Advanced Imazethapyr Manufacturing: Overcoming Crystal Form and Cost Barriers for Global Supply Chains
Advanced Imazethapyr Manufacturing: Overcoming Crystal Form and Cost Barriers for Global Supply Chains
The global demand for high-efficiency, broad-spectrum herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for imidazolinone class compounds like Imazethapyr. A pivotal advancement in this domain is detailed in patent CN102453022A, which discloses a novel preparation method that fundamentally alters the production landscape for this critical active ingredient. This technology addresses long-standing industrial pain points regarding product crystallinity, filtration efficiency, and raw material utilization. By shifting away from traditional acidic hydrolysis pathways towards a streamlined anhydride coupling and oxidative cyclization strategy, manufacturers can achieve a superior Crystal Form II product. This specific polymorph is characterized by exceptional stability and low turbidity, making it ideally suited for downstream formulation processes. For a reliable agrochemical intermediate supplier, mastering this synthetic route represents a significant competitive advantage in delivering high-purity materials that meet stringent international quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
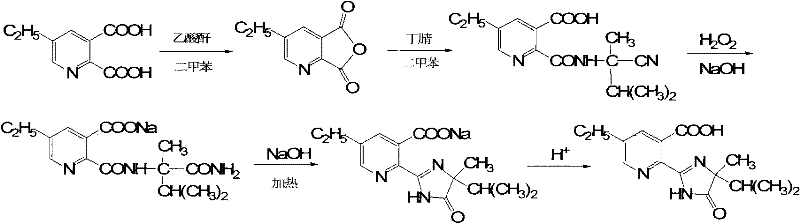
Historically, the industrial production of Imazethapyr has relied heavily on methodologies described in earlier patents such as US4861887 and US4925944, which typically involve the hydrolysis of 2-amino-2,3-dimethylbutyronitrile under harsh sulfuric acid conditions prior to coupling. This conventional approach presents severe operational drawbacks, primarily stemming from the generation of massive volumes of acidic wastewater, which imposes a heavy burden on environmental treatment facilities and increases overall processing costs. Furthermore, the resulting product often crystallizes into Form I, a polymorph characterized by extremely fine particle sizes and high turbidity levels exceeding 800 NTU. These physical properties create significant bottlenecks in solid-liquid separation units, leading to prolonged filtration times, increased energy consumption for drying, and potential losses of valuable material in the mother liquor. The uncertainty of crystal form also complicates formulation stability, posing risks for the final herbicide efficacy and shelf life.
The Novel Approach
In stark contrast, the methodology outlined in CN102453022A introduces a paradigm shift by utilizing 5-ethyl-2,3-pyridinedicarboxylic anhydride as the key electrophilic species, reacting directly with 2-amino-2,3-dimethylbutyronitrile. This strategic modification bypasses the need for preliminary nitrile hydrolysis, thereby drastically simplifying the synthetic sequence and reducing the chemical oxygen demand (COD) of the effluent. The subsequent cyclization is achieved through a controlled oxidative environment using sodium hydroxide and hydrogen peroxide, rather than strong mineral acids. This gentle yet effective cyclization promotes the nucleation of the thermodynamically stable Crystal Form II. The resulting material exhibits superior rheological properties, with turbidity levels consistently below 100 NTU, ensuring rapid filtration and high recovery rates. This approach not only enhances cost reduction in herbicide manufacturing but also aligns with modern green chemistry principles by minimizing hazardous waste streams.
Mechanistic Insights into Anhydride Coupling and Oxidative Cyclization
The core of this innovative synthesis lies in the precise control of the acylation and subsequent cyclization steps. The process initiates with the in situ generation of 5-ethyl-2,3-pyridinedicarboxylic anhydride from 5-ethylpicolinic acid and acetic anhydride in a xylene solvent system under reflux conditions. This activation step is critical, as the anhydride serves as a highly reactive acylating agent that facilitates the formation of the amide bond with the sterically hindered amine, 2-amino-2,3-dimethylbutyronitrile. The reaction is meticulously maintained at a low temperature range of 8-12°C during the addition of the amine to suppress side reactions and ensure high regioselectivity. Following the coupling, the intermediate 2-[(1-cyano-1,2-dimethylpropyl)-formamido]-5-ethyl nicotinic acid is isolated. This intermediate stability is crucial for maintaining batch-to-batch consistency in large-scale operations.
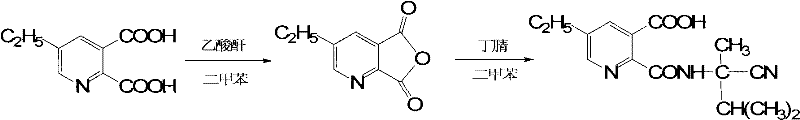
The transformation of the linear amide intermediate into the cyclic imidazolinone ring is achieved through a unique alkaline oxidative hydrolysis mechanism. Unlike traditional acid-catalyzed cyclization, this method employs a NaOH/H2O2 system where the cyano group is first hydrolyzed to an amide and subsequently undergoes intramolecular condensation. The temperature profile is rigorously controlled, starting at 20-25°C for the initial peroxide addition, then ramping to 70°C and finally 90°C to drive the cyclization to completion. This thermal gradient ensures that the kinetic barriers for ring closure are overcome without degrading the sensitive pyridine moiety. The final acidification step at pH 3-4 triggers the precipitation of the free acid in its preferred Crystal Form II. This mechanistic pathway effectively controls impurity profiles, preventing the formation of open-ring byproducts that often plague acidic hydrolysis routes, thereby securing high-purity Imazethapyr suitable for premium agricultural applications.
How to Synthesize Imazethapyr Efficiently
Implementing this advanced synthetic route requires strict adherence to the optimized reaction parameters defined in the patent literature to ensure the exclusive formation of the desired polymorph. The process integrates solvent selection, temperature gradients, and stoichiometric precision to maximize yield and purity. Operators must focus on the efficient removal of solvents and the precise control of pH during the final isolation stage to guarantee the physical specifications of the final powder. The following guide outlines the standardized operational framework derived from the patented technology, providing a roadmap for technical teams aiming to replicate these results in a pilot or commercial setting. Detailed standard operating procedures for each unit operation are essential for maintaining safety and quality compliance.
- Prepare 5-ethyl-2,3-pyridinedicarboxylic anhydride by reacting 5-ethylpicolinic acid with acetic anhydride in xylene under reflux, followed by cooling to 8-12°C.
- React the anhydride with 2-amino-2,3-dimethylbutyronitrile at 8-12°C to form the intermediate 2-[(1-cyano-1,2-dimethylpropyl)-formamido]-5-ethyl nicotinic acid.
- Hydrolyze the cyano group using NaOH and H2O2, followed by cyclization at elevated temperatures (70-90°C) and acidification to pH 3-4 to isolate Crystal Form II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of expensive activating agents and the simplification of the workflow directly translate to a more resilient and cost-effective supply chain. By removing the dependency on scarce or costly reagents like 4-picoline, which was required in older activation methods, manufacturers can stabilize their raw material sourcing and mitigate price volatility risks. Furthermore, the enhanced physical properties of the final product significantly reduce processing time in the finishing department, allowing for higher throughput without capital investment in new filtration equipment. These factors collectively contribute to substantial cost savings and improved margin protection in a competitive market.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive 4-picoline and removes the requirement for nitrogen protection during the reaction phase, which significantly lowers both raw material expenditures and utility costs associated with inert gas blanketing. Additionally, the avoidance of strong sulfuric acid hydrolysis reduces the consumption of neutralizing bases and minimizes the volume of saline wastewater requiring treatment, leading to drastic reductions in waste disposal fees. The overall simplification of the synthetic steps also reduces labor hours and energy consumption per kilogram of product, creating a leaner manufacturing cost structure that enhances competitiveness.
- Enhanced Supply Chain Reliability: The robustness of the anhydride coupling reaction ensures consistent batch quality, reducing the risk of off-spec material that could disrupt downstream formulation schedules. The improved filtration characteristics of Crystal Form II mean that production cycles are shorter and more predictable, allowing for tighter delivery windows and better inventory management. By securing a supply of high-purity intermediates with stable physical properties, formulators can reduce their own quality control burdens and accelerate their time-to-market for finished herbicide products, strengthening the entire value chain.
- Scalability and Environmental Compliance: This methodology is inherently designed for commercial scale-up of complex agrochemical intermediates, as it avoids exothermic hazards associated with large-scale acid hydrolysis and utilizes common, scalable solvents like xylene. The significant reduction in wastewater generation and the absence of heavy metal catalysts align perfectly with increasingly stringent global environmental regulations, ensuring long-term operational continuity without the risk of regulatory shutdowns. This environmental stewardship not only protects the manufacturer's license to operate but also appeals to end-users who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding the implementation of this Imazethapyr synthesis technology. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, focusing on the practical implications for industrial production. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or process optimization projects. The answers provided reflect the specific advantages of the Crystal Form II pathway over legacy methods.
Q: How does the new process improve product filtration compared to conventional methods?
A: The novel method produces Imazethapyr Crystal Form II, which exhibits larger particle sizes and lower turbidity (<100 NTU) compared to the fine, difficult-to-filter Crystal Form I produced by traditional sulfuric acid hydrolysis methods.
Q: What are the primary cost drivers eliminated in this synthesis route?
A: This route eliminates the need for expensive 4-picoline as a solvent/activator and removes the requirement for nitrogen protection during the reaction, significantly lowering raw material and operational costs.
Q: Does this method reduce environmental waste generation?
A: Yes, by avoiding the preliminary hydrolysis of the nitrile group under strong acidic conditions, the process generates substantially less wastewater, easing the burden on three-waste treatment facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imazethapyr Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature and pH controls required for Imazethapyr Crystal Form II are maintained with absolute precision. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical tools to verify crystal form and turbidity metrics, guaranteeing that every batch meets the high standards demanded by global agrochemical leaders. Our commitment to process excellence ensures that the theoretical benefits of this patented route are fully realized in commercial supply.
We invite you to collaborate with us to optimize your herbicide supply chain and leverage these technological advancements for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical performance data rather than theoretical projections. Let us partner to deliver high-quality, cost-effective solutions that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
