation addresses long-standing challenges in stereochemical control and process efficiency by transforming a historically cumbersome 12-step sequence into a streamlined, high-yielding operation. By leveraging specific acid-catalyzed conditions in nitrile solvents, the method achieves exceptional preservation of stereochemical integrity at the C-2 position of the indene oxide starting material. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize supply chains for antiviral medications, reducing both the environmental footprint and the overall cost of goods sold through simplified chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1-amino-2-indanols relied on inefficient multi-step sequences that plagued manufacturing scalability and cost-effectiveness. Traditional routes typically involved treating indene oxide with ammonia water to generate trans-1-amino-2-indanol, which is the undesired stereoisomer for many protease inhibitor applications. To correct this stereochemistry, chemists were forced to undertake a laborious series of transformations including acylation with acid halides to form amide intermediates, activation of the hydroxyl group via mesylation, and subsequent induced cyclization to form oxazoline derivatives. This convoluted pathway not only consumed excessive amounts of reagents and solvents but also necessitated the isolation and purification of multiple unstable intermediates, leading to significant yield losses and extended production lead times that strained global supply chains.
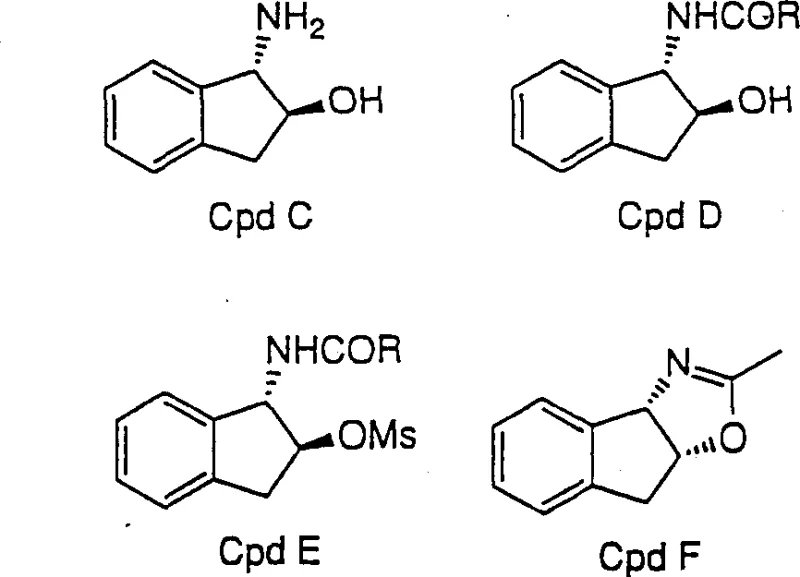
The Novel Approach
In stark contrast to prior art, the disclosed invention utilizes a direct, regiospecific ring-opening strategy that bypasses the formation of trans-isomers entirely. By treating indene oxide with strong acids in the presence of nitrile solvents, the process facilitates the formation of an oxazoline intermediate in situ, which is subsequently hydrolyzed to yield the desired cis-1-amino-2-indanol directly. This one-pot methodology eliminates the need for isolating intermediate species, thereby drastically reducing unit operations and handling time. The ability to proceed from the epoxide starting material to the final amino alcohol in essentially two chemical steps (ring opening and hydrolysis) without intermediate workups represents a paradigm shift in process chemistry, offering a cleaner, faster, and more economically viable pathway for producing high-purity pharmaceutical intermediates.
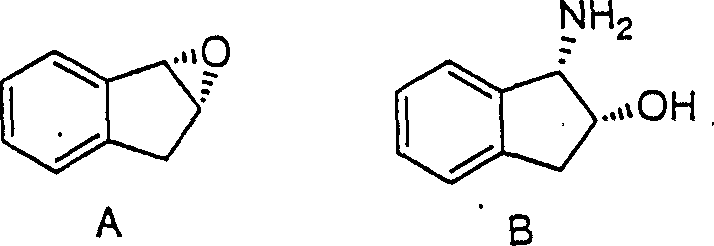
Mechanistic Insights into Acid-Catalyzed Regiospecific Ring Opening
The core mechanistic advantage of this process lies in its ability to maintain the stereochemical integrity of the carbon-oxygen bond at the C-2 position during the ring-opening event. When indene oxide is exposed to strong protic acids like sulfuric acid or methanesulfonic acid in a nitrile solvent such as acetonitrile, the epoxide ring is activated towards nucleophilic attack. The nitrile solvent acts as an internal nucleophile, attacking the benzylic position to form a nitrilium ion species which rapidly cyclizes to form the oxazoline ring system. Crucially, this transformation occurs with inversion of configuration at the site of attack while retaining the stereochemistry at the adjacent carbon, ensuring that the spatial arrangement of substituents aligns perfectly with the requirements for the downstream cis-configuration. This precise control prevents the scrambling of chiral centers that often plagues acid-catalyzed epoxide openings.
Following the formation of the oxazoline intermediate, the addition of excess water triggers a hydrolysis step that cleaves the heterocyclic ring to reveal the free amine and alcohol functionalities. This hydrolysis is conducted at elevated temperatures ranging from 25°C to 100°C, ensuring complete conversion of the oxazoline to the target amino alcohol. The reaction conditions are carefully tuned to avoid racemization, with the patent specifying that mixtures of epoxide enantiomers yield corresponding mixtures of amino alcohol enantiomers with high fidelity. For quality control teams, this mechanism implies a highly predictable impurity profile, as the regiospecificity minimizes the formation of regioisomeric byproducts that would otherwise require costly chromatographic separation, thus enhancing the overall purity of the crude product before final crystallization.
How to Synthesize cis-1-Amino-2-Indanol Efficiently
The implementation of this regiospecific synthesis requires careful attention to solvent selection and temperature control to maximize yield and enantiomeric excess. The process begins by dissolving the indene oxide starting material in a suitable nitrile solvent, with acetonitrile being the preferred choice due to its balance of polarity and reactivity. An acid catalyst, such as methanesulfonic acid or oleum, is then introduced under controlled thermal conditions to initiate the ring opening. Following the formation of the intermediate, aqueous hydrolysis completes the transformation. The detailed standardized synthetic steps for replicating this high-efficiency protocol are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- Dissolve indene oxide in an alkyl or aryl nitrile solvent such as acetonitrile, optionally with a co-solvent.
- Add approximately 2 equivalents of a strong protic acid, Lewis acid, or organic acid while maintaining temperature between -70°C and +30°C.
- Hydrolyze the resulting mixture with excess water at temperatures between 25°C and 100°C to yield cis-1-amino-2-indanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere chemical elegance. The reduction in step count from twelve to essentially two operational stages translates directly into a significantly reduced capital expenditure on reactor time and a drastic simplification of logistics. By eliminating the need to isolate and store hazardous intermediates like mesylates or acid halides, facilities can lower their inventory holding costs and reduce the regulatory burden associated with handling reactive chemical species. Furthermore, the use of commodity chemicals such as acetonitrile and sulfuric acid ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from volatility associated with exotic or proprietary reagents often required in older synthetic methodologies.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps serves as a primary driver for cost optimization, as it removes the need for multiple filtration, drying, and purification cycles that consume energy and labor. By performing the reaction in a telescoped manner, manufacturers can achieve substantial cost savings through reduced solvent volumes and decreased waste disposal fees, as the overall mass intensity of the process is lowered. Additionally, the higher overall yield reported in the patent examples means that less starting material is required to produce the same amount of final API intermediate, further driving down the variable cost per kilogram and improving the gross margin profile for the finished pharmaceutical product.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently increases supply chain resilience by reducing the number of potential failure points where batch rejection could occur. With fewer unit operations, there is less opportunity for human error or equipment malfunction to disrupt production schedules, leading to more consistent on-time delivery performance for downstream drug manufacturers. The robustness of the chemistry, which tolerates a range of temperatures and uses stable reagents, ensures that production can be maintained even under varying operational conditions, providing a reliable source of high-purity pharmaceutical intermediates that keeps clinical and commercial timelines on track.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to large-scale industrial reactors without significant re-engineering. The reduction in solvent usage and the avoidance of heavy metal catalysts or toxic reagents align with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces the risk of regulatory fines but also enhances the corporate sustainability profile of the manufacturing entity, appealing to eco-conscious partners and investors who prioritize responsible chemical production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this regiospecific process. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy for technical decision-makers evaluating this technology for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this route into existing manufacturing frameworks.
Q: How does this new process improve stereochemical integrity compared to conventional methods?
A: The novel process substantially maintains the stereochemical integrity of the carbon-oxygen bond at the C-2 position of the indene oxide starting material, ensuring high enantiomeric purity in the final cis-1-amino-2-indanol product without requiring complex resolution steps early in the synthesis.
Q: What are the primary cost drivers eliminated by this regiospecific method?
A: By converting a previously 12-step sequence into a concise one-pot reaction that avoids the isolation of intermediates like oxazolines, the method drastically reduces solvent consumption, labor hours, and purification costs associated with multi-step batch processing.
Q: Can this process be scaled for commercial API intermediate production?
A: Yes, the process utilizes common industrial solvents like acetonitrile and standard acids such as methanesulfonic acid or sulfuric acid, operating within manageable temperature ranges (-70°C to +100°C), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-1-Amino-2-Indanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV and other viral diseases. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of cis-1-amino-2-indanol meets the exacting standards required for GMP API synthesis. We are committed to delivering not just a chemical product, but a reliable partnership that supports your long-term development goals.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this regiospecific method for your supply chain. We encourage potential partners to contact us today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your pharmaceutical manufacturing operations.
