Scalable Production of High-Purity Dehydroepiandrosterone via Advanced Oil-Free Biotransformation
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the synthesis of critical steroid intermediates. A significant advancement in this domain is detailed in patent CN110656146A, which discloses a novel method for preparing dehydroepiandrosterone (DHEA) through the oil-free conversion of phytosterol by growing cells. This technology represents a paradigm shift from traditional chemical synthesis and oil-based microbial fermentation, addressing long-standing challenges regarding yield, purity, and environmental impact. By leveraging a specific strain of Mycobacterium sp. B-NRRL 3683 and a unique substrate protection strategy, this process achieves high conversion efficiency while eliminating the generation of waste oil, a common byproduct in conventional biotransformation. For global supply chains, this innovation offers a robust alternative for sourcing high-quality steroid precursors, ensuring that the production of downstream hormonal therapies remains both economically viable and ecologically responsible.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of dehydroepiandrosterone has relied heavily on multi-step chemical synthesis or microbial fermentation methods that require the addition of organic oils. Traditional chemical routes often involve the selective reduction of 3-position ketone groups from androstenedione, a process fraught with difficulties such as the formation of 3-position alpha-hydroxyl isomers and other impurities that complicate purification. Furthermore, these chemical methods frequently utilize hazardous organic reagents, leading to significant environmental pollution and high disposal costs. On the biological front, earlier fermentation technologies, such as those disclosed in patent CN201210316197.0, utilized oil-fermentation conversion. While effective to a degree, these oil-based systems suffer from prolonged fermentation times, difficult product separation due to the presence of emulsified oils, and the generation of substantial waste oil that requires complex treatment. These inefficiencies create bottlenecks in manufacturing throughput and inflate the overall cost of goods sold for pharmaceutical manufacturers.
The Novel Approach
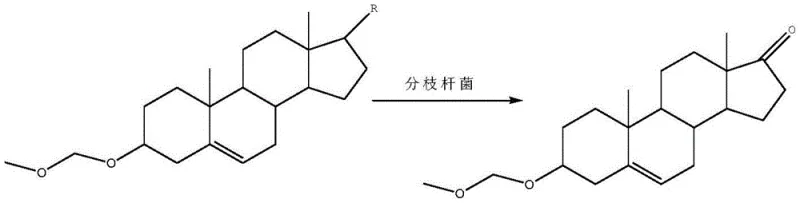
In stark contrast, the method described in CN110656146A introduces a streamlined, oil-free biotransformation process that fundamentally alters the substrate interaction with the microbial catalyst. The core innovation lies in the pre-protection of the 3-position hydroxyl group of the phytosterol raw material using methylal. This chemical modification dramatically increases the solubility and dispersibility of the substrate within the aqueous fermentation broth, allowing the growing cells of Mycobacterium sp. to access and convert the substrate much more efficiently without the need for oil carriers. This approach not only shortens the reaction route by omitting several post-treatment steps associated with oil removal but also significantly enhances the yield and purity of the final DHEA product. By transitioning to an aqueous-based system, the process inherently reduces the environmental footprint, aligning with modern green chemistry principles while delivering a superior economic profile for large-scale production.
Mechanistic Insights into Methylal-Protection and Biotransformation
The success of this synthetic route hinges on the precise chemical protection of the phytosterol substrate prior to biological exposure. The 3-position hydroxyl group on the steroid nucleus is highly reactive and prone to oxidation during the vigorous conditions of fermentation, which can lead to conversion failure or the formation of undesired byproducts. By reacting phytosterol with methylal (dimethoxymethane) in the presence of phosphorus pentoxide as a catalyst, the hydroxyl group is converted into a stable etherate. This protection step is critical because it not only safeguards the reactive site but also modifies the lipophilicity of the molecule, enabling it to disperse effectively in the water-based fermentation medium without the need for solubilizing oils. The use of diatomite as a filter aid during this protection phase further ensures that the resulting etherate is of high quality and free from particulate contaminants that could inhibit microbial growth.
Following protection, the biotransformation utilizes the specific enzymatic machinery of Mycobacterium sp. B-NRRL 3683 to cleave the side chain of the phytosterol derivative. Unlike genetically engineered strains that require complex construction and regulatory approval, this naturally occurring strain is optimized through process control parameters such as temperature, pH, and aeration to maximize the cleavage efficiency. The absence of oil in the fermentation tank prevents the formation of stable emulsions, which typically trap the product and reduce recovery rates. Instead, the product remains more accessible for extraction, leading to a cleaner fermentation broth. The subsequent hydrolysis step using hydrochloric acid efficiently removes the protecting group, regenerating the 3-beta-hydroxyl functionality essential for DHEA's biological activity. This seamless integration of chemical protection and biological catalysis ensures a high-fidelity conversion process.
How to Synthesize Dehydroepiandrosterone Efficiently
The synthesis of dehydroepiandrosterone via this oil-free method involves a carefully orchestrated sequence of protection, fermentation, and purification steps designed to maximize yield and minimize waste. The process begins with the etherification of phytosterol, followed by inoculation into a specialized transformation medium containing corn steep liquor and hydroxypropyl cyclodextrin to further enhance substrate availability. The fermentation is conducted under controlled aeration and pressure to maintain optimal cell growth and enzymatic activity. Once the biotransformation is complete, the product is extracted, hydrolyzed to remove the protecting group, and finally refined through crystallization to achieve pharmaceutical-grade purity. For a detailed breakdown of the specific reagent quantities, culture conditions, and operational parameters required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Protect the 3-position hydroxyl group of phytosterol using methylal and phosphorus pentoxide to enhance solubility and stability.
- Conduct biotransformation using Mycobacterium sp. B-NRRL 3683 in an aqueous fermentation medium without added oil.
- Perform hydrolysis with hydrochloric acid followed by crystallization and refining to obtain high-purity DHEA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oil-free biotransformation technology presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of organic oils from the fermentation process removes a major source of variability and complexity in downstream processing. Traditionally, separating the product from fermentation oil requires extensive solvent extraction and washing steps, which consume significant amounts of energy and raw materials. By removing this step, the new method drastically simplifies the workflow, leading to substantial cost savings in utilities and solvent procurement. Furthermore, the reduction in waste oil generation translates directly to lower environmental compliance costs and reduced liability associated with hazardous waste disposal, making the supply chain more resilient against tightening environmental regulations.
- Cost Reduction in Manufacturing: The streamlined nature of the oil-free process inherently lowers the cost of goods sold by reducing the consumption of organic solvents and energy-intensive separation equipment. Since the substrate is rendered soluble through chemical protection rather than oil emulsification, the volume of extraction solvents required is significantly decreased. Additionally, the higher conversion yield reported in the patent examples implies that less raw material is wasted per unit of finished product, optimizing the utilization of phytosterol feedstock. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on margin, providing a distinct advantage in the highly price-sensitive market for steroid intermediates.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are easy to obtain and process is critical for maintaining uninterrupted production schedules. Phytosterol, the primary feedstock for this method, is a widely available natural product, reducing the risk of supply shortages associated with specialized synthetic precursors. Moreover, the robustness of the Mycobacterium sp. strain and the simplicity of the aqueous fermentation medium reduce the likelihood of batch failures due to contamination or process instability. This reliability ensures consistent delivery timelines for downstream pharmaceutical customers, mitigating the risks of production delays that can ripple through the entire drug development and manufacturing pipeline.
- Scalability and Environmental Compliance: The transition from oil-based to oil-free fermentation significantly eases the burden of scaling up production from pilot to commercial volumes. The absence of viscous oil phases improves oxygen transfer rates in large fermenters, a common bottleneck in aerobic biotransformations, thereby facilitating smoother scale-up. From an environmental perspective, the drastic reduction in waste oil and organic solvent discharge aligns with global sustainability goals and strict effluent standards. This compliance capability future-proofs the manufacturing facility against evolving regulatory landscapes, ensuring long-term operational continuity and enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oil-free DHEA synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What is the primary advantage of the oil-free conversion method for DHEA?
A: The oil-free method eliminates the need for organic oil carriers in fermentation, significantly reducing waste oil pollution and simplifying the downstream separation and purification processes compared to traditional oil-fermentation techniques.
Q: How does the 3-position hydroxyl protection improve the yield?
A: Protecting the 3-position hydroxyl group with methylal prevents unwanted oxidation during fermentation and increases the substrate's solubility in the aqueous medium, leading to higher conversion rates and product purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available phytosterol raw materials and avoids complex genetic engineering or hazardous chemical reagents, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroepiandrosterone Supplier
The technological advancements presented in patent CN110656146A highlight the immense potential for optimizing steroid intermediate production through innovative biocatalysis. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex laboratory-scale protocols into robust, industrial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of high yield and purity observed in the lab are fully realized in mass production. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of dehydroepiandrosterone meets the exacting standards required by the global pharmaceutical industry.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for steroid intermediates. By leveraging our advanced fermentation and chemical synthesis capabilities, we can help you achieve significant efficiencies and cost reductions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
