Advanced Synthesis of Treprostinil Intermediates via Weinreb Amide Coupling for Commercial Scale-up
Introduction to Next-Generation Treprostinil Manufacturing
The global demand for effective treatments for Pulmonary Arterial Hypertension (PAH) continues to drive innovation in the synthesis of prostacyclin analogs, specifically Treprostinil. As detailed in patent CN111116419B, a significant technological breakthrough has been achieved in the preparation of key Treprostinil intermediates, addressing long-standing challenges in yield, purity, and environmental safety. This novel methodology leverages a Weinreb amide coupling strategy that fundamentally alters the economic and operational landscape of producing this critical cardiovascular API intermediate. By shifting away from hazardous heavy metal oxidants and extreme cryogenic conditions, this process offers a robust pathway for reliable API intermediate suppliers aiming to secure supply chains for rare disease medications. The technical implications of this patent extend beyond mere chemical curiosity; they represent a viable solution for cost reduction in pharmaceutical manufacturing where regulatory compliance and process safety are paramount concerns for multinational stakeholders.
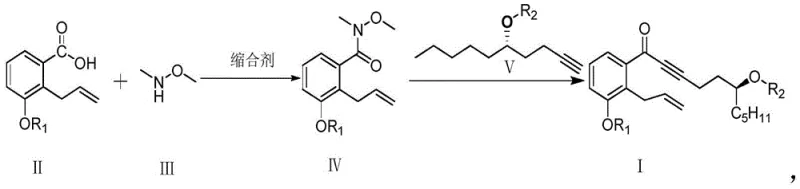
The core innovation lies in the strategic construction of the carbon skeleton through a highly selective nucleophilic addition to a Weinreb amide. Unlike traditional ester-based approaches that often suffer from over-addition or require harsh workup conditions, the Weinreb amide intermediate provides a stable chelating environment that ensures the reaction stops cleanly at the ketone stage. This precision is critical for maintaining the stereochemical integrity of the side chain, which is derived from chiral epoxyheptane precursors. For R&D directors evaluating technology transfer opportunities, this route presents a compelling case due to its demonstrated ability to produce solid intermediates that are easily purified, contrasting sharply with the difficult-to-handle oily substances characteristic of legacy synthetic routes. The integration of these chemical advancements positions manufacturers to meet the stringent quality specifications required for parenteral formulations used in PAH therapy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Treprostinil intermediates has been plagued by significant operational hurdles that impact both cost and environmental compliance. As illustrated in earlier literature such as WO9921830A1 and optimized in J.Org.Chem.2004, the conventional pathway relies heavily on the condensation of an aldehyde with a lithiated alkyne side chain. A major bottleneck in this traditional approach is the oxidation step utilizing Pyridinium Chlorochromate (PCC), a chromium(VI) reagent that poses severe environmental hazards and requires complex waste stream management to remove trace heavy metals from the final product. Furthermore, the generation of the nucleophilic alkyne species typically necessitates the use of n-butyllithium at cryogenic temperatures ranging from -40°C to -50°C. Maintaining such low temperatures on an industrial scale imposes a massive energy burden and introduces significant safety risks associated with pyrophoric reagents.
Another critical deficiency in the prior art is the physical state of the intermediates produced during the sequence. The legacy process generates predominantly oily substances that resist crystallization, forcing manufacturers to rely on repeated column chromatography for purification. This reliance on chromatographic separation is not only solvent-intensive, driving up raw material costs and waste disposal fees, but it also severely limits throughput and scalability. The accumulation of high-molecular-weight by-products during the Claisen rearrangement step further complicates purification, often requiring extensive downstream processing to meet the purity standards necessary for pharmaceutical applications. These cumulative inefficiencies create a fragile supply chain vulnerable to disruptions and cost volatility, making the conventional method suboptimal for meeting the growing global demand for PAH therapeutics.
The Novel Approach
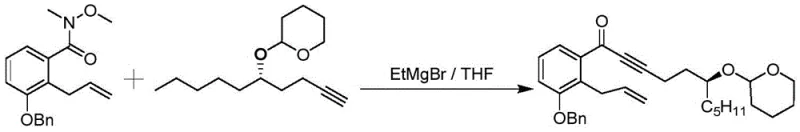
In stark contrast to these legacy challenges, the novel process disclosed in CN111116419B introduces a paradigm shift by utilizing a Weinreb amide intermediate to facilitate the key carbon-carbon bond formation. This approach completely circumvents the need for PCC oxidation, thereby eliminating the risk of heavy metal contamination and simplifying the environmental compliance profile of the manufacturing facility. Instead of relying on cryogenic lithiation, the new method employs Grignard reagents, such as ethylmagnesium bromide, which react efficiently at mild temperatures between room temperature and 45°C. This dramatic reduction in thermal severity translates directly into lower energy consumption and enhanced operational safety, allowing for the use of standard reactor equipment without the need for specialized low-temperature cooling infrastructure.
Moreover, the new synthetic route is engineered to produce solid intermediates rather than oils, a feature that revolutionizes the purification workflow. For instance, the benzyloxy-protected benzoic acid precursor can be recrystallized to achieve purities exceeding 99.0%, effectively removing impurities without the need for chromatography. The final coupling step to form the ketone intermediate proceeds with high selectivity, yielding products with HPLC purity greater than 95% directly after workup. This transition from oil-based to solid-state chemistry significantly reduces solvent consumption and processing time, offering substantial cost reduction in pharmaceutical manufacturing. By addressing the root causes of inefficiency in the traditional route, this novel approach provides a scalable, environmentally friendly, and economically superior alternative for the production of high-purity Treprostinil intermediates.
Mechanistic Insights into Weinreb Amide Coupling
The mechanistic elegance of this synthesis centers on the unique reactivity of the Weinreb amide functional group during nucleophilic attack. When the Weinreb amide (Compound IV) reacts with the alkyne anion generated in situ, the methoxy-N-methylamide moiety forms a stable five-membered chelate ring with the magnesium cation of the Grignard reagent. This chelation effect stabilizes the tetrahedral intermediate, preventing the collapse that would typically lead to a ketone capable of undergoing a second nucleophilic addition to form a tertiary alcohol. Consequently, the reaction halts cleanly at the ketone stage (Compound I) even in the presence of excess organometallic reagent. This intrinsic control over chemoselectivity is vital for maintaining the structural fidelity of the complex Treprostinil scaffold, ensuring that the sensitive allyl and alkyne functionalities remain intact throughout the transformation.
Furthermore, the preparation of the chiral alkyne side chain demonstrates exceptional stereocontrol through the regioselective ring-opening of epoxides. By utilizing (S)-1,2-epoxyheptane as a starting material, the process leverages copper-catalyzed Grignard addition or acetylide opening to establish the requisite stereocenter with high fidelity. The subsequent protection of the hydroxyl group as a tetrahydropyranyl (THP) ether ensures stability during the coupling reaction. The robustness of this mechanistic pathway is evidenced by the consistent GC purity of the side chain precursors, which routinely exceeds 90% and can reach up to 96.6% after purification. This high level of stereochemical purity is essential for the biological activity of the final drug substance, as the wrong enantiomer could be inactive or potentially toxic. The ability to achieve such purity without resorting to chiral resolution steps represents a significant advantage in terms of atom economy and process efficiency.
Impurity control is inherently built into the design of this synthetic route. The use of solid intermediates allows for the physical exclusion of soluble impurities through recrystallization, a mechanism that is far more efficient than chromatographic separation. For example, the hydrolysis of the Claisen rearrangement by-products is facilitated by the solubility differences between the desired carboxylic acid and the polymeric oils formed during heating. Additionally, the mild conditions of the Weinreb coupling minimize thermal degradation and side reactions that often plague high-temperature processes. The result is a cleaner reaction profile with fewer unknown impurities, simplifying the analytical burden on quality control laboratories. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the critical quality attributes of the intermediate are controlled by fundamental chemical principles rather than empirical optimization.
How to Synthesize Treprostinil Intermediate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity while maintaining safety. The process begins with the activation of the benzoic acid derivative using standard peptide coupling reagents such as EDCI and HOBT in the presence of N,O-dimethylhydroxylamine. This step must be monitored carefully to ensure complete conversion to the Weinreb amide, as residual acid can interfere with the subsequent Grignard reaction. Following isolation, the amide is reacted with the lithiated or magnesiated alkyne side chain. It is crucial to maintain the reaction temperature within the specified range of 25°C to 45°C to balance reaction rate with selectivity. Quenching the reaction with saturated ammonium chloride solution effectively breaks the magnesium chelate and liberates the ketone product. Detailed standardized synthetic steps see the guide below.
- React 2-allyl-3-benzyloxy-benzoic acid with N,O-dimethylhydroxylamine hydrochloride using EDCI and HOBT coupling agents to form the Weinreb amide intermediate.
- Prepare the alkyne side chain by reacting a protected epoxyheptane derivative with a Grignard reagent or acetylide species under mild conditions.
- Couple the Weinreb amide with the alkyne anion generated from ethylmagnesium bromide and the terminal alkyne at temperatures between 25°C and 45°C to yield the ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend well beyond the laboratory bench. The elimination of cryogenic conditions removes the dependency on specialized low-temperature reactors and the high energy costs associated with maintaining -50°C environments. This shift allows for the utilization of standard glass-lined or stainless steel reactors available in most multipurpose chemical facilities, significantly lowering the barrier to entry for contract manufacturing organizations. Furthermore, the removal of chromium-based oxidants like PCC eradicates the need for expensive heavy metal scavenging resins and complex wastewater treatment protocols, leading to substantial cost savings in waste management and regulatory compliance. These operational efficiencies translate directly into a more competitive cost structure for the final API, providing a strategic advantage in price-sensitive markets.
- Cost Reduction in Manufacturing: The transition from oily to solid intermediates fundamentally changes the economics of purification. By enabling recrystallization instead of column chromatography, the process drastically reduces solvent consumption, which is often one of the largest cost drivers in pharmaceutical synthesis. The avoidance of expensive reagents like n-butyllithium and trimethylsilylpropyne in favor of more economical Grignard reagents further lowers the raw material bill. Additionally, the higher yields observed in key steps, such as the 97.6% yield in Weinreb amide formation and 77.9% in the coupling step, mean less raw material is wasted, enhancing overall process mass intensity. These factors combine to create a leaner, more cost-effective manufacturing model that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The use of commercially available and stable starting materials, such as (S)-1,2-epoxyheptane and allyl bromide, mitigates the risk of supply disruptions associated with exotic or custom-synthesized reagents. Moreover, the ability to store solid intermediates for extended periods without degradation provides greater flexibility in production scheduling, allowing manufacturers to build inventory buffers against demand fluctuations. This reliability is critical for ensuring the continuous supply of life-saving medications to patients with pulmonary arterial hypertension, where treatment interruptions can have severe clinical consequences.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and easier to scale from pilot plant to commercial production. The reduced environmental footprint aligns with the increasing regulatory pressure on pharmaceutical companies to adopt green chemistry principles. By minimizing hazardous waste generation and energy consumption, manufacturers can achieve better sustainability ratings and avoid potential fines or shutdowns related to environmental violations. This forward-thinking approach not only protects the company's reputation but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term viability and market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of technology transfer or sourcing strategies. The information covers aspects ranging from reaction conditions to purity profiles, ensuring a comprehensive overview of the process capabilities.
Q: How does the new Weinreb amide route improve upon traditional Treprostinil synthesis?
A: The new route eliminates the use of toxic PCC oxidants and avoids cryogenic conditions (-40°C to -50°C) required for n-butyllithium reactions, replacing them with mild Grignard reactions at room temperature to 45°C.
Q: What purity levels can be achieved with this intermediate synthesis process?
A: The process yields intermediates with HPLC purity exceeding 95%, with specific steps achieving up to 99.0% purity after recrystallization, significantly reducing the need for column chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the conversion of oily intermediates into solid crystalline compounds allows for easier purification via recrystallization rather than chromatography, making it highly scalable and cost-effective for industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Treprostinil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex pharmaceutical intermediates like those used in Treprostinil production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel patent can be fully realized in a practical industrial setting. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the exacting standards required for cardiovascular drug manufacturing. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis and sensitive coupling reactions with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our capabilities align with your project goals. Together, we can ensure the uninterrupted availability of this vital medication for patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
