Scaling Methyl 2-Amino-3-Nitrobenzoate Production via Advanced Continuous Flow Technology
The pharmaceutical industry is constantly seeking robust manufacturing pathways for critical intermediates like methyl 2-amino-3-nitrobenzoate, a key building block for angiotensin II receptor blockers such as candesartan and azilsartan. Patent CN111253271A introduces a groundbreaking continuous flow methodology that fundamentally transforms the production landscape for this high-value compound. By leveraging advanced micro-reactor technology, this innovation addresses longstanding challenges regarding safety, yield, and scalability that have plagued traditional batch synthesis. The disclosed method replaces hazardous batch operations with a streamlined, multi-step continuous process that ensures precise control over reaction parameters. This technical breakthrough not only enhances operational safety but also delivers substantial improvements in product purity and overall process efficiency. For global supply chain leaders, this represents a pivotal shift towards more reliable and sustainable manufacturing protocols for complex aromatic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methyl 2-amino-3-nitrobenzoate has been fraught with significant technical and safety hurdles that impede efficient large-scale production. Traditional routes often rely on the Curtius rearrangement involving hazardous sodium azide or direct esterification processes that suffer from prolonged reaction times and mediocre yields. For instance, prior art documents indicate that conventional batch esterification methods frequently achieve yields as low as 67%, necessitating extensive downstream purification to meet pharmaceutical grade standards. Furthermore, the handling of energetic intermediates and strong nitrating agents in large batch vessels poses inherent risks of thermal runaway and explosion. These safety concerns mandate expensive engineering controls and rigorous monitoring systems, driving up the capital expenditure and operational costs for manufacturers. The cumulative effect of these limitations is a supply chain that is vulnerable to disruptions and incapable of meeting the surging demand for antihypertensive medications globally.
The Novel Approach
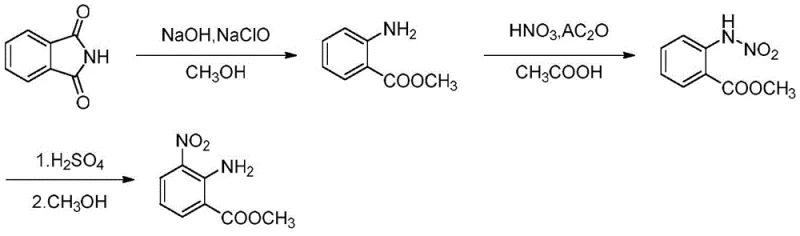
The novel approach detailed in the patent utilizes a sophisticated continuous flow system that integrates micro-channel reactors with tubular reactors to optimize every stage of the synthesis. This method initiates with a Hofmann rearrangement of phthalimide under tightly controlled低温 conditions, immediately followed by a nitration step that leverages the superior mixing capabilities of micro-fluidics. By confining the reaction volume to microscopic channels, the system achieves rapid heat dissipation, effectively neutralizing the exothermic risks associated with nitration and acid-catalyzed rearrangement. The continuous nature of the process allows for immediate quenching and separation of intermediates, drastically reducing the formation of unwanted by-products and degradation compounds. Consequently, this technological leap enables the production of high-purity intermediates with significantly reduced waste generation and energy consumption. The result is a manufacturing paradigm that is not only safer but also economically superior, offering a compelling value proposition for cost-conscious procurement strategies.
Mechanistic Insights into Micro-Reactor Catalyzed Synthesis
The core of this innovative synthesis lies in the precise manipulation of reaction kinetics through micro-reactor engineering, specifically during the critical nitration and rearrangement phases. The process begins by pumping a phthalimide solution and sodium hypochlorite into a micro-channel reactor maintained at temperatures between -5°C and 15°C, facilitating a controlled Hofmann rearrangement. The effluent then transitions into a tubular reactor heated to 55°C to 90°C, where the conversion to methyl anthranilate is completed with high selectivity. Subsequently, the methyl anthranilate undergoes nitration using fuming nitric acid and acetic anhydride within a micro-reactor at 18°C to 35°C, a step where traditional mixing would fail to control the heat release effectively. The final transformation involves an acid-catalyzed rearrangement of the N-nitro intermediate using concentrated sulfuric acid at 0°C to 20°C, yielding the target amino-nitro benzoate structure. This sequential flow architecture ensures that each reactive species encounters the optimal environment for conversion without accumulating thermal energy.
Impurity control is inherently superior in this continuous flow regime due to the minimization of residence time distribution and the prevention of local hot spots. In batch reactors, concentration gradients often lead to over-nitration or hydrolysis side reactions, generating difficult-to-remove impurities that compromise the quality of the final API intermediate. The micro-reactor system maintains a uniform concentration profile throughout the reaction channel, ensuring that the stoichiometry remains ideal at every point of the reaction trajectory. Furthermore, the immediate separation of the crude product via annular continuous centrifugation prevents prolonged exposure to acidic or oxidative conditions that could degrade the sensitive amino-nitro functionality. This mechanistic precision translates directly into a cleaner crude product profile, reducing the burden on the final crystallization step. For R&D directors, this means a more predictable impurity profile that simplifies regulatory filing and quality assurance processes.
How to Synthesize Methyl 2-Amino-3-Nitrobenzoate Efficiently
Implementing this continuous flow synthesis requires a systematic approach to equipment setup and parameter optimization to fully realize the benefits of micro-reaction technology. The process demands precise metering pumps to maintain the strict flow rate ratios defined in the patent, ensuring that the residence time in each reactor zone aligns with the kinetic requirements of the specific transformation. Operators must carefully monitor the temperature gradients across the micro-channel and tubular reactors to prevent any deviation that could trigger side reactions or safety incidents. The integration of inline separation units, such as the annular gap centrifuge mentioned in the examples, is critical for handling the solid-liquid separations efficiently without interrupting the continuous stream. Detailed standard operating procedures must be established to manage the startup and shutdown sequences, which differ significantly from traditional batch protocols.
- Pump phthalimide and sodium hypochlorite solutions into a microchannel reactor at -5 to 15°C, followed by a tubular reactor at 55 to 90°C to obtain methyl anthranilate.
- Mix methyl anthranilate with acetic anhydride and fuming nitric acid in a micro-reactor at 18 to 35°C to synthesize methyl N-nitrobenzoate.
- React methyl N-nitrobenzoate with concentrated sulfuric acid in a micro-reactor at 0 to 20°C, followed by methanol crystallization to isolate the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous flow technology offers transformative benefits that extend far beyond simple yield improvements. The shift from batch to continuous processing fundamentally alters the cost structure of manufacturing by reducing the footprint of the production facility and minimizing the inventory of hazardous intermediates held on-site. This lean manufacturing approach leads to substantial cost savings by eliminating the need for large-scale safety containment systems and reducing the energy load required for heating and cooling massive reaction vessels. Moreover, the enhanced safety profile reduces insurance premiums and regulatory compliance costs, further contributing to the overall economic efficiency of the supply chain. The ability to scale production simply by increasing run time or numbering up reactor units provides unparalleled flexibility in responding to market demand fluctuations without heavy capital investment.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents like sodium azide and the reduction in solvent usage significantly lower the raw material and waste disposal costs associated with production. By achieving higher conversion rates and selectivity, the process minimizes the loss of valuable starting materials, directly improving the cost of goods sold for the final intermediate. The continuous nature of the operation also reduces labor costs per unit of production, as the system requires less manual intervention compared to repetitive batch cycles. These cumulative efficiencies create a robust margin structure that allows suppliers to offer competitive pricing while maintaining high profitability. Ultimately, this cost optimization strengthens the supply chain resilience against raw material price volatility.
- Enhanced Supply Chain Reliability: The modular design of micro-reactor systems ensures that production can be maintained even if individual units require maintenance, preventing total line shutdowns common in single-vessel batch plants. The consistent quality output from continuous flow processes reduces the rate of batch failures and rejections, ensuring a steady flow of compliant material to downstream API manufacturers. This reliability is crucial for long-term supply agreements where consistency and on-time delivery are paramount contractual obligations. Additionally, the reduced reaction times allow for faster turnover of production slots, enabling suppliers to respond more agilely to urgent orders or unexpected demand spikes. Such operational agility is a key differentiator in the highly competitive pharmaceutical intermediate market.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with patent data indicating potential annual outputs reaching 100 tons using commercially available micro-reactor series, proving its viability for industrial application. The intensified reaction conditions lead to a significant reduction in solvent volumes and waste generation, aligning with increasingly stringent global environmental regulations and sustainability goals. By minimizing the use of chlorinated solvents and hazardous reagents, the process simplifies wastewater treatment and lowers the environmental impact of the manufacturing site. This green chemistry approach not only mitigates regulatory risk but also enhances the brand reputation of the supplier among environmentally conscious partners. Scalability combined with compliance ensures long-term viability and uninterrupted supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this continuous flow synthesis technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance.
Q: What are the safety advantages of using micro-reactors for this nitration process?
A: Micro-reactors offer intrinsic safety by managing highly exothermic nitration reactions through superior heat transfer, preventing thermal runaway risks associated with traditional batch reactors.
Q: How does the continuous flow method compare to traditional batch synthesis yields?
A: Patent data indicates that the continuous flow method achieves overall yields exceeding 74%, significantly outperforming traditional batch methods which often struggle to reach 40% efficiency due to side reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes scalable micro-reactor systems like the Chemtrix PlantrixMR series, capable of reaching annual outputs of 100 tons while maintaining consistent quality and purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-Amino-3-Nitrobenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging cutting-edge technologies like the continuous flow methods described in recent patents, we continuously optimize our manufacturing processes to enhance efficiency and safety. Our dedication to technical excellence makes us a trusted partner for pharmaceutical companies seeking reliable sources for complex organic intermediates.
We invite you to collaborate with us to explore how our advanced manufacturing capabilities can support your project goals and drive value for your organization. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our commitment to transparency and partnership. Let us help you secure a stable and cost-effective supply chain for your critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
