Advanced Biocatalytic Synthesis of Pregna-5,7-diene-3β,21-diol for Commercial Pharmaceutical Manufacturing
Advanced Biocatalytic Synthesis of Pregna-5,7-diene-3β,21-diol for Commercial Pharmaceutical Manufacturing
The global demand for high-purity steroid intermediates continues to surge, driven by the expanding markets for hormonal therapies and Vitamin D analogs. A pivotal development in this sector is detailed in patent CN110951815A, which discloses a highly efficient method for preparing pregna-5,7-diene-3β,21-diol through the biological fermentation of ergosterol etherate using resting cells. This technology represents a significant paradigm shift from traditional multi-step chemical synthesis to a streamlined biocatalytic approach. By leveraging a specifically mutated strain of Mycobacterium sp. B-NRRL 3683, the process achieves precise side-chain cleavage while maintaining the stereochemical integrity of the steroid nucleus. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: a shorter synthetic route that minimizes impurity profiles and reduces reliance on hazardous chemical reagents. The structural complexity of the target molecule, as illustrated below, demands a synthesis strategy that balances selectivity with scalability.
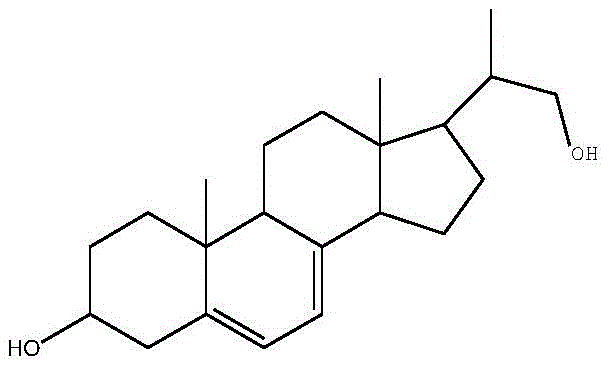
The core breakthrough lies in the strategic protection of the 3-hydroxyl group prior to fermentation. In conventional pathways, the presence of a free hydroxyl group can lead to non-specific oxidation or reduction by microbial enzymes, resulting in a mixture of 3-alpha and 3-beta isomers that are difficult and costly to separate. By converting ergosterol into its etherate form, the patent ensures that the biocatalyst focuses exclusively on the oxidative cleavage of the C17 side chain. This pre-emptive protection step not only enhances the regioselectivity of the biotransformation but also modifies the physicochemical properties of the substrate, making it more compatible with the fermentation medium when aided by solubility enhancers. Consequently, this method addresses the chronic pain points of low yield and poor product quality that have historically plagued the manufacturing of this critical pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pregna-5,7-diene-3β,21-diol has relied heavily on chemical methodologies that are fraught with inefficiencies. Traditional routes typically involve the selective chemical reduction of a 3-keto precursor to the desired 3-beta-hydroxyl configuration. However, achieving high stereoselectivity in this reduction step is notoriously challenging. Chemical reducing agents often produce a significant quantity of the thermodynamically stable 3-alpha-hydroxyl isomer as a byproduct. Separating these stereoisomers requires rigorous and repetitive purification steps, such as column chromatography or multiple recrystallizations, which drastically erode the overall process yield. Furthermore, these chemical methods frequently necessitate the use of expensive, moisture-sensitive reagents and generate substantial volumes of organic solvent waste, creating both economic and environmental burdens. The cumulative effect of these multi-step chemical transformations is a prolonged production cycle, increased operational costs, and a final product that may still contain trace levels of difficult-to-remove impurities, posing risks for downstream drug formulation.
The Novel Approach
In stark contrast, the methodology described in CN110951815A introduces a sophisticated chemo-enzymatic sequence that circumvents these stereochemical pitfalls. The process initiates with the protection of the ergosterol 3-hydroxyl group using methylal in the presence of phosphorus pentoxide. This reaction is elegantly simple yet highly effective, driving the equilibrium forward by removing the methanol byproduct through the dehydrating action of the catalyst. The resulting ergosterol etherate serves as an ideal substrate for the subsequent biotransformation. By masking the reactive hydroxyl group, the molecule becomes less polar and more soluble in the lipid phases of the fermentation broth, especially when formulated with hydroxypropyl-beta-cyclodextrin. This modification allows the resting cells to access the substrate more efficiently without interfering with the 3-position stereochemistry. The result is a direct, high-yield conversion to the protected intermediate, effectively bypassing the isomerization issues inherent in purely chemical reductions.
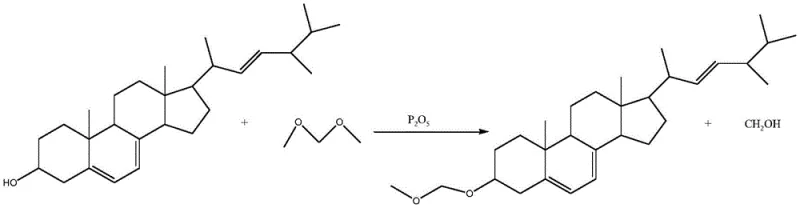
Mechanistic Insights into Resting Cell Biotransformation
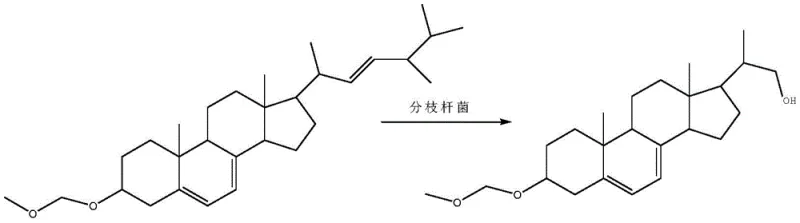
The heart of this innovative process is the utilization of a mutated Mycobacterium sp. B-NRRL 3683 strain in a resting cell configuration. Unlike growing cell fermentations where energy is diverted towards biomass accumulation, resting cells are metabolically primed to focus their enzymatic machinery solely on the bioconversion task. The patent details a rigorous mutagenesis protocol using nitrosoguanidine to enhance the strain's ability to cleave the C17 side chain of the protected ergosterol specifically. This targeted mutation is crucial because wild-type strains often lack the specificity or activity required for high-titer production of this specific diene-diol intermediate. The resting cells act as a reservoir of oxidases and lyases that systematically degrade the side chain while leaving the steroid nucleus intact. The inclusion of hydroxypropyl-beta-cyclodextrin in the fermentation broth is a masterstroke of process engineering; it forms inclusion complexes with the hydrophobic ergosterol etherate, effectively solubilizing the substrate in the aqueous buffer and preventing substrate inhibition or precipitation, which are common failure modes in steroid fermentations.
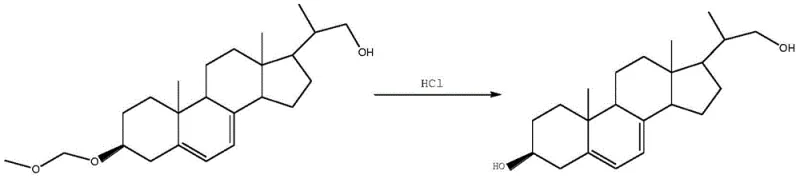
Following the biotransformation, the process employs a mild hydrolysis step to reveal the final active hydroxyl group. The protected intermediate is extracted and then treated with dilute hydrochloric acid under controlled thermal conditions. This deprotection step is designed to be orthogonal to the sensitive diene system in the B-ring of the steroid, ensuring that the conjugated double bonds remain untouched. The mechanism here relies on the acid-catalyzed cleavage of the acetal linkage formed during the initial protection phase. Because the protecting group was introduced to prevent enzymatic side-reactions rather than to withstand harsh chemical conditions, it can be removed under relatively mild acidic conditions (e.g., 5% HCl at 60°C). This gentleness is vital for preserving the integrity of the pregna-5,7-diene system, which is prone to isomerization or degradation under stronger acidic or basic conditions. The final refining step, involving crystallization from methanol and petroleum ether, capitalizes on the high purity achieved in the fermentation step, requiring minimal effort to reach pharmaceutical-grade specifications.
How to Synthesize Pregna-5,7-diene-3β,21-diol Efficiently
The synthesis of this high-value steroid intermediate via the patented resting cell method offers a robust framework for industrial adoption. The process integrates chemical protection with biological precision, requiring careful control over fermentation parameters such as pH, temperature, and aeration to maximize the activity of the mutated Mycobacterium strain. Operators must ensure the substrate is adequately solubilized using cyclodextrin prior to inoculation to prevent mass transfer limitations. The downstream processing is equally critical, utilizing standard liquid-liquid extraction followed by mild acid hydrolysis to recover the product. For a comprehensive understanding of the specific operating parameters, reagent ratios, and safety protocols required to replicate this high-yield pathway, please refer to the standardized technical guide below.
- Protect the 3-hydroxyl group of ergosterol using methylal and phosphorus pentoxide to form ergosterol etherate.
- Perform resting cell transformation using mutated Mycobacterium sp. B-NRRL 3683 in a buffered system with cyclodextrin.
- Extract the product, hydrolyze the protecting group with hydrochloric acid, and refine via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic route translates into tangible strategic advantages beyond mere technical feasibility. The primary benefit is the drastic simplification of the supply chain for raw materials. By starting from ergosterol, a widely available and renewable natural product derived from yeast, manufacturers can decouple their production from volatile petrochemical feedstocks. The elimination of multiple chemical reduction and purification steps significantly reduces the consumption of organic solvents and expensive chiral reagents, leading to a leaner, more cost-effective manufacturing process. Furthermore, the high stereoselectivity of the biological step means that less material is lost to isomeric byproducts, directly improving the mass balance and reducing the cost of goods sold (COGS). This efficiency gain is compounded by the reduced need for extensive wastewater treatment, as the fermentation broth is largely aqueous and biodegradable compared to the toxic effluents generated by traditional heavy metal-catalyzed reductions.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates several unit operations associated with traditional chemical synthesis, such as complex chromatographic separations and multiple recrystallizations required to remove alpha-isomers. By achieving high stereochemical purity directly through the enzymatic action of the resting cells, the need for costly chiral resolving agents and extensive purification infrastructure is substantially diminished. This reduction in processing intensity lowers both the capital expenditure (CAPEX) for equipment and the operational expenditure (OPEX) for labor and utilities. Additionally, the use of a mutated strain that offers higher conversion rates means that less starting material is required to produce the same amount of API intermediate, further driving down the variable costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: Relying on a biological fermentation process anchored by a robust, mutated bacterial strain provides a more stable and predictable supply source compared to chemical synthesis dependent on scarce reagents. The strain Mycobacterium sp. B-NRRL 3683 is well-characterized, and the mutagenesis protocol ensures a consistent biological catalyst that is less susceptible to the batch-to-batch variability often seen in complex organic synthesis. The ability to scale fermentation from liters to cubic meters using standard bioreactor technology ensures that production capacity can be ramped up quickly to meet surges in market demand without the long lead times associated with sourcing specialized chemical catalysts. This biological resilience adds a layer of security to the supply chain, mitigating risks associated with geopolitical disruptions in the chemical supply market.
- Scalability and Environmental Compliance: The process is inherently designed for green manufacturing, aligning with increasingly stringent global environmental regulations. The replacement of harsh chemical reducing agents with a water-based fermentation system significantly reduces the generation of hazardous waste streams. The downstream hydrolysis uses dilute hydrochloric acid, which is easily neutralized and managed, unlike the heavy metal waste from traditional catalytic hydrogenation or hydride reductions. This environmental compatibility simplifies the permitting process for new manufacturing facilities and reduces the long-term liability associated with waste disposal. Moreover, the high solubility of the substrate facilitated by cyclodextrin allows for higher substrate loading in the fermenter, improving the volumetric productivity of the bioreactors and making the process economically viable at a multi-ton commercial scale.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resting cell fermentation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the operational realities and potential hurdles. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers below clarify the specific advantages regarding impurity control, substrate solubility, and the scalability of the biological components.
Q: How does this biocatalytic method improve upon traditional chemical synthesis?
A: Traditional methods often rely on multi-step chemical reductions of 3-keto groups which suffer from poor stereoselectivity (generating unwanted 3-alpha isomers). This patent utilizes a specific resting cell fermentation of protected ergosterol, which inherently avoids the formation of these isomers and significantly shortens the synthetic route.
Q: What is the role of hydroxypropyl-beta-cyclodextrin in the fermentation process?
A: Hydroxypropyl-beta-cyclodextrin acts as a solubility enhancer. Since the protected ergosterol substrate has reduced polarity, the cyclodextrin facilitates its dispersion and solubility within the aqueous fermentation broth, thereby increasing the bioavailability of the substrate to the resting cells and improving overall conversion yields.
Q: Is this process scalable for industrial production of steroid intermediates?
A: Yes, the patent explicitly demonstrates scalability through examples ranging from small-scale flask cultures to 10L tank fermentations with substantial substrate loading (up to 10% ergosterol etherate). The use of robust resting cells and standard downstream processing (extraction, hydrolysis, crystallization) aligns well with existing industrial infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregna-5,7-diene-3β,21-diol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of fermentation parameters and downstream processing described in CN110951815A is maintained at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of Pregna-5,7-diene-3β,21-diol meets the most stringent purity specifications required by global regulatory bodies. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance ensures that your production schedules are never compromised by material shortages or specification failures.
We invite you to collaborate with us to optimize your supply chain for this critical steroid intermediate. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this biocatalytic route can lower your total landed cost. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-efficient source of high-purity intermediates, allowing you to focus on your core drug development goals while we manage the complexities of advanced manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
