Advanced Solid-State Synthesis of Milrinone Intermediates for Commercial Scale-Up
Advanced Solid-State Synthesis of Milrinone Intermediates for Commercial Scale-Up
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for safer and more efficient manufacturing processes for critical active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN113493405A, which discloses a robust preparation method for 4-(dimethylamino)-3-(pyridine-4-yl)but-3-en-2-one, a vital intermediate in the synthesis of Milrinone. Milrinone, a potent phosphodiesterase III inhibitor, remains a cornerstone treatment for refractory heart failure and low-cardiac-ejection syndromes, necessitating a supply chain that guarantees both high purity and consistent availability. The structural complexity of Milrinone, characterized by its bipyridine core and nitrile functionality, presents significant synthetic challenges that this new methodology aims to resolve through innovative solid-state chemistry.
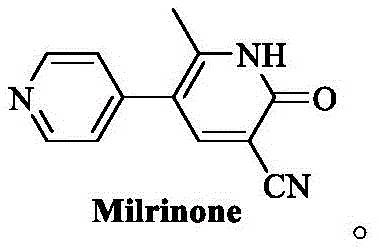
This patented approach fundamentally shifts the paradigm from handling unstable liquid intermediates to utilizing stable, high-purity solid precursors. For R&D directors and process chemists, this transition represents a critical optimization in impurity control and reaction reproducibility. By replacing the traditional liquid 1-(4-pyridyl)-2-acetone with a solid acetylated pyridine derivative, the process mitigates the risks associated with high-temperature vacuum distillation and exothermic instability. Furthermore, the method employs greener solvent systems and avoids the use of highly toxic reagents like hexamethylphosphoramide (HMPA), aligning with modern environmental, health, and safety (EHS) standards required by global regulatory bodies. This technical breakthrough not only enhances the quality of the final API but also streamlines the path toward commercial viability for generic manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Milrinone has been plagued by significant operational hazards and inefficiencies inherent in legacy 'linear' and 'convergent' routes. Traditional linear pathways often rely on the use of strong bases such as n-butyllithium or phenyllithium to generate reactive anions, which pose severe safety risks including pyrophoricity and potential explosion upon scale-up due to uncontrolled exotherms. Additionally, these methods frequently require stringent anhydrous conditions and generate toxic byproducts like benzene, complicating waste management and increasing production costs. Another common conventional route involves the use of liquid 1-(4-pyridyl)-2-acetone, which necessitates high-temperature reduced-pressure distillation for purification. This step is not only energy-intensive but also prone to thermal degradation, leading to variable purity and difficult-to-control feeding amounts in subsequent reaction stages.
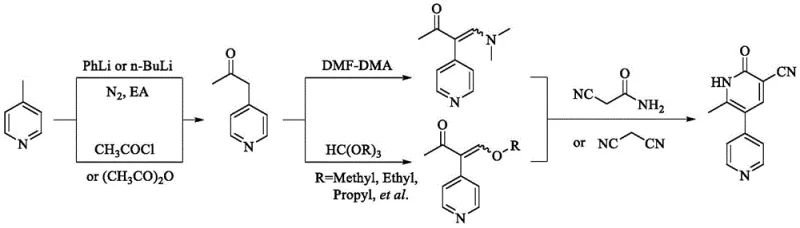
Furthermore, convergent synthetic strategies reported in earlier literature often suffer from low overall yields and complex purification protocols. For instance, routes utilizing malononitrile and triethyl orthoformate to generate alpha-ethoxymethylene malononitrile involve multiple isolation steps that erode material throughput. The final cyclization steps in these older methods frequently result in colored crude products that are difficult to refine to injection-grade standards without extensive recrystallization or chromatography. The reliance on toxic solvents like HMPA for the synthesis of key enaminone intermediates further exacerbates the environmental burden, requiring specialized disposal procedures and increasing the total cost of ownership for manufacturing facilities. These cumulative drawbacks render many traditional processes unsuitable for modern, large-scale industrial production where safety and cost-efficiency are paramount.
The Novel Approach
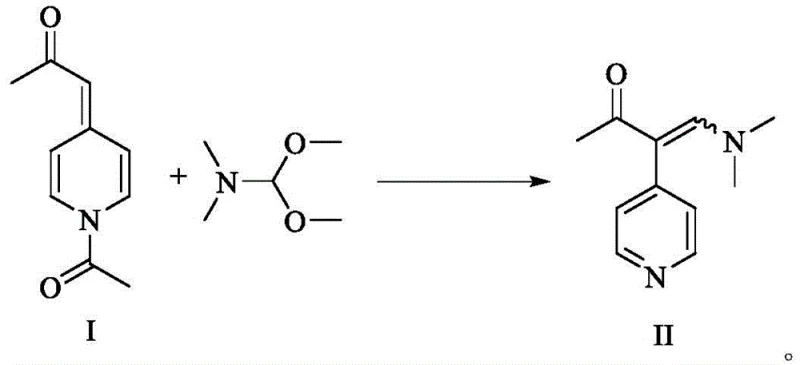
The methodology outlined in CN113493405A introduces a transformative solution by leveraging a stable solid intermediate, designated as Compound I, to bypass the limitations of liquid precursors. This novel route begins with the acetylation of 4-methylpyridine to form a solid 1-(1-acetylpyridine-4(1H)-ylidene)-2-acetone, which can be purified to exceptional standards (over 99.8% purity) through a simple recrystallization with acetic anhydride. This solid-state advantage eliminates the need for hazardous high-vacuum distillation and ensures precise stoichiometric control during the subsequent enaminone formation. The conversion of this solid intermediate to the key enaminone (Compound II) is achieved using N,N-dimethylformamide dimethyl acetal (DMF-DMA) in safe solvents like 1,4-dioxane, proceeding under mild reflux conditions with high conversion rates. This shift from liquid to solid handling significantly reduces operational complexity and enhances the safety profile of the entire synthesis campaign.
Moreover, the final cyclization step utilizes alpha-cyanoacetamide instead of the more toxic and expensive malononitrile, reacting with the enaminone intermediate in alcoholic solvents such as ethanol. This one-pot cyclization strategy simplifies the workflow by reducing the number of unit operations and avoiding the need for complex extraction procedures like Soxhlet extraction. The resulting Milrinone crude is obtained as an off-white solid with high purity, minimizing the need for aggressive refining steps. By integrating these improvements, the novel approach offers a streamlined, safer, and more economically viable pathway that is inherently scalable. For procurement and supply chain leaders, this translates to a more reliable source of high-quality intermediates with reduced risk of production delays caused by safety incidents or purification bottlenecks.
Mechanistic Insights into Solid-State Precursor Stability and Condensation
The core mechanistic advantage of this process lies in the stabilization of the reactive ketone functionality through N-acetylation, forming the solid zwitterionic or tautomeric species known as Intermediate I. In traditional liquid 1-(4-pyridyl)-2-acetone, the free pyridine nitrogen can interfere with electrophilic attacks or lead to polymerization under acidic or thermal stress. By temporarily blocking the nitrogen with an acetyl group, the molecule gains significant thermal stability and crystallinity, allowing for rigorous purification via recrystallization. This purification step is critical for removing trace impurities that could otherwise propagate through the synthesis, affecting the crystal form and bioavailability of the final API. The solid nature of Intermediate I also facilitates accurate weighing and dosing, which is essential for maintaining consistent reaction kinetics in large-scale reactors where heat transfer limitations can amplify minor stoichiometric deviations.
The subsequent transformation of Intermediate I into the enaminone Intermediate II proceeds via a nucleophilic attack of the enolizable methyl ketone on the electrophilic carbon of DMF-DMA. This condensation reaction is facilitated by the electron-withdrawing nature of the N-acetyl pyridinium moiety, which enhances the acidity of the alpha-protons. The use of DMF-DMA serves a dual purpose: it acts as both a reagent and a protecting group carrier, installing the dimethylamino vinyl group necessary for the final ring closure. The reaction mechanism avoids the formation of hazardous byproducts associated with lithium-halogen exchange or strong base deprotonation. Instead, it relies on thermal activation in polar aprotic solvents, generating methanol and dimethylamine as volatile byproducts that are easily removed. This clean reaction profile minimizes the formation of tars and oligomers, directly contributing to the high isolated yields reported in the patent examples.
How to Synthesize 4-(dimethylamino)-3-(pyridine-4-yl)but-3-en-2-one Efficiently
The synthesis of this critical enaminone intermediate is the linchpin of the entire Milrinone production process, dictating the quality and yield of the final drug substance. The patented procedure outlines a precise sequence involving the preparation of the solid precursor followed by condensation and purification. Operators must strictly adhere to temperature controls during the acetylation and reflux stages to prevent decomposition. The workup procedure involves a unique acid-base extraction strategy where the crude product is dissolved in dilute acid, washed with organic solvents to remove non-basic impurities, and then precipitated by adjusting the pH with alkali. This purification technique effectively removes unreacted starting materials and side products, yielding needle-shaped crystals of high purity suitable for direct use in the next step without further chromatography.
- Preparation of Solid Intermediate I: React 4-methylpyridine with acetyl chloride in dichloromethane, followed by alkaline workup and recrystallization with acetic anhydride to obtain high-purity solid 1-(1-acetylpyridine-4(1H)-ylidene)-2-acetone.
- Synthesis of Enaminone Intermediate II: Reflux solid Intermediate I with N,N-dimethylformamide dimethyl acetal (DMF-DMA) in 1,4-dioxane, followed by water precipitation and acid-base purification to isolate needle crystals.
- Cyclization to Milrinone: React Intermediate II with alpha-cyanoacetamide in ethanol under alkaline reflux conditions, followed by acidification and hot water pulping to yield high-purity Milrinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical superiority. The elimination of hazardous reagents like n-butyllithium and toxic solvents like HMPA drastically reduces the regulatory burden and insurance costs associated with manufacturing. Facilities no longer require specialized infrastructure for handling pyrophoric materials or complex waste treatment systems for phosphoramide residues. This simplification of the safety profile allows for production in a wider range of certified facilities, thereby diversifying the supply base and reducing the risk of single-source bottlenecks. Furthermore, the use of commodity chemicals such as acetyl chloride, acetic anhydride, and ethanol ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by eliminating expensive purification steps such as alumina column chromatography and high-vacuum distillation. The ability to purify intermediates through simple crystallization and filtration reduces solvent consumption and energy usage, directly lowering the cost of goods sold (COGS). Additionally, the higher yields obtained at each stage mean less raw material is wasted, maximizing the output per batch and improving overall asset utilization. The replacement of malononitrile with the more cost-effective alpha-cyanoacetamide further contributes to raw material cost optimization without compromising reaction efficiency.
- Enhanced Supply Chain Reliability: The stability of the solid Intermediate I allows for long-term storage and transportation without degradation, enabling manufacturers to build strategic inventory buffers against market fluctuations. Unlike liquid intermediates that may require cold chain logistics or immediate use, this solid precursor can be stockpiled safely, ensuring continuous production even if upstream supply lines are temporarily disrupted. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supplies (e.g., cooling water temperature), leading to more consistent batch cycles and reliable delivery schedules for downstream API customers.
- Scalability and Environmental Compliance: The route is designed with industrial scale-up in mind, utilizing standard reactor equipment and avoiding exothermic runaways that limit batch sizes in traditional methods. The reduced toxicity profile aligns with increasingly stringent global environmental regulations, facilitating easier permitting and audit compliance for manufacturing sites. By minimizing the generation of hazardous waste and reducing the E-factor of the process, companies can achieve sustainability goals while maintaining high production volumes. This environmental stewardship is increasingly becoming a key differentiator in supplier selection criteria for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-state synthesis route for Milrinone intermediates. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on quality and reliability metrics.
Q: Why is the solid intermediate route superior to traditional liquid methods?
A: Traditional methods rely on liquid 1-(4-pyridyl)-2-acetone which requires difficult high-temperature vacuum distillation and poses handling risks. The novel solid intermediate I eliminates distillation, offers higher purity through recrystallization, and simplifies feeding control for industrial scale-up.
Q: How does this process address toxicity concerns in Milrinone synthesis?
A: Conventional routes often utilize highly toxic solvents like hexamethylphosphoramide (HMPA) or hazardous reagents like n-butyllithium. This patented method replaces HMPA with safer solvents like 1,4-dioxane or ethanol and avoids pyrophoric reagents, significantly improving operational safety and environmental compliance.
Q: What represents the key yield improvement in this synthesis pathway?
A: By utilizing a stable solid precursor and optimizing the condensation with DMF-DMA, the process achieves yields exceeding 90% for the key enaminone intermediate, compared to significantly lower yields in older convergent routes that suffered from side reactions and difficult purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patented solid-state process are fully realized at an industrial level. Our state-of-the-art facilities are equipped to handle the specific solvent systems and crystallization requirements of this route, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of global regulatory agencies, providing our clients with a secure and compliant supply chain foundation.
We invite you to collaborate with us to optimize your Milrinone supply chain and reduce manufacturing costs through the adoption of this superior technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your cardiovascular drug portfolio. Let us engineer the bottleneck out of your production process and deliver the reliability your business demands.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
