Advanced Regiospecific Synthesis of Cis-1-Amino-2-Indanol for High-Purity HIV Protease Inhibitors
Introduction to Regiospecific Amination Technology
The pharmaceutical industry constantly seeks more efficient pathways to synthesize complex chiral intermediates, particularly those serving as the backbone for potent antiviral therapies. Patent CN1077881C introduces a groundbreaking regiospecific process for synthesizing cis-1-amino-2-alkanol derivatives, specifically focusing on cis-1-amino-2-indanol, a critical scaffold for HIV protease inhibitors. This technology represents a paradigm shift from traditional multi-step syntheses, offering a direct route from readily available 1,2-indanediol or 2-halo-1-indanol precursors. By leveraging acid-catalyzed rearrangement in nitrile solvents, this method achieves high regioselectivity and stereochemical fidelity, addressing long-standing challenges in the manufacturing of oligopeptide analogue inhibitors. For R&D directors and procurement specialists, understanding this proprietary chemistry is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with improved economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cis-1-amino-2-indanol and related compounds has been plagued by inefficiency and complexity. Prior art methods, such as those described in earlier European patent applications, often necessitated a cumbersome 12-step sequence to reach the target molecule. These legacy routes typically involved the treatment of indene oxide with ammonia, which predominantly yielded the undesired trans-1-amino-2-indanol isomer, requiring additional steps to invert stereochemistry or resolve racemic mixtures. Furthermore, these conventional pathways relied heavily on expensive starting materials and reagents, coupled with numerous isolation and purification stages that drastically reduced overall yield. The accumulation of impurities across such a long synthetic chain posed significant risks to final drug quality, while the extensive use of solvents and reagents generated substantial chemical waste, creating environmental and disposal burdens that are increasingly untenable in modern green chemistry frameworks.
The Novel Approach
In stark contrast, the novel regiospecific process detailed in the patent data streamlines production into a concise, high-yielding operation. The core innovation lies in the direct conversion of 1,2-indanediols or 2-halo-1-indanols into the desired cis-amino alcohol using a strong acid catalyst in a nitrile solvent. This approach effectively bypasses the need for separate amination and stereochemical inversion steps by integrating them into a single pot reaction sequence. As illustrated in the reaction scheme below, the process allows for the precise manipulation of stereochemistry based on the choice of starting material: diol precursors retain configuration at the C-2 position, while halohydrin precursors undergo specific inversion. This flexibility enables manufacturers to access specific enantiomers, such as 1R-amino-2S-indanol or 1S-amino-2R-indanol, directly from the corresponding chiral pool starting materials without the need for costly chiral chromatography or resolution.
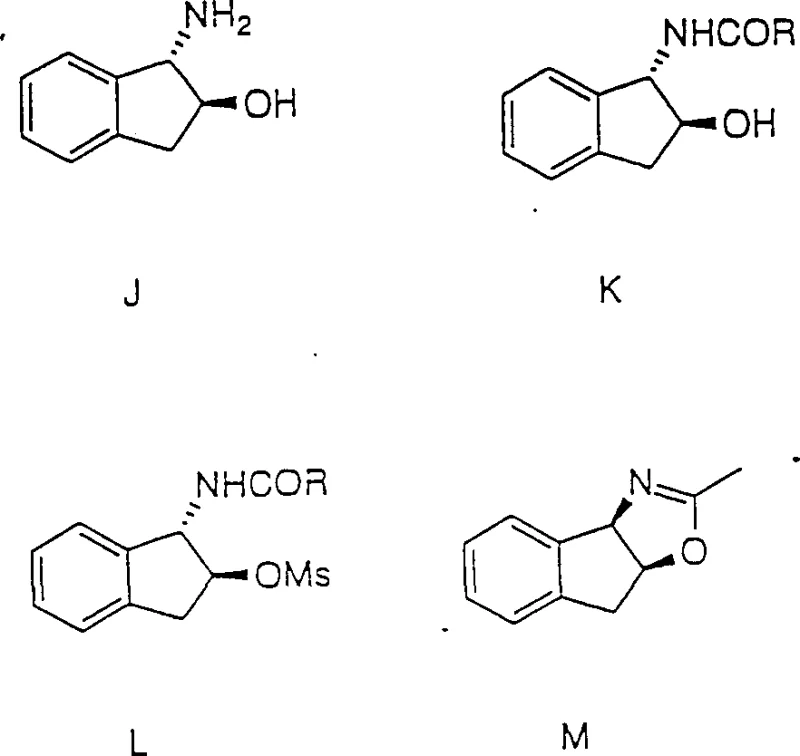
The operational simplicity of this new route cannot be overstated. By conducting the reaction in solvents like acetonitrile, which also serves as the nitrogen source for the amine group via a Ritter-type mechanism, the process eliminates the need for external ammonia sources or hazardous azide reagents. The reaction conditions are robust, tolerating temperatures ranging from -70°C to 30°C during the initial activation phase, followed by hydrolysis at moderate temperatures between 25°C and 80°C. This broad operating window facilitates easier thermal management on a commercial scale, reducing the risk of runaway reactions and ensuring consistent batch-to-batch quality. Ultimately, this novel approach transforms a previously arduous synthesis into a commercially viable, cost-effective process suitable for the demands of the global antiviral market.
Mechanistic Insights into Acid-Catalyzed Regiospecific Rearrangement
At the heart of this technology is a sophisticated mechanistic pathway that ensures high regioselectivity and stereochemical control. When a 1,2-indanediol precursor is treated with a strong protic acid, such as sulfuric acid or methanesulfonic acid, or a Lewis acid like boron trifluoride, one of the hydroxyl groups is protonated and leaves as water, generating a benzylic carbocation intermediate. In the presence of a nitrile solvent, this electrophilic center is immediately trapped by the nitrogen lone pair of the nitrile, forming a nitrilium ion. This step is crucial as it establishes the carbon-nitrogen bond at the specific regiochemical position required for the final amino alcohol. Subsequent hydrolysis of the nitrilium intermediate with water converts the nitrile group into the primary amine, completing the transformation. The beauty of this mechanism lies in its ability to preserve the stereochemical integrity of the adjacent carbon center when starting from diols, as the reaction proceeds with retention of configuration at the non-reacting stereocenter.
For R&D teams focused on impurity control, understanding the divergence in mechanism when using halohydrin precursors is vital. In the case of 2-halo-1-indanols, the presence of the halogen atom alters the reaction trajectory. The acid-catalyzed displacement of the halogen or the participation of the neighboring hydroxyl group leads to an intermediate that undergoes inversion of configuration at the C-2 position. This stereospecific inversion is a powerful tool for accessing the complementary enantiomer of the target amino alcohol without changing the fundamental reagents. Furthermore, the patent data highlights that the final product can be isolated directly as a crystalline free base or as a stable amine salt derivative, such as a tartrate, simply by adjusting the pH of the reaction medium. This inherent ability to crystallize the product directly from the crude mixture significantly reduces the burden on downstream purification processes, minimizing the carryover of trace impurities and ensuring the high-purity pharmaceutical intermediate standards required for clinical applications.
How to Synthesize Cis-1-Amino-2-Indanol Efficiently
The practical implementation of this regiospecific synthesis is designed for scalability and ease of execution in a standard chemical manufacturing facility. The process begins with the dissolution of the chosen precursor, either a 1,2-indanediol or a 2-halo-1-indanol, in a nitrile solvent such as acetonitrile or benzonitrile. To initiate the rearrangement, a stoichiometric excess of acid is added under controlled temperature conditions, typically maintaining the mixture between -70°C and 30°C to manage the exotherm and prevent side reactions. Following the formation of the intermediate, water is introduced to effect hydrolysis, converting the nitrile adduct into the final amine. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- Dissolve 1 equivalent of 1,2-indanediol or 2-halo-1-indanol in a suitable alkyl or aryl nitrile solvent such as acetonitrile.
- Add at least 1.5 equivalents of a strong protic acid, Lewis acid, or organic acid while maintaining the temperature between -70°C and 30°C for 0.25 to 6 hours.
- Quench the reaction with excess water and stir at 25°C to 80°C for 0.5 to 8 hours to effect hydrolysis and isolate the cis-1-amino-2-indanol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this regiospecific process offers tangible strategic benefits that extend beyond simple chemistry. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like indanediols and sulfuric acid, manufacturers can decouple their production from the volatility of specialized reagent markets. This shift not only stabilizes costs but also enhances supply continuity, as the reliance on obscure or single-source intermediates is eliminated. The reduction in synthetic steps from twelve to just a few key operations means that the overall throughput time is significantly compressed, allowing for faster response to market demand fluctuations and shorter lead times for high-purity amino alcohols.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the elimination of expensive reagents and the reduction of unit operations. Traditional routes required multiple protection and deprotection cycles, each adding cost in terms of materials, labor, and waste disposal. By consolidating these into a single acid-catalyzed step, the new method substantially lowers the cost of goods sold. Additionally, the use of acetonitrile as both solvent and reagent reduces the total volume of organic solvents required, leading to lower procurement costs for solvents and reduced expenses associated with solvent recovery and waste treatment. The ability to isolate the product via simple crystallization further cuts down on energy-intensive distillation and chromatography steps, resulting in significant operational savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of starting materials. The process tolerates a wide range of temperatures and uses stable, non-hazardous acids that are easily sourced globally. This reduces the risk of production delays caused by reagent shortages or strict regulatory controls on hazardous materials. Furthermore, the high yield and selectivity of the reaction mean that less starting material is needed to produce the same amount of final product, effectively increasing the capacity of existing manufacturing assets. This efficiency allows suppliers to maintain larger safety stocks and meet urgent orders without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles. The reduction in step count inherently reduces the E-factor (mass of waste per mass of product), minimizing the environmental footprint of the manufacturing process. The absence of heavy metal catalysts or toxic azide reagents simplifies waste stream management and reduces the regulatory burden associated with hazardous waste disposal. Scalability is ensured by the use of standard batch reactor operations and straightforward workup procedures, making the transition from laboratory scale to commercial scale-up of complex pharmaceutical intermediates seamless and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this regiospecific synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this process control stereochemistry compared to traditional methods?
A: Unlike conventional methods that often yield mixtures or require resolution, this regiospecific process allows for precise stereochemical control. Using 1,2-indanediol precursors retains the stereochemical integrity at the C-2 position, while 2-halo-1-indanol precursors allow for specific inversion, enabling the targeted synthesis of either the 1R,2S or 1S,2R enantiomer directly.
Q: What are the primary cost drivers eliminated in this new synthetic route?
A: The primary cost reductions come from eliminating the lengthy 12-step sequence previously required. By utilizing a direct acid-catalyzed rearrangement in a nitrile solvent, the process removes the need for multiple protection/deprotection steps, expensive reagents for amine introduction, and complex purification of intermediates, significantly lowering the overall cost of goods.
Q: Is this process scalable for commercial API production?
A: Yes, the process is highly scalable. It utilizes common industrial solvents like acetonitrile and standard mineral acids like sulfuric acid. The workup involves straightforward hydrolysis and crystallization of salt derivatives, which are unit operations well-suited for large-scale manufacturing environments ranging from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-1-Amino-2-Indanol Supplier
As the global demand for advanced antiviral therapeutics continues to grow, the need for efficient and scalable synthesis of key intermediates like cis-1-amino-2-indanol has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging the breakthroughs described in patent CN1077881C to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major pharmaceutical companies. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the highest standards for safety and efficacy required for API synthesis.
We invite you to explore how our optimized manufacturing processes can drive value for your organization. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific supply chain needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our regiospecific synthesis can reduce your overall production costs and improve supply security. Contact us today to discuss your requirements and discover how NINGBO INNO PHARMCHEM can become your trusted partner in the development and supply of high-quality pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
