Advanced Fullerene Derivatives for High-Efficiency Organic Photovoltaics and Sensor Applications
The landscape of organic electronics is continuously evolving, driven by the demand for materials that offer superior photoelectric conversion efficiency and stability. Patent CN105210205B introduces a significant advancement in this field by disclosing a novel class of fullerene derivatives designed specifically for use as n-type semiconductor materials. These compounds, characterized by a pyrrolidine ring fused to the C60 fullerene cage, are engineered to achieve exceptional purity levels exceeding 99%, a critical factor often overlooked in standard commercial grades. The patent meticulously defines this purity through a rigorous elemental analysis protocol, ensuring that the deviation between theoretical and actual elemental composition is minimized. This level of precision is not merely academic; it translates directly into enhanced device performance, particularly in organic thin-film solar cells where impurity-induced trap states can severely degrade efficiency. By leveraging a robust synthetic pathway involving 1,3-dipolar cycloaddition, the invention provides a reliable route to structurally diverse derivatives that overcome the limitations of traditional acceptors like PCBM.
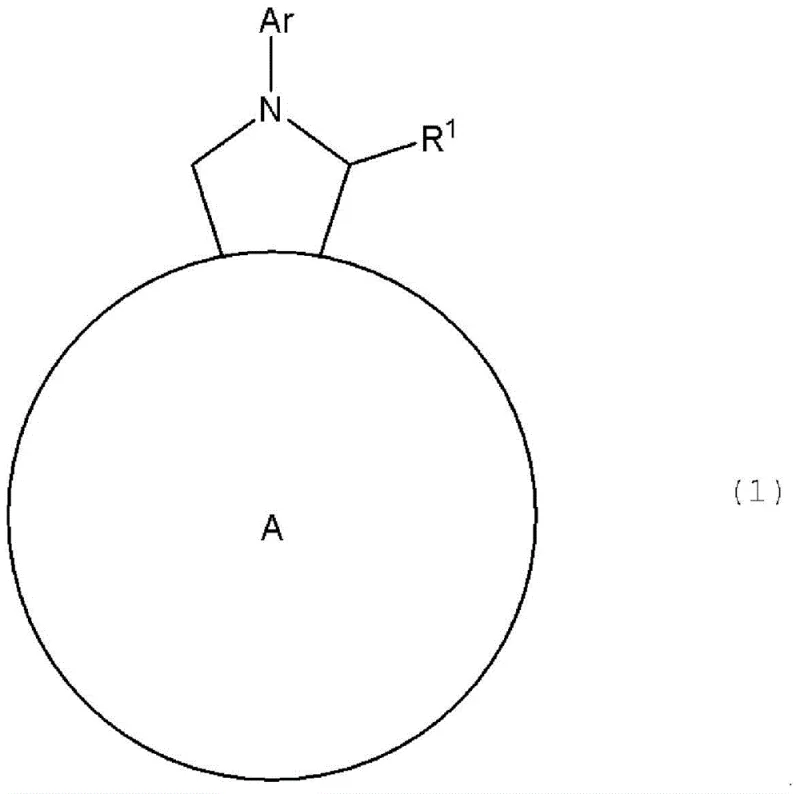
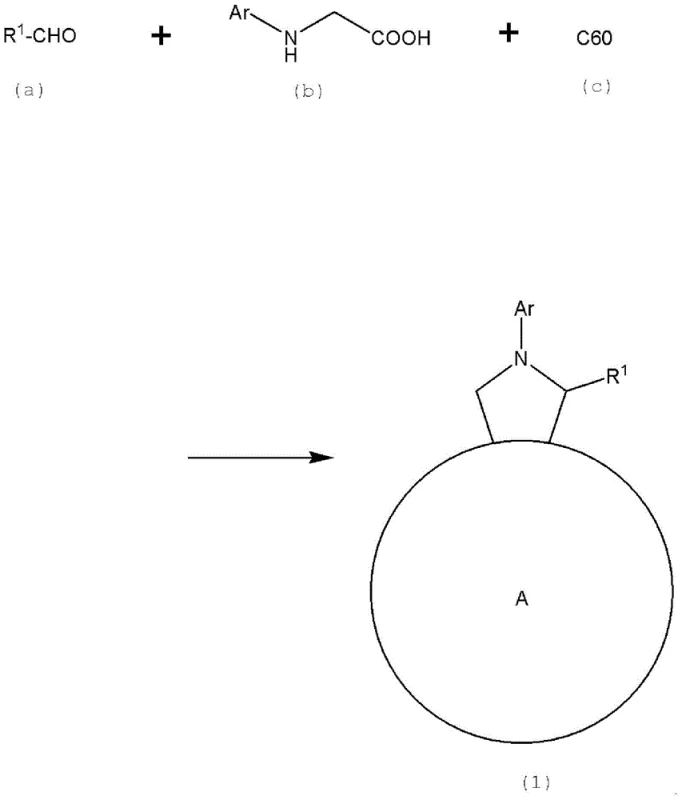
For procurement managers and supply chain directors seeking a reliable electronic chemical supplier, understanding the distinction between conventional methods and this novel approach is vital for strategic sourcing. Traditional fullerene derivatives, such as PCBM, while widely used, often suffer from limitations in structural tunability and may require complex post-synthetic modifications that introduce variability. The conventional synthesis of three-membered ring fullerene derivatives often yields mixtures that are difficult to separate, limiting the ability to fine-tune energy levels for specific donor-acceptor pairs. In contrast, the method described in CN105210205B utilizes a five-membered ring formation via the Prato reaction, which inherently offers greater structural diversity. This allows for the precise modification of solubility and energy levels by varying the aldehyde and N-substituted glycine precursors. Furthermore, the patent emphasizes a multi-stage purification process that goes beyond standard chromatography, incorporating sequential solvent washing and recrystallization to remove trace impurities that HPLC might miss. This ensures a consistent supply of high-performance material, reducing the risk of batch-to-batch variability that can plague large-scale manufacturing of organic photovoltaic modules.
From a mechanistic perspective, the synthesis relies on the generation of an azomethine ylide intermediate, which undergoes a concerted 1,3-dipolar cycloaddition with the electron-deficient double bonds of the C60 fullerene cage. This reaction is highly regioselective, typically occurring at the 6,6-junctions of the fullerene sphere to form the stable pyrrolidine ring. The choice of solvent plays a crucial role in this mechanism; high-boiling aromatic solvents like chlorobenzene or toluene are preferred to maintain the solubility of the fullerene reactant while providing sufficient thermal energy for the cycloaddition to proceed to completion. The reaction kinetics are further optimized by controlling the molar ratios of the aldehyde and glycine relative to the fullerene, typically maintaining a slight excess of the organic precursors to drive the equilibrium forward without generating excessive byproducts. Understanding this mechanism is essential for R&D directors aiming to replicate or scale this process, as deviations in temperature or stoichiometry can lead to the formation of bis-adducts or unreacted starting materials, complicating the downstream purification efforts required to meet the stringent 99% purity specification.
Mechanistic Insights into 1,3-Dipolar Cycloaddition for Fullerene Functionalization
The control of impurities in this synthesis is achieved through a combination of kinetic control during the reaction and thermodynamic control during purification. During the reaction phase, maintaining a temperature range of 80°C to 120°C ensures that the activation energy for the cycloaddition is met while minimizing thermal degradation of the sensitive azomethine ylide. Post-reaction, the removal of unreacted C60 is critical, as residual fullerene can act as a recombination center in the final device. The patent describes a sophisticated purification sequence starting with silica gel column chromatography to remove bulk impurities, followed by preparative Gel Permeation Chromatography (GPC). However, the true innovation lies in the final polishing steps: sequential washing with solvents of varying polarity (methanol, acetone, dichloromethane, THF, hexane) effectively strips away surface-adsorbed impurities and residual solvents that co-elute during chromatography. This is followed by recrystallization from a hexane-chlorobenzene mixture, which leverages the differential solubility of the target derivative versus isomeric byproducts. This multi-faceted approach ensures that the final product meets the strict elemental analysis criteria, guaranteeing the electronic quality required for high-efficiency high-purity OLED material and photovoltaic applications.
How to Synthesize High-Purity Fullerene Derivatives Efficiently
The synthesis of these advanced n-type semiconductor materials requires careful attention to reaction conditions and purification protocols to ensure the structural integrity and electronic purity of the final product. The process begins with the reflux of C60, an aldehyde, and an N-substituted glycine in an aromatic solvent, followed by a rigorous workup involving column chromatography and GPC. To achieve the necessary purity levels for commercial device fabrication, the crude material must undergo sequential solvent washing and recrystallization. Detailed standardized synthesis steps for replicating this high-yield pathway are provided in the guide below.
- React C60 fullerene with an N-substituted glycine and an aldehyde in a solvent like toluene or chlorobenzene under reflux conditions for 10 to 24 hours to form the crude pyrrolidine-fused fullerene derivative.
- Purify the crude reaction mixture using silica gel column chromatography with a hexane-toluene gradient, followed by preparative Gel Permeation Chromatography (GPC) using chloroform or toluene as the eluent.
- Perform sequential solvent washing with methanol, acetone, dichloromethane, THF, and hexane, followed by recrystallization from hexane-chlorobenzene and vacuum drying to achieve purity greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in battery & energy storage materials manufacturing, the adoption of this technology offers several strategic benefits rooted in process efficiency and material performance. The synthesis utilizes readily available commodity chemicals such as benzaldehyde, N-phenylglycine, and C60 fullerene, which simplifies the raw material supply chain and reduces dependency on exotic or proprietary reagents. The reaction conditions are relatively mild, operating at atmospheric pressure and moderate temperatures, which lowers the capital expenditure required for specialized high-pressure reactors. Furthermore, the use of common organic solvents like toluene and chlorobenzene facilitates solvent recovery and recycling, contributing to a more sustainable and cost-effective manufacturing process. By achieving higher photoelectric conversion efficiencies with these purified derivatives, manufacturers can potentially reduce the active layer thickness or the overall material loading per device, leading to substantial cost savings in the long run without compromising power output.
- Cost Reduction in Manufacturing: The elimination of complex transition metal catalysts, which are often required in other fullerene functionalization strategies, significantly reduces the cost of goods sold and simplifies the removal of metal residues. This catalyst-free approach not only lowers raw material costs but also reduces the environmental burden associated with heavy metal waste disposal. Additionally, the high yield of the primary cycloaddition reaction minimizes the loss of expensive C60 starting material, ensuring that the process remains economically viable even at large scales. The streamlined purification protocol, while rigorous, relies on standard unit operations like chromatography and crystallization that are easily scalable in existing chemical manufacturing facilities.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials ensures a robust supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. Since the precursors are bulk chemicals produced by multiple global suppliers, procurement teams can negotiate better pricing and secure long-term contracts with confidence. The scalability of the synthesis from gram-scale laboratory batches to multi-kilogram production runs has been demonstrated in the patent examples, indicating a clear path for commercial scale-up of complex polymer additives and semiconductor materials. This reliability is crucial for maintaining continuous production lines in the fast-paced organic electronics industry.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to methods involving toxic reagents or harsh conditions. The solvents used are standard industrial chemicals with established recycling infrastructure, allowing for effective waste management and compliance with increasingly stringent environmental regulations. The ability to produce high-purity materials without the need for extensive post-synthetic modifications reduces the overall process footprint. This aligns with the growing industry demand for green chemistry solutions, making these fullerene derivatives an attractive option for companies aiming to improve their sustainability profiles while maintaining high product performance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these fullerene derivatives in organic electronic devices. The answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on performance metrics and compatibility. Understanding these details is essential for integrating this material into existing product lines or developing new applications in the renewable energy sector.
Q: How is the purity of these fullerene derivatives defined in the patent?
A: The patent defines purity based on elemental analysis deviations. Specifically, purity (%) = 100 - Dmax(%), where Dmax is the maximum absolute difference between the theoretical and analytical values for Carbon, Hydrogen, and Nitrogen. A purity of 99% or higher is required for optimal performance.
Q: What p-type semiconductor materials are compatible with these n-type derivatives?
A: The derivatives show excellent compatibility with standard p-type materials such as P3HT (poly-3-hexylthiophene) and donor-acceptor type pi-conjugated polymers like PTB7. They facilitate the formation of bulk heterojunction structures essential for high photoelectric conversion efficiency.
Q: Can these materials be used in applications other than solar cells?
A: Yes, beyond organic thin-film solar cells, these high-purity n-type semiconductor materials are suitable for photosensor arrays and image sensors for digital cameras, offering high sensitivity and the ability to function effectively in both bright and low-light conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fullerene Derivatives Supplier
As the demand for high-efficiency organic photovoltaics continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge materials and scalable manufacturing solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fullerene derivatives meets the exacting standards required for next-generation electronic applications. Our commitment to quality assurance means that you can rely on us for materials that deliver reproducible device performance.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how optimizing your material supply chain can impact your overall production economics. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the suitability of these high-purity n-type semiconductors for your unique formulation needs. Let us help you accelerate your development timeline with our proven expertise in fine chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
