Scaling High-Purity Prednisolone Production via Optimized Low-Temperature Hydrolysis
The pharmaceutical industry constantly seeks robust manufacturing routes for critical corticosteroid intermediates, and the technology disclosed in patent CN100375751C offers a compelling solution for the production of prednisone hydrolyzate, commonly known as prednisolone. This specific intellectual property details a refined hydrolysis process that transforms prednisone acetate into the active hydrolyzed form using a sophisticated mixed-solvent system. Unlike traditional methods that rely on单一 solvents and ambient conditions, this innovation utilizes a precise blend of chloroform and methanol alongside a potassium hydroxide catalyst at strictly controlled low temperatures. The strategic adjustment of reaction parameters addresses long-standing inefficiencies in steroid processing, specifically targeting the enhancement of both yield and product quality. For R&D directors and procurement specialists, understanding this shift from conventional room-temperature saponification to a cryogenic mixed-solvent approach is vital for evaluating potential supply chain partnerships. The data indicates a consistent ability to achieve high purity levels, making this route highly attractive for the commercial scale-up of complex pharmaceutical intermediates where impurity profiles are strictly regulated.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of prednisolone from its acetate precursor relied heavily on using methanol as a single solvent with solid potassium hydroxide acting as the catalyst at room temperature, typically around 24-26°C. This traditional approach suffers from significant thermodynamic and kinetic drawbacks, primarily due to the poor solubility characteristics of the lipophilic steroid nucleus in pure methanol, necessitating excessive solvent volumes to drive the reaction to completion. Furthermore, operating at ambient temperatures increases the risk of unwanted side reactions, such as epimerization or degradation of the sensitive conjugated enone system located in Ring A of the steroid structure. The use of solid base catalysts also introduces heterogeneity issues, leading to inconsistent reaction rates and difficulties in heat dissipation, which ultimately compromises the uniformity of the final batch. These factors collectively result in lower overall yields and a product quality that often requires extensive downstream purification, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative technique described in the patent fundamentally alters the reaction environment by introducing a chloroform-methanol mixed solvent system with a specific volume ratio ranging from 1.10 to 1.15. This binary solvent mixture creates an optimal polarity balance that significantly enhances the solubility of the steroid substrate while maintaining the necessary conditions for effective nucleophilic attack by the hydroxide ion. By switching to a potassium hydroxide methanol solution and lowering the reaction temperature to a precise window of 0-5°C, the process minimizes thermal stress on the molecule, effectively suppressing degradation pathways. This controlled environment allows for a rapid reaction completion within approximately 30 minutes, followed by a streamlined workup involving neutralization, concentration, and crystallization. The result is a dramatic improvement in process efficiency, delivering yields consistently above 83% and purity levels exceeding 97%, which represents a substantial leap forward in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Base-Catalyzed Ester Hydrolysis
The core chemical transformation in this process is a classic base-catalyzed ester hydrolysis, often referred to as saponification, where the acetate group at the C21 position is cleaved to reveal the primary alcohol. In this specific steroid context, the mechanism involves the nucleophilic attack of the hydroxide ion on the carbonyl carbon of the ester linkage, forming a tetrahedral intermediate. The presence of chloroform in the solvent mixture plays a crucial role beyond simple solubility; it likely stabilizes the transition state through specific solvent-solute interactions, facilitating the breakdown of the intermediate into the carboxylate salt and the free alcohol. The strict temperature control at 0-5°C is mechanistically significant because it lowers the energy available for competing reactions, such as the base-catalyzed isomerization of the double bonds in the A-ring, which would otherwise generate difficult-to-remove impurities. This precision ensures that the reaction proceeds selectively at the ester moiety without disturbing the pharmacophore essential for biological activity.
Controlling the impurity profile is paramount in steroid synthesis, and this low-temperature protocol offers superior selectivity compared to ambient methods. At higher temperatures, the basic conditions could promote aldol-like condensations or retro-aldol reactions given the presence of ketone functionalities at C3 and C20. By maintaining the system at near-freezing conditions, the kinetic energy of the molecules is restricted, effectively 'freezing out' these high-energy degradation pathways. Additionally, the use of a liquid catalyst solution ensures homogeneous mixing, preventing localized hot spots of high pH that could degrade the sensitive steroid backbone. This results in a cleaner crude product, simplifying the subsequent crystallization steps and ensuring that the final API intermediate meets stringent regulatory specifications for related substances. The structural integrity of the molecule is preserved, as evidenced by the high assay values reported in the experimental data.
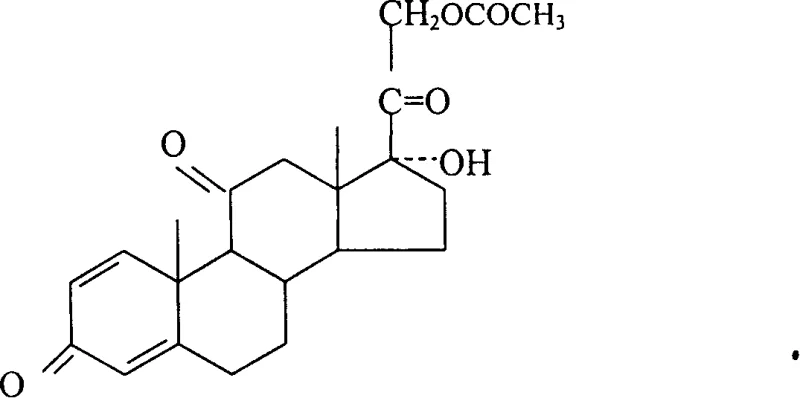
How to Synthesize Prednisolone Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and thermal management to replicate the high yields observed in the patent literature. The process begins with the dissolution of the raw material in the optimized chloroform-methanol mixture, ensuring a clear solution before the introduction of the catalyst. The addition of the base must be controlled to maintain the exotherm within the narrow 0-5°C range, which is critical for product quality. Following the reaction, the workup procedure involves neutralization to quench the base, followed by concentration to induce crystallization. The detailed standardized synthetic steps, including specific charging capacities and stirring times, are outlined in the guide below for technical teams evaluating feasibility.
- Dissolve prednisone acetate raw material in a mixed solvent of chloroform and methanol with a specific volume ratio of 1.10 to 1.15 to ensure complete solubility.
- Add a 4% to 6% potassium hydroxide methanol solution as the catalyst while maintaining the reaction temperature strictly between 0°C and 5°C.
- After reacting for 30 minutes, neutralize the mixture, concentrate to crystallize, then cool, filter, and dry to obtain high-purity prednisolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized hydrolysis process translates directly into tangible operational benefits and risk mitigation. The shift from a high-volume single-solvent system to a mixed-solvent approach significantly reduces the total volume of organic solvents required per kilogram of product, which lowers both raw material costs and waste disposal expenses. Furthermore, the improved yield consistency reduces the variability in production planning, allowing for more accurate forecasting and inventory management. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in ambient temperature, enhancing the reliability of supply during different seasons or in varying geographic manufacturing locations. These factors combine to create a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of excessive solvent usage and the improvement in yield directly contribute to a lower cost of goods sold (COGS). By achieving higher conversion rates with less raw material waste, manufacturers can realize substantial cost savings without compromising on quality. Additionally, the simplified purification process reduces the consumption of auxiliary chemicals and energy required for extensive recrystallization or chromatography. This economic efficiency makes the final product more competitive in the marketplace, offering better margins for downstream drug manufacturers who rely on these critical intermediates for their own formulations.
- Enhanced Supply Chain Reliability: The use of common, commercially available reagents such as chloroform, methanol, and potassium hydroxide ensures that the supply chain is not dependent on exotic or scarce catalysts. This availability mitigates the risk of production stoppages due to raw material shortages, a critical consideration for long-term supply agreements. The robustness of the process also implies a lower rate of batch failures, ensuring a steady flow of material to customers. This reliability is essential for maintaining continuous production schedules in the fast-paced pharmaceutical industry, where delays can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as mixing, cooling, and filtration that are easily transferable from pilot plant to commercial scale. The reduction in solvent volume also aligns with green chemistry principles by minimizing the environmental footprint associated with solvent recovery and waste treatment. This compliance with environmental regulations reduces the administrative burden and potential liabilities associated with hazardous waste disposal. Consequently, manufacturers can scale up production to meet increasing demand while adhering to strict environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of prednisolone using this specific hydrolysis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the process. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios or sourcing strategies.
Q: Why is a chloroform-methanol mixed solvent preferred over pure methanol?
A: The mixed solvent system improves the solubility of the lipophilic steroid backbone while maintaining the necessary polarity for the hydrolysis reaction, significantly reducing the total solvent volume required compared to single-solvent methods.
Q: What is the critical temperature range for this hydrolysis reaction?
A: The reaction must be maintained at a low temperature between 0°C and 5°C to prevent degradation of the sensitive enone system in the steroid ring and to minimize side reactions, ensuring high product purity.
Q: How does this process impact the final yield of prednisolone?
A: By optimizing solvent ratios and controlling reaction temperature, this method achieves yields exceeding 83%, representing a significant improvement over traditional room-temperature processes which suffer from lower conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain, and our expertise aligns perfectly with the advanced synthesis routes described in recent patents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of prednisolone meets the exacting standards required for hormonal therapies. Our capability to implement low-temperature hydrolysis processes demonstrates our technical depth and commitment to delivering superior chemical solutions.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our advanced process technologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential of this optimized hydrolysis method for your projects. Let us be your partner in driving efficiency and quality in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
