Advanced Synthetic Route for Methylprednisolone: Enhancing Purity and Commercial Scalability for Global API Manufacturers
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of high-value corticosteroids, and the synthesis of methylprednisolone stands as a prime example of process innovation. Patent CN101418029A discloses a groundbreaking method that fundamentally alters the traditional approach to constructing the critical C21 side-chain functionality required for this potent anti-inflammatory agent. By shifting away from conventional iodination strategies, this technology introduces a streamlined three-step sequence involving catalytic bromination, acetate substitution, and hydrolysis, delivering exceptional yields and purity profiles. The structural complexity of methylprednisolone, characterized by its specific stereochemistry at the C6, C11, and C17 positions as depicted below, demands precise chemical control to avoid isomeric impurities that could compromise therapeutic efficacy.
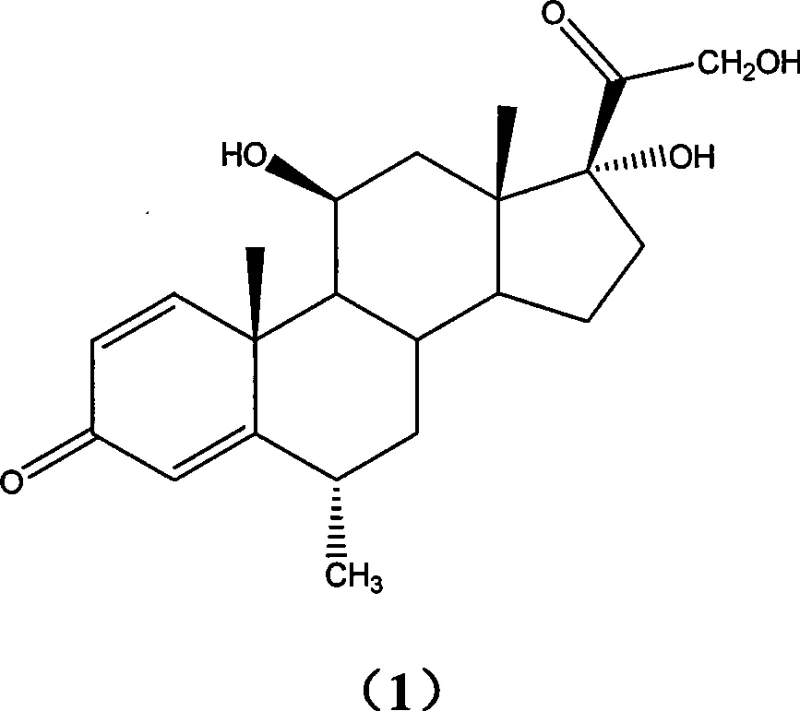
For R&D directors and process chemists evaluating this technology, the significance lies in the replacement of hazardous and costly reagents with benign, commercially available alternatives. The method utilizes 6-alpha-methyl-11-beta,17-alpha-dihydroxypregn-1,4-diene-3,20-dione as the starting material, subjecting it to a highly selective bromination using N-bromosuccinimide or similar agents under the influence of an ammonium salt catalyst. This specific catalytic system is not merely an additive but a critical enabler that drives the reaction to completion with yields ranging from 80% to 99.5%, thereby establishing a reliable foundation for subsequent transformations. The ability to isolate a stable 21-bromo intermediate without extensive purification represents a major operational advantage, simplifying the workflow and reducing solvent consumption in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of methylprednisolone has been plagued by convoluted synthetic routes that involve multiple protection and deprotection steps, biological oxidations, and the use of prohibitively expensive reagents. Prior art, such as the methods referenced in GB 2318790, often relies on complex sequences starting from 6-methyl progesterone acetate, requiring microbial hydroxylation followed by chemical dehydrogenation and side-chain modification using liquid bromine or iodine. These legacy processes are fraught with inefficiencies, including low overall yields, the generation of toxic by-products, and the necessity for harsh reaction conditions that complicate waste treatment and environmental compliance. Furthermore, the reliance on iodine-based reagents for side-chain functionalization imposes a significant financial burden due to the high cost of iodine and the poor atom economy associated with iodination by-products, making cost reduction in API manufacturing a persistent challenge for producers adhering to these outdated protocols.
The Novel Approach
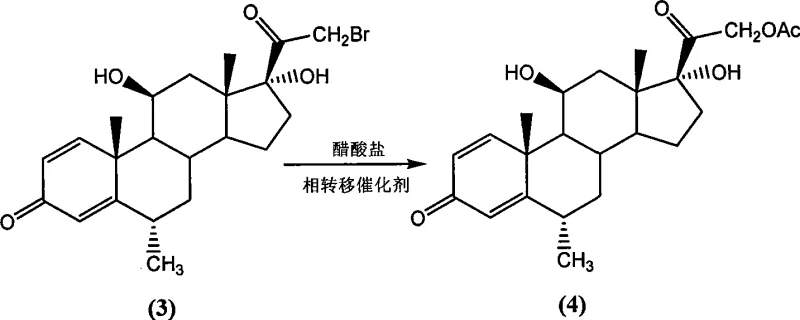
In stark contrast, the methodology outlined in CN101418029A offers a direct and efficient pathway that bypasses the need for complex protecting group chemistry and biological fermentation steps. The core innovation involves a direct bromination of the C21 position followed by a phase-transfer catalyzed substitution with acetate, as illustrated in the reaction scheme below, which seamlessly converts the bromo-intermediate into the acetoxy precursor. This approach not only eliminates the use of expensive iodine but also leverages the superior reactivity and stability of bromine derivatives, allowing for milder reaction temperatures and shorter processing times. The transition from the bromo-intermediate to the acetate derivative is achieved with remarkable efficiency, utilizing common acetate salts and catalytic amounts of phase-transfer agents, which facilitates the reaction in homogeneous or biphasic systems with yields consistently exceeding 75% and often reaching near-quantitative levels under optimized conditions.
Mechanistic Insights into Ammonium Salt-Catalyzed Bromination and PTC Substitution
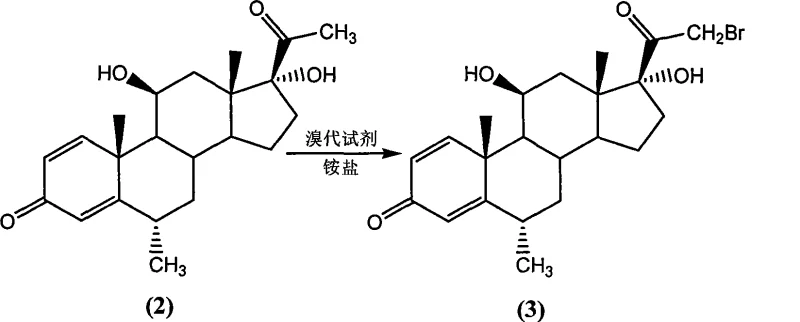
The mechanistic elegance of this synthesis lies in the dual catalytic systems employed to drive the key transformations with high selectivity. In the initial bromination step, the inclusion of an ammonium salt, such as ammonium acetate or ammonium formiate, acts as a crucial promoter that activates the brominating reagent, likely N-bromosuccinimide, towards the enolizable C21 position of the steroid nucleus. Without this catalyst, the patent explicitly notes that the reaction fails to proceed or results in negligible conversion, highlighting the unique role of the ammonium cation in stabilizing the transition state or facilitating the generation of the active brominating species. This catalytic effect ensures that the bromination occurs exclusively at the desired C21 position without affecting the sensitive C11 hydroxyl group or the conjugated diene system in Ring A, thereby preserving the structural integrity of the steroid backbone and minimizing the formation of regio-isomeric impurities that are difficult to remove downstream.
Following the bromination, the subsequent nucleophilic substitution utilizes a phase-transfer catalyst (PTC) to bridge the solubility gap between the organic-soluble bromo-steroid and the inorganic acetate salt. The quaternary ammonium salt PTC transports the acetate anion into the organic phase, where it attacks the C21 carbon, displacing the bromide leaving group in an SN2-type mechanism. This process is highly efficient, operating effectively at temperatures between 50°C and 120°C, and allows for the use of a wide range of solvents including alcohols, ketones, and chlorinated hydrocarbons. The robustness of this substitution step is critical for commercial scale-up of complex pharmaceutical intermediates, as it tolerates variations in reagent quality and maintains high conversion rates even at lower catalyst loadings. The final hydrolysis step is equally controlled, performed at low temperatures (0-5°C) to prevent degradation of the sensitive steroid skeleton while ensuring complete cleavage of the acetate ester to reveal the primary alcohol functionality essential for biological activity.
How to Synthesize Methylprednisolone Efficiently
Implementing this synthetic route requires careful attention to reaction parameters, particularly the stoichiometry of the brominating agent and the selection of the ammonium salt catalyst to maximize the yield of the 21-bromo intermediate. The process begins by dissolving the starting ketone in a solvent such as tetrahydrofuran or ether, followed by the addition of N-bromosuccinimide and a catalytic amount of ammonium acetate, stirring at ambient or slightly elevated temperatures until conversion is complete. The resulting intermediate can be isolated or used directly in the next step, where it is reacted with potassium acetate or sodium acetate in the presence of a phase-transfer catalyst like tetrabutylammonium bromide. Detailed standardized synthesis steps and specific workup procedures for each stage are provided in the guide below to ensure reproducibility and safety during pilot and production runs.
- React 6-alpha-methyl-11-beta,17-alpha-dihydroxypregn-1,4-diene-3,20-dione with a brominating agent like N-bromosuccinimide in the presence of an ammonium salt catalyst to form the 21-bromo intermediate.
- Perform a nucleophilic substitution on the 21-bromo intermediate using an acetate salt and a phase-transfer catalyst in a suitable solvent to generate the 21-acetate derivative.
- Hydrolyze the 21-acetate derivative under mild alkaline conditions at low temperatures (0-5°C) to yield the final methylprednisolone product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this bromination-based synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the substantial cost savings derived from replacing high-cost iodine reagents with inexpensive brominating agents, which directly impacts the bill of materials for the final API. Additionally, the stability of the 21-bromo intermediate allows for flexible production scheduling and inventory management, as the intermediate can be stored for extended periods without significant degradation, decoupling the upstream and downstream manufacturing stages. This flexibility enhances supply chain reliability by reducing the risk of batch failures and enabling just-in-time production models that align with fluctuating market demand for corticosteroid medications.
- Cost Reduction in Manufacturing: The elimination of expensive iodine reagents and the reduction in the number of synthetic steps significantly lower the overall production costs. By avoiding complex protection and deprotection sequences and utilizing cheap, commercially available catalysts like ammonium salts and phase-transfer agents, manufacturers can achieve a leaner cost structure. The high atom economy of the bromination reaction further contributes to waste minimization, reducing the costs associated with raw material procurement and waste disposal, ultimately leading to a more competitive pricing strategy for the final methylprednisolone product in the global marketplace.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction conditions ensures a consistent supply of high-quality material. Unlike biological oxidation methods which are susceptible to fermentation variability and contamination, this purely chemical route offers predictable kinetics and reproducible outcomes. The ability to source reagents like N-bromosuccinimide and ammonium acetate from multiple global suppliers mitigates the risk of single-source dependency, ensuring continuity of supply even during market disruptions. This reliability is crucial for maintaining uninterrupted production of life-saving anti-inflammatory medications.
- Scalability and Environmental Compliance: The mild reaction conditions and the ability to recover and recycle solvents make this process highly scalable from kilogram to multi-tonne production levels. The reduction in toxic by-products and the avoidance of heavy metal catalysts simplify the wastewater treatment process, aligning with increasingly stringent environmental regulations. This eco-friendly profile not only reduces the environmental footprint of the manufacturing facility but also minimizes the regulatory burden and potential fines associated with hazardous waste discharge, making it a sustainable choice for long-term industrial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology, drawing directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this route against existing supply chains.
Q: Why is the ammonium salt catalyst critical in the bromination step?
A: According to patent CN101418029A, the reaction between the starting ketone and the brominating reagent does not proceed effectively without the ammonium salt catalyst. Its addition significantly improves reaction preference and yield, ensuring the formation of a stable 21-bromo intermediate.
Q: What are the economic advantages of using bromine over iodine in this synthesis?
A: Traditional methods often rely on expensive iodine reagents. This novel route utilizes low-cost brominating agents like N-bromosuccinimide, which offers superior atom economy and significantly lowers raw material costs while reducing the toxicity and environmental impact associated with iodine-based processes.
Q: What purity levels can be achieved with this synthetic method?
A: The patent data indicates that through simple purification steps such as recrystallization, the final methylprednisolone product can achieve an HPLC purity content greater than 99%, meeting stringent requirements for pharmaceutical active ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylprednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering methylprednisolone and its key intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative bromination route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your position as a leader in the corticosteroid market.
