Advanced Synthesis of 1,4-bis(2,4-diaminophenoxy)benzene for High-Performance Polyimide Applications
The rapid evolution of the flexible electronics sector has placed unprecedented demands on the thermal and mechanical stability of substrate materials, driving intense research into advanced polyimide formulations. At the heart of this innovation lies the critical need for high-purity aromatic diamine monomers, specifically 1,4-bis(2,4-diaminophenoxy)benzene, which serves as a foundational building block for next-generation Flexible Copper Clad Laminates (FCCL). Patent CN101215241A introduces a groundbreaking preparation method that addresses the historical scarcity of efficient synthetic routes for this valuable intermediate. By leveraging a catalytic transfer hydrogenation strategy using hydrazine hydrate and palladium on carbon, this technology offers a streamlined pathway that bypasses the complexities of traditional high-pressure hydrogenation. For R&D directors and procurement strategists alike, understanding the nuances of this patent is essential for securing a reliable electronic chemical supplier capable of meeting the rigorous specifications of modern optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of polynitro aromatic compounds to their corresponding polyamines has been fraught with significant engineering and economic challenges that hinder scalable manufacturing. Traditional catalytic hydrogenation often necessitates the use of high-pressure reactors operating under dangerous conditions, requiring substantial capital investment in safety infrastructure and specialized maintenance protocols. Furthermore, alternative chemical reduction methods, such as those employing iron powder in acidic media, generate massive quantities of toxic sludge and wastewater, creating severe environmental compliance burdens and escalating waste disposal costs. These conventional approaches frequently suffer from poor selectivity, leading to the formation of azo or azoxy by-products that are notoriously difficult to separate, thereby compromising the final purity required for high-performance polymer synthesis. The inability to efficiently isolate the target diamine without extensive purification steps has long been a bottleneck, limiting the availability of high-quality 1,4-bis(2,4-diaminophenoxy)benzene for commercial scale-up of complex polymer additives.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN101215241A utilizes a mild yet highly effective hydrazine hydrate reduction system that operates at atmospheric pressure and moderate temperatures ranging from 70°C to 85°C. This novel approach eliminates the need for hazardous high-pressure hydrogen gas, drastically simplifying the reactor design and enhancing overall operational safety for plant personnel. The process employs a versatile alcoholic solvent system, including options like ethylene glycol or ethanol, which facilitates excellent solubility of the nitro precursor while allowing for the direct crystallization of the amine product upon cooling. By integrating the reduction and isolation steps so seamlessly, the method removes the necessity for subsequent refining or chromatographic purification, which traditionally accounts for a significant portion of manufacturing time and expense. This streamlined workflow not only accelerates production cycles but also ensures a consistent supply of high-purity material, positioning it as a superior choice for cost reduction in polyimide manufacturing.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic breakthrough relies on the efficient transfer of hydrogen atoms from hydrazine to the nitro groups, mediated by the palladium on carbon catalyst surface. In this mechanism, hydrazine acts as a hydrogen donor, decomposing on the active palladium sites to generate nascent hydrogen species that are immediately available for the reduction of the nitro functionalities. The reaction proceeds through a series of intermediate stages, converting the nitro groups first to nitroso and then to hydroxylamine derivatives before finally arriving at the stable primary amine state. The choice of Pd/C is critical, as the carbon support provides a high surface area that maximizes catalyst exposure while preventing the aggregation of palladium particles, ensuring sustained catalytic activity throughout the 1 to 5 hour reaction window. This catalytic cycle is remarkably clean, with nitrogen gas being the primary by-product of hydrazine oxidation, which harmlessly vents from the system without contaminating the reaction mixture.
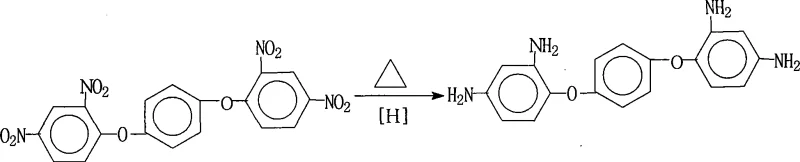
Controlling the impurity profile in such a multi-functional reduction is paramount, especially when the target molecule is destined for electronic grade polymers where trace metal ions or organic contaminants can degrade dielectric properties. The patented process achieves exceptional impurity control by optimizing the molar ratio of hydrazine to substrate between 1:20 and 1:50, ensuring a vast excess of reducing agent that drives the reaction to completion and prevents the accumulation of partially reduced intermediates. Furthermore, the hot filtration step performed immediately after the reaction effectively removes the solid Pd/C catalyst, preventing any potential leaching of palladium into the final product which could act as a pro-oxidant in the final polyimide resin. The subsequent cooling of the mother liquor induces selective crystallization, where the high symmetry and strong intermolecular hydrogen bonding of the diamine product favor its precipitation over any minor soluble by-products. This intrinsic self-purification mechanism is what allows the process to consistently deliver purity levels exceeding 99% without additional downstream processing.
How to Synthesize 1,4-bis(2,4-diaminophenoxy)benzene Efficiently
Implementing this synthesis route requires precise attention to the addition rate of the reducing agent and the maintenance of the specified temperature profile to ensure optimal kinetics and safety. The process begins by suspending the palladium catalyst in the chosen alcoholic solvent, followed by the addition of the dinitro precursor, creating a homogeneous reaction environment prior to the introduction of hydrazine. Operators must carefully monitor the exothermic nature of the hydrazine decomposition during the addition phase, utilizing the 60°C pre-heating step to initiate the reaction gently before ramping to the optimal 70°C to 85°C range. Detailed standard operating procedures regarding the specific weight ratios of catalyst to substrate and the concentration of hydrazine hydrate are critical for reproducibility. For a comprehensive breakdown of the exact experimental parameters and stoichiometry required for successful execution, please refer to the standardized synthesis guide below.
- Charge the reactor with 1,4-bis(2,4-dinitrophenoxy)benzene, Pd/C catalyst (1%-15% Pd content), and an alcoholic solvent such as ethylene glycol or ethanol.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60%-85% concentration) while maintaining stirring.
- Maintain the reaction temperature between 70°C and 85°C for 1 to 5 hours, then filter hot to remove the catalyst.
- Cool the filtrate to precipitate the crystal product, filter, and vacuum dry to obtain high-purity 1,4-bis(2,4-diaminophenoxy)benzene.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this hydrazine-based reduction technology offers profound advantages that extend far beyond simple chemical yield, fundamentally altering the cost structure of producing high-performance polyimide precursors. By eliminating the requirement for high-pressure hydrogenation infrastructure, manufacturers can significantly reduce capital expenditure and lower the barrier to entry for producing this specialized intermediate, fostering a more competitive and resilient supply market. The simplicity of the work-up procedure, which relies on basic filtration and crystallization rather than energy-intensive distillation or complex extraction sequences, translates directly into reduced utility consumption and shorter batch cycle times. These operational efficiencies allow suppliers to respond more agilely to fluctuating market demands, ensuring reducing lead time for high-purity electronic chemicals even during periods of peak industry activity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive purification steps and the use of readily available, commodity-grade reagents like hydrazine hydrate and common alcohols. Unlike methods that require precious metal catalysts in homogeneous forms which are difficult to recover, the heterogeneous Pd/C catalyst used here can be easily filtered off, and while the patent focuses on the reaction, industrial practice often allows for catalyst regeneration or metal recovery, further driving down the cost per kilogram. The high yield reported in the examples, consistently hovering near quantitative conversion, minimizes raw material waste, ensuring that the maximum amount of expensive nitro precursor is converted into saleable product. This efficiency creates a robust margin structure that can withstand volatility in raw material pricing, offering stable pricing models for long-term procurement contracts.
- Enhanced Supply Chain Reliability: The reliance on non-proprietary, bulk chemical inputs such as ethanol, ethylene glycol, and hydrazine ensures that production is not held hostage by the supply constraints of exotic reagents. Since the reaction conditions are mild and do not require specialized high-pressure vessels, the process can be easily transferred between different manufacturing sites or scaled up in existing multipurpose reactors without major retrofitting. This flexibility enhances supply chain continuity, mitigating the risk of production stoppages due to equipment failure or regulatory bottlenecks associated with high-pressure operations. Furthermore, the solid nature of the final product facilitates easy storage and transportation, reducing logistical complexities compared to liquid intermediates that may require stabilization or temperature-controlled shipping.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the environmental profile of a chemical process becomes a key determinant of its long-term viability. This method generates minimal waste, primarily consisting of nitrogen gas and water, avoiding the heavy metal sludge associated with iron reductions or the solvent-heavy waste streams of other organic reductions. The absence of chlorinated solvents or harsh acidic conditions simplifies wastewater treatment protocols, lowering the operational cost of environmental compliance. The straightforward scalability from gram-scale laboratory experiments to multi-ton industrial batches is evidenced by the patent's explicit claim of suitability for industrial production, giving supply chain heads confidence in the ability to ramp up volumes rapidly to meet the growing demand for FCCL and advanced composite materials.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their formulation pipelines, we have compiled answers to the most common inquiries regarding the technical specifications and handling of this material. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring that all information is grounded in verified scientific results. Understanding these details is crucial for quality assurance teams who need to validate the material against internal specifications for electronic grade applications. We encourage stakeholders to review these points closely to appreciate the depth of process control embedded in this manufacturing route.
Q: What is the achievable purity of 1,4-bis(2,4-diaminophenoxy)benzene using this method?
A: According to patent CN101215241A, the described process consistently yields products with a purity exceeding 99%, often reaching up to 99.8% without the need for complex recrystallization steps.
Q: Why is hydrazine hydrate preferred over catalytic hydrogenation for this reduction?
A: Hydrazine hydrate serves as a convenient liquid reducing agent that eliminates the need for high-pressure hydrogen gas equipment, significantly simplifying the operational safety profile and reducing capital expenditure for reactor infrastructure.
Q: Is this process suitable for large-scale industrial production of polyimide precursors?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its simple operation, minimal three-waste generation, and the ability to isolate the product directly via crystallization from the mother liquor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-bis(2,4-diaminophenoxy)benzene Supplier
As the demand for high-performance polyimides continues to surge in the aerospace and flexible electronics sectors, securing a partner with deep technical expertise in aromatic amine synthesis is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. We understand that in the realm of electronic materials, even trace impurities can compromise the performance of the final device, which is why our manufacturing processes are designed with purity as the primary objective.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of 1,4-bis(2,4-diaminophenoxy)benzene can enhance your supply chain resilience. By partnering with us, you gain access to a reliable source of high-purity intermediates that empowers your innovation while optimizing your bottom line through efficient, scalable, and environmentally responsible manufacturing practices.
