Advanced Biocatalytic Synthesis of Pregna-5,7-diene-3β,21-diol for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and sustainable pathways for synthesizing complex steroid intermediates, particularly those serving as precursors for vital hormonal therapies. Patent CN110846370A introduces a groundbreaking methodology for the preparation of Pregna-5,7-diene-3β,21-diol, a critical intermediate in steroid medicine synthesis, by leveraging growth cell biological fermentation of ergosterol etherate. This innovative approach fundamentally shifts the paradigm from traditional multi-step chemical synthesis to a streamlined biocatalytic process that addresses long-standing challenges in stereoselectivity and yield. By integrating a specific 3-position protection strategy with the use of a mutated Mycobacterium sp. B-NRRL 3683 strain, the technology achieves precise side-chain cleavage while maintaining the integrity of the steroid nucleus. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates, offering a route that is not only chemically elegant but also economically viable for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing Pregna-5,7-diene-3β,21-diol have historically relied heavily on chemical transformations that are fraught with inefficiencies and purity issues. A primary bottleneck in conventional methods involves the manipulation of the 3-position ketone group, which typically requires selective chemical reduction to achieve the desired beta-hydroxyl configuration. However, chemical reduction agents often lack the absolute stereoselectivity required, resulting in the co-production of the 3-position alpha-hydroxyl isomer. This impurity profile necessitates rigorous and costly downstream purification steps to separate the isomers, significantly driving up production costs and reducing overall process yield. Furthermore, these chemical routes often involve multiple reaction steps and the use of expensive, environmentally hazardous reagents, creating substantial waste management burdens and complicating regulatory compliance for green manufacturing standards.
The Novel Approach
In stark contrast, the novel approach detailed in the patent circumvents these stereochemical pitfalls by employing a strategic protection-deprotection sequence coupled with highly specific enzymatic activity. By initially protecting the 3-hydroxyl group of ergosterol to form an etherate, the process effectively masks the reactive site, preventing unwanted side reactions during the subsequent fermentation. This modification not only enhances the solubility of the substrate in the fermentation medium, thereby improving bioavailability to the microorganism, but also ensures that the biocatalyst focuses exclusively on the oxidative cleavage of the C17 side chain. The result is a direct and clean conversion to the target intermediate with minimal formation of alpha-isomer impurities, drastically simplifying the purification workflow and enhancing the economic feasibility of the entire manufacturing process.
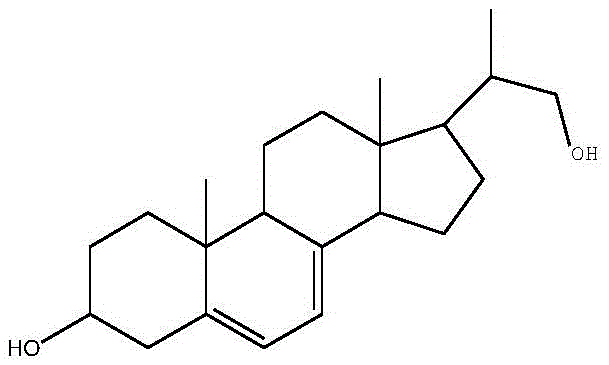
Mechanistic Insights into Mycobacterium-Mediated Side-Chain Cleavage
The core of this technological breakthrough lies in the sophisticated application of microbial biocatalysis using a specifically mutated strain of Mycobacterium sp. B-NRRL 3683. Unlike wild-type strains that may exhibit broad or non-specific metabolic activities, the mutated strain described in the patent has been engineered through nitrosoguanidine treatment to possess heightened specificity for the ergosterol etherate substrate. The mechanism involves the uptake of the protected ergosterol derivative by the growing cells, where specific oxygenase enzymes initiate the oxidative degradation of the aliphatic side chain at the C17 position. Crucially, the presence of the 3-position ether protecting group acts as a steric and electronic shield, ensuring that the enzymatic machinery does not inadvertently oxidize or modify the A-ring of the steroid nucleus. This precise molecular recognition allows for the accumulation of the desired diene-diol skeleton with exceptional fidelity, a feat that is difficult to replicate with purely chemical catalysts without extensive protecting group manipulations.
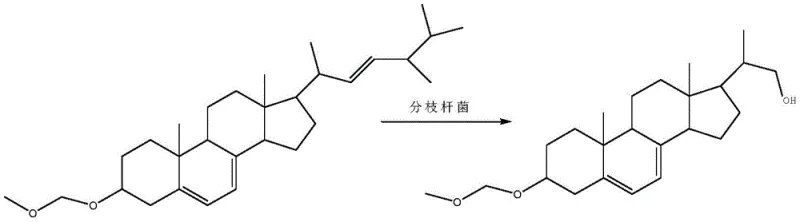
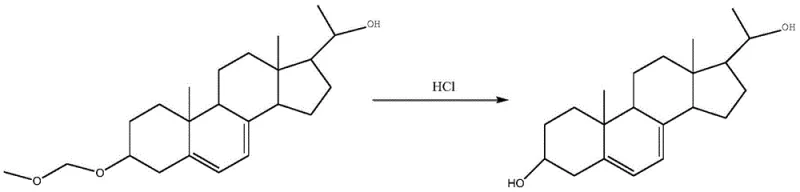
Following the biotransformation, the process employs a mild hydrolysis step to reveal the final active pharmacophore. The removal of the methylal protecting group is achieved under controlled acidic conditions, typically using hydrochloric acid in an ethyl acetate solvent system. This step is designed to be orthogonal to the sensitive diene system and the newly formed hydroxyl groups, ensuring that the structural integrity of the molecule is preserved during deprotection. The hydrolysis mechanism proceeds via protonation of the ether oxygen, followed by nucleophilic attack by water, regenerating the free 3-beta-hydroxyl group and releasing formaldehyde byproducts which are easily removed during workup. This final chemical step bridges the gap between the biotechnological fermentation and the isolation of the pure pharmaceutical intermediate, completing the hybrid chemo-enzymatic synthesis.
How to Synthesize Pregna-5,7-diene-3β,21-diol Efficiently
The synthesis protocol outlined in the patent provides a comprehensive framework for executing this high-efficiency transformation, emphasizing the critical parameters for strain maintenance and fermentation control. The process begins with the preparation of the ergosterol etherate substrate, followed by inoculation of the mutated Mycobacterium seed culture into a specialized transformation medium containing nutrients like corn steep liquor and soybean oil to support cell growth and substrate emulsification. Strict control of temperature, pH, and aeration during the fermentation phase is essential to maximize the expression of the side-chain cleavage enzymes while minimizing cellular stress. For a detailed breakdown of the specific operational parameters, media formulations, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Protect the 3-hydroxyl group of ergosterol using methylal and phosphorus pentoxide to form ergosterol etherate, enhancing substrate solubility.
- Perform biotransformation using a mutated Mycobacterium sp. B-NRRL 3683 strain in a optimized fermentation medium to cleave the side chain.
- Extract the fermented product, followed by acid hydrolysis to remove the protecting group and refine to obtain high-purity Pregna-5,7-diene-3β,21-diol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this biocatalytic route offers profound advantages for procurement managers and supply chain heads tasked with securing reliable sources of steroid intermediates. The most significant impact is observed in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply chain resilience. By eliminating the need for multiple chemical reduction and separation steps associated with traditional methods, manufacturers can significantly lower their consumption of expensive reagents and solvents. This reduction in material intensity not only lowers the direct cost of goods sold but also mitigates the risks associated with the supply volatility of specialized chemical catalysts, ensuring a more stable and predictable production schedule for high-volume API manufacturing.
- Cost Reduction in Manufacturing: The streamlined nature of the biocatalytic process inherently drives down manufacturing costs by consolidating multiple synthetic operations into a single fermentation step. The elimination of costly chiral reducing agents and the associated purification columns required to remove alpha-isomers results in substantial savings on raw material procurement. Furthermore, the use of renewable biological catalysts that can be propagated indefinitely reduces the dependency on finite chemical resources, creating a more sustainable and cost-effective production model that improves overall profit margins for the final drug product.
- Enhanced Supply Chain Reliability: Implementing a fermentation-based strategy diversifies the supply chain risk profile by reducing reliance on complex chemical synthesis networks that are often vulnerable to geopolitical or logistical disruptions. The raw materials required for this process, such as ergosterol and standard fermentation nutrients, are widely available commodities with stable global markets. This accessibility ensures that production can be scaled up rapidly to meet surging demand without the bottlenecks typically associated with sourcing specialized fine chemicals, thereby guaranteeing continuity of supply for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard industrial fermentation equipment that can be easily expanded from pilot to commercial scale. From an environmental standpoint, the reduction in hazardous chemical waste and the use of biodegradable fermentation byproducts align perfectly with increasingly stringent global environmental regulations. This eco-friendly profile not only simplifies waste disposal logistics and costs but also enhances the corporate sustainability credentials of the manufacturer, making the supply chain more attractive to environmentally conscious partners and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the mutated strain and the protection strategy employed in the patented method.
Q: How does the 3-position protection improve fermentation yield?
A: Protecting the 3-hydroxyl group reduces the polarity of the substrate, significantly increasing its solubility in the fermentation broth and vegetable oil phases. This enhanced bioavailability allows the Mycobacterium strain to access the substrate more efficiently, leading to higher conversion rates and reduced formation of unwanted isomers.
Q: What are the advantages of using the mutated Mycobacterium sp. B-NRRL 3683 strain?
A: The mutated strain, developed through nitrosoguanidine treatment, exhibits superior specificity for cleaving the C17 side chain of ergosterol derivatives while preserving the 3-beta-hydroxyl configuration. This eliminates the need for complex chemical reduction steps that typically generate difficult-to-separate alpha-isomer impurities.
Q: Is this biocatalytic process scalable for industrial production?
A: Yes, the process utilizes standard fermentation techniques with readily available raw materials like corn steep liquor and soybean oil. The elimination of harsh chemical reagents and the use of robust growing cells make the pathway highly suitable for large-scale commercial manufacturing with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregna-5,7-diene-3β,21-diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in CN110846370A for producing high-quality steroid intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Pregna-5,7-diene-3β,21-diol delivered meets the exacting standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this fermentation process, ensuring maximum yield and consistency for our global clientele.
We invite forward-thinking pharmaceutical companies to collaborate with us to unlock the full commercial potential of this advanced synthesis technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
