Advanced Synthesis of 1,5-Bis(2,4-Diaminophenoxy)Naphthalene for High-Performance Polyimides
Advanced Synthesis of 1,5-Bis(2,4-Diaminophenoxy)Naphthalene for High-Performance Polyimides
The rapid evolution of the flexible electronics industry demands materials with exceptional thermal stability and low dielectric constants, driving the need for advanced aromatic diamine monomers. Patent CN101260056A introduces a robust and industrially viable preparation method for 1,5-bis(2,4-diaminophenoxy)naphthalene, a critical building block for next-generation polyimide resins used in Flexible Copper Clad Laminates (FCCL). This technology represents a significant leap forward in process efficiency, utilizing a catalytic transfer hydrogenation strategy that bypasses the complexities of traditional high-pressure reduction methods. By leveraging a palladium on carbon catalyst system with hydrazine hydrate in saturated fatty alcohol solvents, the process achieves remarkable purity levels exceeding 99% without necessitating cumbersome downstream purification. For R&D directors and procurement specialists alike, this patent outlines a pathway to secure a reliable supply of high-performance electronic chemical intermediates while optimizing manufacturing overheads through simplified unit operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitro groups in complex aromatic ethers like 1,5-bis(2,4-dinitrophenoxy)naphthalene has posed significant engineering challenges for chemical manufacturers. Traditional approaches often rely on catalytic hydrogenation using molecular hydrogen gas, which requires specialized high-pressure reactors and stringent safety protocols to manage explosion risks associated with hydrogen handling. Alternatively, chemical reduction using metals such as iron or tin in acidic media generates substantial quantities of hazardous solid waste and wastewater, creating severe environmental compliance burdens and escalating disposal costs. Furthermore, these conventional routes frequently suffer from incomplete conversion or over-reduction side reactions, leading to impurity profiles that complicate the subsequent polymerization steps required for polyimide synthesis. The necessity for multiple recrystallization steps to achieve electronic-grade purity further erodes overall yield and extends production lead times, making the final monomer prohibitively expensive for mass-market electronic applications.
The Novel Approach
The methodology disclosed in the patent data offers a transformative alternative by employing hydrazine hydrate as a liquid hydrogen donor in the presence of a heterogeneous Pd/C catalyst. This catalytic transfer hydrogenation proceeds under atmospheric pressure conditions within a moderate temperature range of 70°C to 85°C, drastically reducing the capital expenditure required for reactor infrastructure. The use of saturated fatty alcohols, such as ethanol or ethylene glycol, provides an excellent solvent medium that ensures high solubility of the reactants while facilitating easy product isolation through simple water addition. This novel approach effectively mitigates the safety risks associated with high-pressure gas handling and eliminates the generation of heavy metal sludge typical of stoichiometric metal reductions. Consequently, the process delivers a streamlined workflow where the crude product can be isolated with exceptional purity directly from the reaction mixture, significantly enhancing the economic feasibility of producing this high-value electronic material intermediate.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic breakthrough lies in the efficient activation of hydrazine by the palladium surface, which facilitates the selective reduction of nitro groups to primary amines without compromising the integrity of the ether linkages. In this catalytic cycle, hydrazine adsorbs onto the active sites of the Pd/C catalyst and decomposes to generate reactive hydrogen species in situ, which are immediately transferred to the nitro functionalities of the substrate. This mechanism is highly chemoselective, ensuring that the sensitive aryl-oxygen bonds remain intact, a critical requirement for maintaining the structural properties necessary for high-performance polyimide formation. The reaction kinetics are optimized by maintaining the temperature between 70°C and 85°C, which provides sufficient energy for the reduction while preventing the thermal decomposition of hydrazine or the solvent system. The use of a heterogeneous catalyst also allows for straightforward separation via hot filtration, enabling the potential recovery and reuse of the precious metal catalyst, thereby further improving the sustainability profile of the manufacturing process.
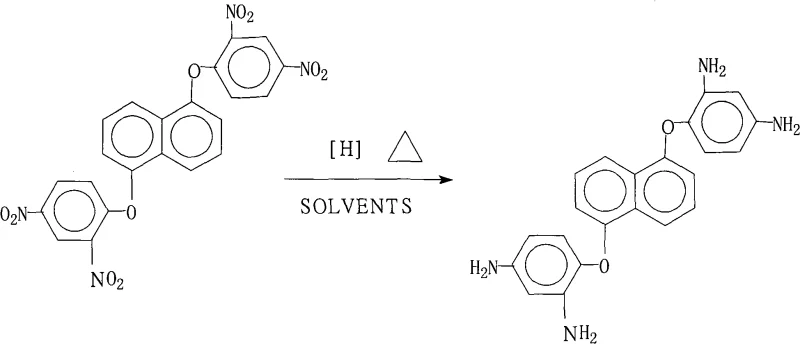
Impurity control is inherently built into this reaction design through the precise control of reagent stoichiometry and the physical properties of the product. By utilizing a molar excess of hydrazine hydrate ranging from 1:20 to 1:50 relative to the substrate, the reaction drives the reduction to completion, minimizing the presence of partially reduced intermediates such as hydroxylamines or azo compounds. The subsequent workup procedure exploits the differential solubility of the product in the alcohol-water mixture; upon cooling the mother liquor and adding pure water, the target diamine crystallizes out selectively while soluble byproducts and residual catalyst fines remain in the solution. This crystallization-induced purification is highly effective, consistently yielding products with purity levels above 99% as confirmed by the experimental data in the patent. Such rigorous control over the impurity spectrum is vital for downstream polymerization, as even trace impurities can act as chain terminators or defects in the final polyimide film, adversely affecting its mechanical and electrical performance in FCCL applications.
How to Synthesize 1,5-Bis(2,4-Diaminophenoxy)Naphthalene Efficiently
To implement this synthesis effectively in a pilot or production setting, operators must adhere to specific parameter controls regarding catalyst loading and solvent composition. The patent specifies a catalyst loading of Pd/C between 1% and 15% palladium content, with a substrate-to-catalyst weight ratio optimized between 100:1 and 100:20 to balance reaction rate and cost. The solvent system is flexible, allowing for mixtures of lower alcohols like methanol or ethanol with glycols such as ethylene glycol to tune the solubility and boiling point characteristics. Detailed standardized operating procedures regarding the rate of hydrazine addition and the specific cooling profiles for crystallization are essential to replicate the high yields reported in the examples. For a comprehensive guide on the exact step-by-step execution of this protocol, please refer to the structured synthesis guide below.
- Charge the reactor with 1,5-bis(2,4-dinitrophenoxy)naphthalene, Pd/C catalyst, and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution, then maintain reaction temperature between 70°C and 85°C.
- Filter hot to remove catalyst, cool the filtrate, add water to precipitate crystals, and vacuum dry to obtain >99% pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route translates into tangible strategic benefits regarding cost stability and supply continuity. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for manufacturing this intermediate, allowing for a broader base of qualified suppliers and reducing the risk of single-source dependency. Furthermore, the use of commodity chemicals such as hydrazine hydrate and common alcohols ensures that raw material sourcing remains resilient against market volatility, unlike specialized reagents that may face supply constraints. The simplified workup process, which avoids complex distillation or chromatographic purification, reduces energy consumption and labor hours per batch, directly contributing to a lower cost of goods sold. These factors combined create a more robust supply chain capable of meeting the growing demand for high-performance materials in the electronics sector without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The transition from high-pressure hydrogenation or metal-acid reduction to this atmospheric hydrazine method removes the need for expensive pressure-rated vessels and extensive waste treatment facilities. By avoiding the generation of heavy metal sludge, the facility saves substantially on hazardous waste disposal fees and environmental compliance costs. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting material to side products, ensuring that the theoretical yield is closely approached in practice. The ability to recover the product through simple crystallization rather than energy-intensive distillation further drives down utility costs, making the overall manufacturing economics highly favorable for large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing reliability is bolstered by the use of widely available raw materials that are not subject to the same geopolitical or logistical bottlenecks as rare earth catalysts or high-purity gases. The operational simplicity of the process means that production schedules are less prone to delays caused by equipment maintenance or complex safety shutdowns associated with high-pressure systems. This operational resilience ensures a consistent flow of material to downstream polymer manufacturers, helping them maintain their own production schedules for FCCL and composite materials. Moreover, the scalability of the reaction from gram to ton scale is well-documented in the patent examples, providing confidence that supply can be ramped up quickly to meet surges in market demand.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns with modern green chemistry principles by reducing the E-factor through minimized waste generation and solvent usage. The absence of corrosive acids and heavy metal reducing agents simplifies the effluent treatment process, allowing facilities to meet stringent discharge regulations with standard wastewater treatment protocols. The use of saturated fatty alcohols, which are generally less toxic and more biodegradable than chlorinated solvents, further enhances the environmental profile of the manufacturing site. This compliance advantage not only mitigates regulatory risk but also appeals to end-users in the electronics industry who are increasingly demanding sustainable supply chains for their components.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,5-bis(2,4-diaminophenoxy)naphthalene based on the patented technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this monomer into their specific material formulations. The answers are derived directly from the experimental data and process descriptions provided in the intellectual property documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of the hydrazine reduction method for BDAPN?
A: The method described in patent CN101260056A eliminates the need for high-pressure hydrogenation equipment and complex post-reaction purification steps, resulting in a simpler, safer, and more cost-effective industrial process with yields exceeding 90%.
Q: Can this synthesis route be scaled for commercial polyimide production?
A: Yes, the process utilizes common solvents like ethanol and ethylene glycol and operates at moderate temperatures (70°C-85°C), making it highly suitable for large-scale commercial manufacturing of electronic grade monomers.
Q: What purity levels can be achieved without recrystallization?
A: The patented workup procedure involving hot filtration and water precipitation directly yields crystals with purity greater than 99%, removing the need for additional refining stages that typically increase production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Bis(2,4-Diaminophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of advanced polyimide materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of the global electronics industry. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in catalytic reduction processes allows us to optimize the synthesis of complex aromatic diamines, guaranteeing a consistent supply of material that enables our partners to innovate without supply chain interruptions.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
