Advanced Biocatalytic Route for ADD Production: Technical Analysis and Commercial Scalability
Introduction to Patent CN110592169B: A Breakthrough in Steroid Biocatalysis
The pharmaceutical industry continuously seeks robust and efficient pathways for synthesizing critical steroid intermediates, and the technology disclosed in patent CN110592169B represents a significant advancement in this domain. This intellectual property details a sophisticated two-step microbial transformation method for producing Androst-1,4-diene-3,17-dione, commonly known as ADD, directly from phytosterol feedstocks. Unlike conventional chemical synthesis routes that rely on harsh reagents and multi-step protection-deprotection sequences, this biological approach leverages the enzymatic specificity of distinct bacterial strains to achieve high conversion rates under mild conditions. The core innovation lies in the sequential use of Mycobacterium sp. B-NRRL 3683 followed by Nocardia simplex, creating a telescoped process that eliminates the need for isolating the intermediate 4-androstene-3,17-dione (4-AD). For R&D directors and process chemists, this patent offers a compelling alternative to traditional dioscin-based synthesis, promising a streamlined workflow that aligns with modern green chemistry principles while maintaining rigorous quality standards required for hormone synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ADD has been plagued by significant inefficiencies inherent to both chemical and early biological methods. The traditional chemical route starting from dioscin involves a cumbersome eleven-step synthesis, requiring expensive reagents, extreme reaction conditions, and generating substantial hazardous waste, which drastically inflates the cost of goods sold and complicates environmental compliance. On the biological front, earlier microbial transformation attempts using strains like Mycobacterium fortuitum often suffered from poor conversion specificity and low yields, with some prior art reporting maximum transformation rates as low as 64 percent. These legacy processes frequently resulted in complex mixtures of by-products, necessitating energy-intensive purification steps that eroded profit margins. Furthermore, the inability to fully convert the side-chain cleavage products often left valuable 4-AD unreacted in the final mixture, representing a direct loss of raw material value and requiring additional downstream processing to separate the desired ADD from the unreacted precursor.
The Novel Approach
The methodology outlined in CN110592169B fundamentally restructures the production workflow by introducing a dual-strain cascade that maximizes substrate utilization and minimizes waste. By first employing Mycobacterium sp. B-NRRL 3683 to convert phytosterol into a mixture rich in 4-AD and ADD, and subsequently utilizing the oil layer of this fermentation broth as the direct substrate for Nocardia simplex, the process effectively telescopes two major reaction stages. This innovative strategy omits the independent extraction and purification of 4-AD, which is typically a bottleneck in steroid manufacturing. The result is a process characterized by strong reaction specificity and relatively quick conversion times, capable of handling high substrate concentrations up to 10 percent w/v. This approach not only simplifies the operational complexity but also ensures that the final product ADD achieves a purity exceeding 99.5 percent, thereby meeting the stringent specifications required for a reliable steroid intermediate supplier in the global market.
Mechanistic Insights into Dual-Strain Microbial Transformation
The success of this biocatalytic route hinges on the complementary enzymatic activities of the two selected microorganisms, which work in concert to degrade the sterol side chain and introduce the requisite double bonds. In the first stage, Mycobacterium sp. B-NRRL 3683 initiates the degradation of the phytosterol side chain at the C17 position, a complex oxidative process that typically involves multiple cytochrome P450 enzymes and oxygenases. This step is critical as it cleaves the aliphatic tail of the sterol to yield the C19 steroids 4-AD and ADD. The patent specifies precise fermentation conditions, maintaining temperatures between 28-32°C and controlling dissolved oxygen via an air flow of 0.1-1.0 vvm, which are vital for sustaining the high metabolic activity required for side-chain cleavage. Following this initial conversion, the broth is heated to 80°C to denature proteins and facilitate phase separation, allowing the recovery of an upper oil layer enriched with the steroid products. This physical separation step is ingenious as it concentrates the hydrophobic steroids while discarding the aqueous phase containing water-soluble metabolic by-products and residual nutrients.
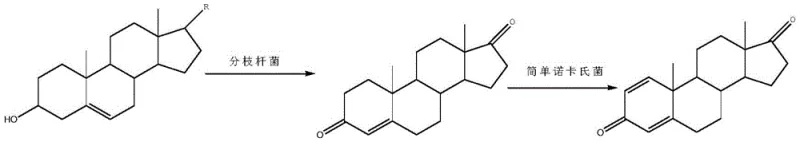
In the second stage, the oil layer serves as the substrate for Nocardia simplex, which possesses potent 1,2-dehydrogenase activity capable of converting the remaining 4-AD into the desired ADD. This enzymatic dehydrogenation introduces a double bond at the C1-C2 position of the steroid nucleus, completing the structural transformation. The use of Nocardia simplex is particularly advantageous due to its high specificity for this dehydrogenation reaction, minimizing the formation of over-oxidized by-products or degraded steroids. The process operates under similar mild thermal conditions (28-32°C), ensuring the stability of the sensitive steroid skeleton. From a mechanistic standpoint, the ability to drive this second conversion to completion without isolating the 4-AD intermediate suggests a high tolerance of the Nocardia strain to the complex matrix of the fermentation oil. This synergy between the two strains effectively creates a continuous flow of material from raw phytosterol to high-purity ADD, demonstrating a level of process integration that significantly enhances the overall atom economy and operational efficiency of the manufacturing line.
How to Synthesize ADD Efficiently
Implementing this dual-strain fermentation process requires careful attention to seed culture preparation and medium optimization to ensure consistent batch-to-batch performance. The patent details specific formulations for slant and shake flask cultures, emphasizing the importance of nitrogen sources like peptone and yeast extract, as well as carbon sources such as glucose and corn steep liquor, to build robust cell biomass before the main production fermentation. Operators must strictly adhere to the sterilization protocols, typically autoclaving at 121°C for 30 minutes, to prevent contamination which could derail the delicate microbial balance. The detailed standardized synthetic steps below outline the precise inoculation sizes, agitation speeds, and aeration rates necessary to replicate the high yields reported in the patent examples.
- Ferment phytosterol using Mycobacterium sp. B-NRRL 3683 at 28-32°C to generate a 4-AD and ADD mixture, then separate the upper oil layer by heating to 80°C.
- Utilize the crude oil layer directly as a substrate for Nocardia simplex fermentation at 28-32°C to convert residual 4-AD into ADD.
- Extract the final product using methanol precipitation and saponification, followed by recrystallization to achieve purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through cost reduction in pharmaceutical manufacturing. The elimination of the intermediate isolation step for 4-AD represents a massive reduction in solvent consumption and processing time, as there is no need for extensive extraction, drying, and recrystallization of the intermediate. This streamlining of the workflow translates to lower utility costs and reduced labor hours per kilogram of finished product. Furthermore, the use of microbial fermentation avoids the need for expensive transition metal catalysts or hazardous oxidizing agents often found in chemical synthesis, thereby reducing the costs associated with raw material procurement and hazardous waste disposal. The mild reaction conditions also imply lower energy requirements for heating and cooling compared to high-temperature chemical processes, contributing to a more sustainable and cost-effective production profile that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The telescoped nature of this process significantly lowers the cost of goods by removing unit operations. By avoiding the isolation of 4-AD, manufacturers save on the substantial volumes of organic solvents typically required for extraction and purification, as well as the energy needed to recover these solvents. Additionally, the high conversion specificity means less raw phytosterol is wasted as unreacted material or degraded by-products, maximizing the yield from every ton of starting material and driving down the effective cost per unit of active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Relying on robust microbial strains like Mycobacterium sp. and Nocardia simplex provides a stable and renewable production platform that is less susceptible to the volatility of petrochemical feedstock prices. The ability to use phytosterol, a widely available by-product of the vegetable oil industry, ensures a secure and diverse supply of raw materials. Moreover, the simplified process flow reduces the number of potential failure points in the manufacturing chain, leading to more predictable lead times and higher on-time delivery rates for customers seeking a reliable steroid intermediate supplier.
- Scalability and Environmental Compliance: The patent data demonstrates successful operation in 10-liter fermenters with high substrate loading, indicating strong potential for commercial scale-up of complex steroid intermediates to multi-ton scales. The aqueous nature of the fermentation media and the absence of heavy metals simplify wastewater treatment, making it easier for facilities to meet increasingly strict environmental regulations. This environmental compatibility reduces the risk of regulatory shutdowns and ensures long-term operational continuity for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual-strain biotransformation technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this method compares to existing industry standards. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing production infrastructure.
Q: How does this dual-strain method improve upon traditional single-strain fermentation?
A: Traditional methods often suffer from low specificity or incomplete conversion. This patent utilizes a sequential approach where Mycobacterium sp. performs the initial side-chain cleavage, and Nocardia simplex specifically targets the remaining 4-AD for dehydrogenation, significantly boosting overall conversion specificity and final purity.
Q: What are the critical control parameters for the fermentation steps?
A: Temperature control between 28-32°C and dissolved oxygen management (0.1-1.0 vvm air flow) are critical. Additionally, the separation of the oil layer at 80°C prior to the second fermentation step is essential to remove aqueous impurities and concentrate the substrate for the second strain.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful operation in 10-liter tanks with high substrate loading (up to 10% phytosterol). The omission of intermediate isolation steps reduces solvent usage and processing time, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ADD Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced biocatalytic routes like the one described in CN110592169B for securing the supply of critical hormone precursors. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for ADD, ensuring that every batch meets the >99.5 percent purity benchmark demonstrated in the patent. We are committed to delivering high-purity ADD that supports the uninterrupted manufacturing of downstream hormonal therapies.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized fermentation route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient, green, and cost-effective technology into your API supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
