Advanced Regioselective Synthesis of Fullerene Derivatives for Commercial Scale-Up
The landscape of advanced material synthesis is undergoing a transformative shift with the introduction of patent CN101535220B, which details a revolutionary process for producing fullerene derivatives with unprecedented regioselectivity and yield. This intellectual property addresses a long-standing bottleneck in the functionalization of carbon clusters, specifically targeting the efficient production of mono-, di-, and tri-adducts that are critical for next-generation electronic and photofunctional applications. Unlike traditional methods that often result in complex mixtures of higher adducts, this novel approach leverages the synergistic interaction between Grignard reagents and specific polar substances to control the addition pattern on the fullerene cage. For R&D directors and procurement specialists seeking a reliable fullerene derivative supplier, understanding the mechanistic advantages of this technology is paramount for securing a stable supply chain of high-purity intermediates. The patent outlines a robust methodology that not only simplifies the synthetic route but also drastically enhances the economic viability of producing these sophisticated carbon nanomaterials.
By integrating this patented technology into our manufacturing portfolio, we can offer clients a distinct competitive edge in cost reduction in electronic chemical manufacturing. The ability to selectively synthesize lower adducts without the need for cumbersome separation processes translates directly into reduced operational expenditures and shorter lead times for high-purity fullerene derivatives. This report delves deep into the technical nuances of the invention, providing a comprehensive analysis of the reaction mechanisms, the critical role of solvent polarity, and the scalable nature of the process. As the demand for organic photovoltaics and semiconductor materials continues to surge, the capacity to deliver structurally defined fullerene derivatives at a commercial scale becomes a strategic asset. We invite industry leaders to explore how this synthesis platform can optimize their material sourcing strategies and accelerate their product development cycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fullerene derivatives has been plagued by significant challenges regarding regioselectivity and product distribution, particularly when aiming for lower addition numbers such as mono- or di-adducts. Conventional methodologies, such as those utilizing phenyl Grignard reagents in conjunction with organocopper catalysts like CuBr·S(CH3)2, have demonstrated efficacy primarily in the production of penta-addition derivatives where five organic groups surround a five-membered ring on the fullerene skeleton. While these methods achieve high yields for hexa-, hepta-, or deca-adducts, they suffer from poor selectivity when the target is a mono-, di-, tri-, or tetra-adduct. The inherent reactivity of the fullerene cage often leads to over-addition, resulting in a complex mixture of products that requires extensive and costly purification steps, such as high-performance liquid chromatography (HPLC), to isolate the desired species. This lack of control not only diminishes the overall yield of the target compound but also introduces significant variability in batch-to-batch consistency, which is unacceptable for high-specification applications in the semiconductor and display industries.
The Novel Approach
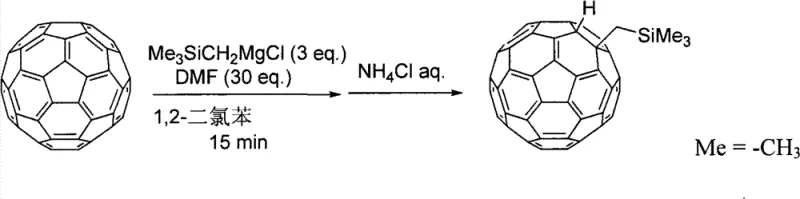
In stark contrast to the limitations of organocopper-mediated pathways, the process disclosed in patent CN101535220B introduces a paradigm shift by employing a Grignard reagent in the presence of a polar substance with a specific donor number (DN) of 25 or greater. This innovative strategy enables the position-selective addition of organic groups to the fullerene or fullerene derivative skeleton with remarkable efficiency. The core of this advancement lies in the organic group addition step A, where the interaction between the Grignard reagent and the polar solvent facilitates a controlled nucleophilic attack on the fullerene cage. This method effectively suppresses the formation of higher adducts, allowing for the high-yield isolation of mono- and di-addition products. For instance, experimental data within the patent demonstrates isolated yields reaching up to 93% for specific mono-adducts, a figure that vastly outperforms traditional techniques for these specific targets. This breakthrough provides a reliable pathway for the commercial scale-up of complex fullerene derivatives, ensuring that manufacturers can meet the rigorous purity standards required for advanced material applications without the burden of excessive waste generation.
Mechanistic Insights into Grignard-Mediated Regioselective Addition
The mechanistic underpinning of this synthesis route is fundamentally rooted in the modulation of the Grignard reagent's reactivity through coordination with high-donor-number polar substances. In the organic group addition step A, the polar substance, such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), acts not merely as a solvent but as a critical ligand that stabilizes the magnesium center and influences the trajectory of the nucleophilic attack. The patent specifies that the donor number of the polar substance must be 25 or more to achieve the desired enhancement in yield, indicating that the Lewis basicity of the solvent plays a pivotal role in the transition state stabilization. When the Grignard reagent reacts with the fullerene in the presence of these polar additives, it promotes the addition of both a hydrogen atom and an organic group to specific positions on the carbon cage, typically surrounding a five-membered ring as depicted in Formula (1). This regioselectivity is crucial because it prevents random poly-addition, thereby simplifying the impurity profile of the crude reaction mixture. The ability to tune the reaction outcome by simply adjusting the equivalents of the polar substance (ranging from 3 to 100 equivalents) offers process chemists a powerful handle to optimize the reaction for different substrate scales.
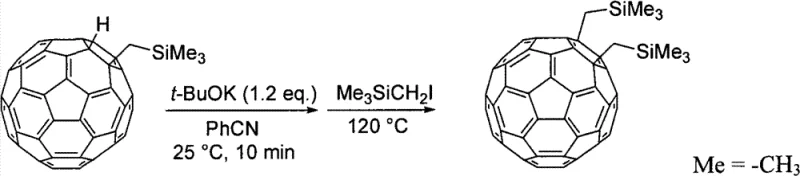
Furthermore, the process allows for a sequential functionalization strategy through organic group addition step B, which expands the structural diversity of the accessible derivatives. After the initial formation of a hydrogen-added fullerene derivative in step A, the subsequent reaction with a basic compound, such as potassium tert-butoxide (t-BuOK), and a halogen compound enables the replacement of the hydrogen atom with a second organic group. This dehydrogenative addition mechanism is highly advantageous for synthesizing symmetrical or asymmetrical di-adducts with precise control over the substitution pattern. The use of mild bases and standard alkylating agents in this second step ensures compatibility with a wide range of functional groups, including silyl, alkoxy, and aryl moieties. This modularity is essential for tailoring the electronic properties of the final fullerene derivative, whether it be for use as an electron acceptor in organic solar cells or as a charge transport material in OLED devices. The mechanistic clarity provided by this patent empowers manufacturers to design derivatives with specific HOMO-LUMO gaps and solubility profiles, directly addressing the custom requirements of downstream application engineers.
How to Synthesize Regioselective Fullerene Derivatives Efficiently
The practical implementation of this patented technology involves a streamlined workflow that is amenable to both laboratory-scale optimization and industrial manufacturing. The process begins with the dissolution of the starting fullerene material, such as C60, in a suitable aromatic solvent like o-dichlorobenzene, followed by the addition of the polar substance and the Grignard reagent under an inert atmosphere. The reaction proceeds rapidly, often completing within minutes to an hour at temperatures ranging from -50°C to 50°C, after which it is quenched with an aqueous ammonium chloride solution. The detailed standardized synthesis steps for replicating this high-yield transformation are outlined in the guide below, providing a clear roadmap for technical teams to validate the process in their own facilities. By adhering to these protocols, manufacturers can consistently achieve the high purity and selectivity metrics reported in the patent examples, ensuring that the final product meets the stringent specifications required for electronic grade materials.
- React fullerene or fullerene derivatives with a Grignard reagent and a polar substance having a donor number of 25 or more to perform the first organic group addition.
- Quench the reaction mixture with an aqueous ammonium chloride solution to stop the reaction and isolate the hydrogen-added intermediate.
- Optionally react the intermediate with a basic compound and a halogen compound to further add organic groups for di- or tri-adduct synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this Grignard-based synthesis route offers substantial strategic benefits that extend beyond mere technical performance. The elimination of expensive and potentially toxic transition metal catalysts, such as copper salts used in conventional methods, results in a significantly simplified workup procedure and reduces the environmental footprint of the manufacturing process. This reduction in chemical complexity translates directly into cost reduction in manufacturing, as there is no longer a need for specialized heavy metal scavenging resins or extensive wastewater treatment protocols associated with copper removal. Furthermore, the reagents utilized in this process, including Grignard reagents and common polar aprotic solvents like DMF, are commodity chemicals available from multiple global suppliers, thereby enhancing supply chain reliability and mitigating the risk of raw material shortages. This diversification of the supply base ensures that production schedules remain uninterrupted even in the face of market volatility, providing a stable foundation for long-term procurement planning.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic improvement in reaction selectivity, which minimizes the formation of difficult-to-separate byproducts. In traditional synthesis, the low yield of mono-adducts necessitates the processing of large volumes of starting material to obtain a small amount of product, inflating the cost per kilogram. By achieving isolated yields exceeding 90% for target adducts, this process maximizes atom economy and reduces the consumption of expensive fullerene starting materials. Additionally, the simplified purification requirements mean that less solvent and stationary phase are needed for chromatography, further lowering the variable costs associated with production. These efficiencies accumulate to provide substantial cost savings that can be passed down to the customer or reinvested into R&D for new material development.
- Enhanced Supply Chain Reliability: The reliance on robust, well-established chemical transformations ensures that the manufacturing process is resilient to disruptions. Grignard reagents are synthesized from widely available alkyl or aryl halides and magnesium metal, commodities that are produced on a massive global scale. This stands in contrast to specialized organometallic catalysts that may have limited suppliers and long lead times. By anchoring the synthesis on these staple reagents, we can guarantee consistent availability and shorter lead times for high-purity fullerene derivatives. Moreover, the process operates under relatively mild conditions and atmospheric pressure, reducing the need for specialized high-pressure reactor infrastructure and allowing for flexible production scheduling across multiple manufacturing sites to meet surging demand.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is straightforward due to the exothermic nature of the Grignard addition being easily manageable with standard cooling systems and the absence of sensitive catalytic species that might deactivate upon scale-up. The use of polar substances like DMF, while requiring careful handling, is a standard practice in the fine chemical industry with established recovery and recycling protocols, supporting environmental compliance goals. The ability to produce multi-kilogram batches with consistent quality metrics makes this technology ideal for supporting the commercialization of new electronic devices that require large quantities of active materials. This scalability ensures that as our clients' products move from pilot testing to mass market release, their supply of critical fullerene intermediates can expand seamlessly without the need for process re-validation.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the technical details provided in the patent documentation. These inquiries address common concerns regarding reaction scope, purification strategies, and the versatility of the functional groups that can be installed. Understanding these aspects is critical for R&D teams who are looking to integrate these derivatives into complex device architectures. The answers provided below reflect the proven capabilities of the synthesis method as demonstrated in the experimental examples, offering a transparent view of what can be achieved through this partnership.
Q: What is the primary advantage of using polar substances in this fullerene synthesis?
A: Using polar substances with a high donor number (DN ≥ 25), such as DMF or DMSO, significantly increases the yield of mono- and di-adducts compared to conventional organocopper methods, which typically favor higher adducts.
Q: Can this process be scaled for industrial production of electronic materials?
A: Yes, the process utilizes readily available Grignard reagents and mild reaction conditions (-50°C to 50°C), making it highly suitable for commercial scale-up from kilogram to tonnage levels without requiring expensive transition metal catalysts.
Q: What types of organic groups can be introduced using this method?
A: The method supports a wide variety of groups including C1-C30 hydrocarbon groups, silyl groups, alkoxy groups, and aryl groups, allowing for precise tuning of the derivative's electronic and solubility properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fullerene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced electronic materials hinges on the availability of high-quality, structurally precise intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your growth from early-stage research to full-scale manufacturing. We are committed to delivering fullerene derivatives that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging the proprietary synthesis methods described in patent CN101535220B, we can offer you a level of customization and reliability that is unmatched in the current market, positioning your projects for success in the competitive landscape of optoelectronics and energy storage.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis platform to your specific molecular targets. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to request specific COA data and route feasibility assessments for novel derivatives, we are ready to provide the support you need. Our goal is to become your strategic partner in material innovation, helping you overcome synthesis bottlenecks and accelerate your time-to-market. Let us collaborate to define the next generation of carbon-based functional materials together.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
