Advanced Continuous Flow Synthesis of Methyl 2-Amino-3-Nitrobenzoate for Commercial API Production
The pharmaceutical industry is constantly seeking robust manufacturing routes for critical intermediates, particularly those serving the Angiotensin II Receptor Blocker (ARB) class of drugs. Patent CN111253271B introduces a transformative approach to synthesizing methyl 2-amino-3-nitrobenzoate, a pivotal building block for medications such as candesartan and azilsartan. This innovation shifts the paradigm from traditional batch processing to a sophisticated continuous flow micro-reaction technology. By leveraging the enhanced heat and mass transfer capabilities of microchannel reactors, this method addresses longstanding challenges regarding safety, yield, and scalability. For R&D directors and supply chain leaders, understanding this technological leap is essential for securing a competitive edge in the production of high-value pharmaceutical intermediates. The patent details a seamless integration of reaction steps that not only improves operational efficiency but also significantly mitigates the risks associated with hazardous reagents typically found in legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methyl 2-amino-3-nitrobenzoate has been plagued by significant operational hurdles and safety concerns. Traditional pathways often rely on the Curtius rearrangement, which necessitates the use of sodium azide, a highly toxic and explosive compound that poses severe safety risks during handling and storage. Furthermore, alternative routes involving direct esterification of 2-amino-3-nitrobenzoic acid frequently suffer from prolonged reaction times and suboptimal yields, often hovering around 67% or lower. These batch processes struggle with heat dissipation during exothermic nitration steps, leading to the formation of unwanted byproducts and complicating downstream purification. The cumulative effect of these inefficiencies is a manufacturing process that is not only costly due to low material throughput but also environmentally burdensome due to the generation of complex waste streams. For procurement managers, these factors translate into volatile supply chains and increased costs for waste management and safety compliance.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111253271B utilizes a continuous flow system that fundamentally alters the reaction dynamics. By employing microchannel reactors, the process achieves rapid mixing and precise temperature control, which are critical for managing the exothermic nature of nitration and rearrangement reactions. This novel approach replaces dangerous batch operations with a streamlined, multi-step flow sequence that enhances intrinsic safety. The continuous removal of products from the reaction zone prevents over-reaction and degradation, thereby boosting overall yield and purity. This shift allows for a drastic simplification of the workflow, eliminating the need for hazardous azide chemistry entirely. For supply chain heads, this represents a move towards a more reliable and scalable production model where output can be increased simply by extending run times or numbering up reactor units, ensuring a steady supply of this critical intermediate without the bottlenecks typical of batch manufacturing.
Mechanistic Insights into Continuous Flow Micro-Reaction Technology
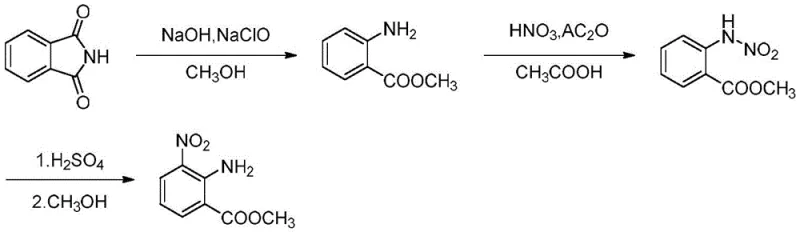
The core of this innovation lies in the precise orchestration of three distinct chemical transformations within a continuous flow environment. The process begins with the reaction of phthalimide and sodium hypochlorite in a microchannel reactor maintained at a低温 range of -5 to 15°C, followed by further reaction in a tubular reactor at 55 to 90°C. This initial step effectively generates methyl anthranilate through a Hofmann-like degradation pathway, where the microreactor's high surface-to-volume ratio ensures immediate heat removal, preventing thermal runaway. Subsequently, the methyl anthranilate undergoes nitration using acetic anhydride and fuming nitric acid within a second microchannel reactor at 18 to 35°C. The short residence time of 20 to 60 seconds in this stage is crucial for controlling the regioselectivity of the nitration, minimizing the formation of dinitro byproducts. Finally, the resulting N-nitrobenzoate intermediate is subjected to an acid-catalyzed rearrangement in the presence of sulfuric acid at 0 to 20°C. This Bamberger-like rearrangement converts the N-nitro group into the desired ortho-amino nitro configuration with high fidelity.
Impurity control is inherently superior in this flow system due to the plug-flow characteristics which ensure uniform residence time distribution for all fluid elements. In traditional batch reactors, gradients in concentration and temperature often lead to localized hot spots where side reactions proliferate. Here, the rapid quenching and separation immediately following each reaction stage halt the chemistry at the optimal point, preserving the integrity of the intermediate. The final recrystallization from methanol further refines the product, consistently delivering purity levels exceeding 99%. For R&D teams, this mechanistic precision means a cleaner impurity profile, which simplifies regulatory filing and reduces the burden on analytical quality control laboratories. The ability to fine-tune parameters such as flow rate and temperature in real-time provides an additional layer of process robustness that is unattainable with static batch vessels.
How to Synthesize Methyl 2-Amino-3-Nitrobenzoate Efficiently
The implementation of this continuous flow synthesis requires a systematic approach to reactor setup and parameter optimization to ensure maximum efficiency and safety. The process is designed to be modular, allowing for the independent control of each reaction stage to accommodate specific kinetic requirements. Detailed standard operating procedures regarding pump calibration, pressure monitoring, and emergency shutdown protocols are essential for successful deployment. The following guide outlines the standardized synthesis steps derived from the patent data to assist technical teams in replicating this high-performance route.
- Pump phthalimide and sodium hypochlorite solutions into a microchannel reactor at -5 to 15°C, followed by a tubular reactor at 55 to 90°C to obtain methyl anthranilate.
- Mix methyl anthranilate with acetic anhydride and fuming nitric acid in a microchannel reactor at 18 to 35°C to synthesize methyl N-nitrobenzoate.
- React methyl N-nitrobenzoate with sulfuric acid in a microchannel reactor at 0 to 20°C for rearrangement, followed by methanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this continuous flow technology offers profound commercial benefits that extend far beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. For procurement managers, the elimination of hazardous reagents like sodium azide removes a significant cost center associated with specialized handling, storage, and disposal of dangerous chemicals. The process intensification achieved through microreactors means that the same production volume can be achieved with a much smaller physical footprint, reducing capital expenditure on large-scale reactor vessels and containment infrastructure. Furthermore, the substantial improvement in yield translates directly into better raw material utilization, lowering the cost of goods sold per kilogram of the final intermediate. These efficiencies create a more competitive pricing structure, allowing suppliers to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or margin.
- Cost Reduction in Manufacturing: The transition to continuous flow eliminates the need for expensive safety measures required for batch azide chemistry and reduces solvent consumption due to higher concentration capabilities. By avoiding the low yields associated with traditional esterification methods, the overall material throughput is significantly enhanced, leading to substantial cost savings. The reduced reaction times also lower energy consumption for heating and cooling cycles, contributing to a leaner operational budget. Additionally, the simplified workup procedures minimize labor costs and reduce the downtime typically associated with cleaning and turning over batch reactors between runs.
- Enhanced Supply Chain Reliability: Continuous manufacturing provides a steady stream of product rather than the sporadic output of batch processing, which greatly stabilizes inventory levels and delivery schedules. The scalability of microreactor systems allows for rapid capacity expansion by adding more reactor modules rather than building new facilities, ensuring that supply can quickly adapt to market demand surges. This flexibility is crucial for maintaining the continuity of supply for critical ARB drug intermediates, mitigating the risk of stockouts that can disrupt downstream API production. The robustness of the process against minor fluctuations in conditions further ensures consistent quality, reducing the likelihood of batch rejections and supply delays.
- Scalability and Environmental Compliance: The inherent safety of the microreaction technology simplifies regulatory compliance regarding process safety management and environmental protection. The precise control over reaction conditions minimizes the generation of hazardous waste and byproducts, aligning with green chemistry principles and reducing the burden on wastewater treatment facilities. Scaling up from pilot to commercial production is straightforward and predictable, as the transport phenomena remain consistent regardless of the number of reactor units employed. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to meet global demand efficiently while adhering to stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility of adopting this technology for industrial production. Understanding these details is vital for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios.
Q: What are the safety advantages of using microreactors for this synthesis compared to batch processes?
A: The microreactor method eliminates the need for hazardous sodium azide used in traditional Curtius rearrangements and provides intrinsic safety through superior heat dissipation during exothermic nitration steps.
Q: How does the continuous flow process impact the purity of the final intermediate?
A: Precise control over residence time and temperature in the microchannel reactors minimizes side reactions and byproduct formation, consistently achieving purity levels greater than 99% after recrystallization.
Q: Is this method suitable for large-scale industrial production of ARB drug intermediates?
A: Yes, the process demonstrates excellent scalability with negligible amplification effects, allowing for annual production capacities reaching hundreds of tons using series-connected microreactor systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-Amino-3-Nitrobenzoate Supplier
The technological advancements detailed in patent CN111253271B underscore the immense potential of continuous flow chemistry in modern pharmaceutical manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is backed by rigorous QC labs and stringent purity specifications, ensuring that every batch of methyl 2-amino-3-nitrobenzoate meets the highest international standards. We understand the critical nature of this intermediate in the synthesis of life-saving ARB medications and are dedicated to providing a supply chain that is both resilient and responsive to the dynamic needs of the global pharmaceutical market.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our continuous flow expertise can optimize your supply chain. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and quality in your API production processes.
