Revolutionizing Corticosteroid Production: A Scalable Biocatalytic Route for Prednisone Acetate
Revolutionizing Corticosteroid Production: A Scalable Biocatalytic Route for Prednisone Acetate
The pharmaceutical industry is constantly seeking sustainable and economically viable pathways for the synthesis of critical adrenocortical hormones like prednisone acetate. A significant technological breakthrough is documented in patent CN112342261B, which outlines a novel preparation method that strategically combines microbial fermentation with targeted chemical oxidation. This approach fundamentally shifts the paradigm from traditional, waste-intensive chemical synthesis to a hybrid biocatalytic process. By utilizing pregn-4-ene-17 alpha, 21-diol-3, 20-dione-21-acetate (RSA) as the foundational raw material, the method leverages existing industrial infrastructure that has become underutilized, thereby transforming a potential liability into a high-value asset. The core innovation lies in the sequential application of fermentation hydroxylation, fermentation dehydrogenation, and a final oxidation reaction, achieving high conversion rates and exceptional purity suitable for global regulatory standards.
This integrated pathway not only addresses the environmental concerns associated with heavy metal usage in traditional routes but also drastically simplifies the supply chain by eliminating the need for complex iodine substitution steps. For R&D directors and procurement managers, this patent represents a tangible opportunity to reduce the cost of goods sold (COGS) while enhancing the sustainability profile of their steroid portfolios. The ability to achieve a purity of up to 99.4% through this streamlined process underscores its viability for commercial-scale manufacturing of high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of prednisone acetate has relied heavily on either biotransformation of cortisone acetate or fully chemical synthesis starting from dehydropregnenolone acetate. The traditional chemical synthesis routes are notoriously cumbersome, often involving an eight-step reaction sequence that includes the use of elemental iodine for substitution reactions. The reliance on iodine introduces significant economic and environmental burdens; elemental iodine is a high-cost reagent, and its usage generates substantial hazardous waste, complicating disposal and increasing the overall environmental footprint of the manufacturing process. Furthermore, the multi-step nature of these chemical routes inherently accumulates impurities at each stage, necessitating rigorous and costly purification protocols to meet pharmacopeial standards. The complexity of these legacy processes often results in lower overall yields and higher production lead times, creating bottlenecks for supply chain heads who require consistent and reliable volumes of active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the method disclosed in patent CN112342261B introduces a streamlined strategy that revitalizes the use of RSA, a raw material whose production facilities have largely been idled in the industry. By repurposing these existing assets, manufacturers can bypass the capital-intensive requirements of setting up entirely new production lines. The novel approach replaces the lengthy chemical sequences with two highly specific biological fermentation steps followed by a single, controlled oxidation. This hybrid model capitalizes on the regioselectivity of microorganisms to install critical functional groups—specifically the 11-alpha-hydroxyl and the 1,2-double bond—with precision that chemical catalysts struggle to match without expensive protecting group strategies. Consequently, this route offers a shorter synthetic timeline, reduced solvent consumption, and a markedly lower generation of hazardous by-products, positioning it as a superior alternative for cost reduction in corticosteroid manufacturing.
Mechanistic Insights into Biocatalytic Hydroxylation and Oxidation
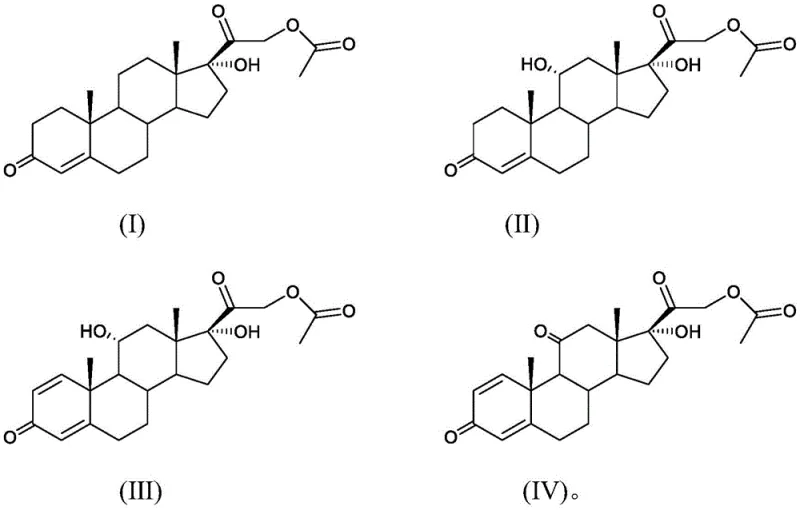
The success of this synthesis hinges on the precise orchestration of biological and chemical transformations. The first critical stage involves the microbial hydroxylation of Compound (I) using strains such as Aspergillus ochraceus. This biocatalytic step is engineered to introduce a hydroxyl group at the 11-alpha position of the steroid nucleus, a transformation that is chemically challenging to achieve with high selectivity. The fermentation process is meticulously controlled, with parameters such as temperature (25-35°C), airflow, and agitation speed optimized to maximize the metabolic activity of the fungus. Following this, the intermediate Compound (II) undergoes a second fermentation using Arthrobacter simplex. This step is responsible for the dehydrogenation reaction, introducing the characteristic 1,2-double bond in the A-ring of the steroid structure. The synergy between these two biological steps ensures that the complex stereochemistry of the molecule is established early in the process, minimizing the risk of isomeric impurities that could compromise the efficacy of the final drug product.
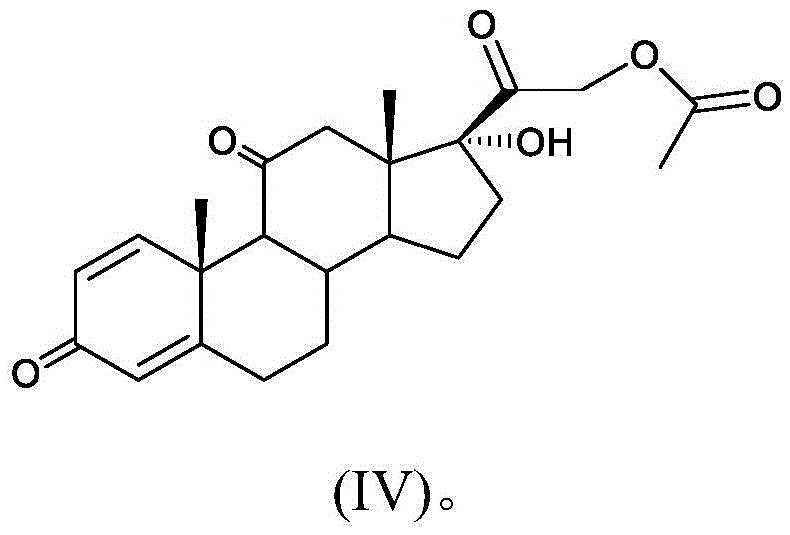
The final chemical transformation involves the oxidation of the 11-alpha-hydroxyl group to a ketone to yield the final prednisone acetate (Compound IV). This is achieved using a Jones reagent (chromic anhydride in sulfuric acid) under strictly controlled low-temperature conditions, typically between -10°C and 0°C. The低温 environment is crucial to prevent over-oxidation or degradation of the sensitive steroid backbone. The mechanistic precision here ensures that the 11-keto functionality, essential for the anti-inflammatory activity of prednisone, is formed cleanly. This step demonstrates how combining robust chemical oxidation with preceding biological specificity creates a powerful synthesis platform. The result is a molecule with a defined impurity profile, where the HPLC area normalization content can reach up to 99.4%, validating the robustness of this mechanistic approach for producing high-purity pharmaceutical intermediates.
How to Synthesize Prednisone Acetate Efficiently
Implementing this synthesis route requires strict adherence to the fermentation parameters and oxidation conditions detailed in the patent to ensure reproducibility and safety. The process begins with the preparation of seed cultures and fermentation media, followed by the stepwise addition of substrates under sterile conditions. The transition from biological to chemical processing must be managed carefully to maintain product integrity. For technical teams looking to adopt this methodology, the following guide outlines the critical operational phases derived from the patent examples. Detailed standardized synthesis steps are provided in the section below to facilitate immediate process evaluation.
- Perform first biological fermentation using Aspergillus ochraceus to hydroxylate compound (I) at the 11-alpha position, yielding compound (II).
- Conduct second biological fermentation using Arthrobacter simplex to introduce the 1,2-double bond via dehydrogenation, forming compound (III).
- Execute a controlled Jones oxidation reaction on compound (III) at low temperatures (-10 to 0°C) to convert the 11-alpha-hydroxyl group to a ketone, finalizing prednisone acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented method offers compelling strategic advantages beyond mere technical feasibility. The primary value driver is the significant reduction in raw material costs achieved by utilizing RSA, a compound that is industrially available but currently underutilized. By tapping into this existing supply, companies can avoid the volatility associated with sourcing specialized precursors required for traditional iodine-based synthesis. Furthermore, the elimination of the iodine substitution step removes a major cost center related to both reagent purchase and hazardous waste disposal. This shift towards a greener, bio-based workflow aligns with modern ESG (Environmental, Social, and Governance) goals, potentially reducing regulatory compliance costs and enhancing the marketability of the final product as sustainably sourced.
- Cost Reduction in Manufacturing: The economic benefits of this route are substantial, primarily driven by the simplification of the synthetic sequence. By replacing multiple chemical steps with efficient fermentation processes, the consumption of organic solvents and energy is drastically lowered. The avoidance of expensive iodine reagents and the associated waste treatment protocols translates directly into improved margins. Additionally, the high conversion rates reported in the fermentation steps (up to 95%) mean that less raw material is wasted, further optimizing the cost structure. This efficiency allows for a more competitive pricing strategy in the global market for corticosteroid intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of multi-step chemical syntheses, where a failure in any single step can halt production. This biocatalytic route mitigates such risks by relying on robust microbial strains that are less prone to the variability seen in complex chemical catalysis. The use of RSA as a starting material also diversifies the supply base, as it leverages established production capacities that are currently idle, ensuring a steady flow of inputs. This resilience is critical for maintaining consistent delivery schedules to downstream pharmaceutical customers, reducing the risk of stockouts and production delays.
- Scalability and Environmental Compliance: Scaling up biological processes is generally more straightforward than scaling complex chemical reactions involving hazardous reagents. The fermentation steps described operate under mild conditions (ambient pressure and moderate temperatures), which reduces the engineering demands on production facilities. Moreover, the reduced reliance on heavy metals and halogenated reagents simplifies the effluent treatment process, making it easier to meet stringent environmental regulations. This ease of compliance facilitates faster approval for commercial scale-up of complex steroid intermediates, allowing manufacturers to respond quickly to market demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this prednisone acetate synthesis route. These insights are derived directly from the experimental data and beneficial effects described in patent CN112342261B, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What is the primary advantage of using RSA as a starting material for prednisone acetate?
A: Using pregn-4-ene-17 alpha, 21-diol-3, 20-dione-21-acetate (RSA) allows manufacturers to utilize existing, often idle industrial production facilities. This repurposing significantly lowers capital expenditure compared to building new lines for traditional chemical synthesis routes that require expensive iodine substitution steps.
Q: How does the biocatalytic route improve impurity profiles compared to chemical synthesis?
A: The enzymatic specificity of strains like Aspergillus ochraceus and Arthrobacter simplex ensures highly regioselective hydroxylation and dehydrogenation. This biological precision minimizes the formation of structural isomers and by-products common in harsh chemical conditions, resulting in a crude product with purity exceeding 99% before final refinement.
Q: Is the Jones oxidation step scalable for industrial production?
A: Yes, the patent details a robust oxidation protocol using Jones reagent at controlled low temperatures (-10 to 0°C). While chromium-based oxidations require careful waste management, the process is streamlined into a single step following fermentation, avoiding the multi-step chemical sequences of older methods, thus maintaining overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in CN112342261B for the global supply of corticosteroids. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle both the fermentation and chemical oxidation stages of this synthesis, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We are committed to helping our clients navigate the complexities of steroid synthesis, offering a partnership that combines technical expertise with reliable manufacturing capacity.
We invite pharmaceutical companies and chemical distributors to explore how this advanced synthesis method can optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality prednisone acetate and related intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
