Advanced Manufacturing of Treprostinil Intermediates via Weinreb Amide Coupling
Advanced Manufacturing of Treprostinil Intermediates via Weinreb Amide Coupling
The landscape of pulmonary arterial hypertension (PAH) treatment has evolved significantly with the advent of prostacyclin analogs, among which Treprostinil stands out for its stability and subcutaneous administration capabilities. As global demand for this critical cardiovascular medication rises, the efficiency of its supply chain becomes paramount. Patent CN111116419A introduces a transformative synthetic methodology for producing key Treprostinil intermediates, specifically addressing the bottlenecks of traditional manufacturing. This technical insight report analyzes the strategic advantages of this novel route, focusing on the transition from hazardous oxidation processes to a streamlined Weinreb amide coupling strategy. For R&D directors and procurement leaders, understanding this shift is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a sustainable cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical ketone intermediate for Treprostinil has been plagued by significant operational and environmental challenges. As illustrated in the prior art routes such as WO9921830A1 and optimizations published in J.Org.Chem.2004, the traditional pathway relies heavily on the oxidation of alcohol precursors using Pyridinium Chlorochromate (PCC). This reliance on chromium-based oxidants introduces severe environmental liabilities, necessitating expensive heavy metal removal protocols and generating hazardous waste streams that complicate regulatory compliance. Furthermore, the construction of the chiral side chain in these legacy methods often employs n-butyllithium at cryogenic temperatures ranging from -40°C to -50°C. Such extreme conditions demand specialized refrigeration infrastructure, drastically increasing energy consumption and limiting the feasibility of large-scale batch processing. Additionally, the intermediates generated in these older pathways are frequently oily substances that resist crystallization, forcing manufacturers to rely on repetitive column chromatography for purification, a technique that is notoriously solvent-intensive and difficult to scale for commercial production.
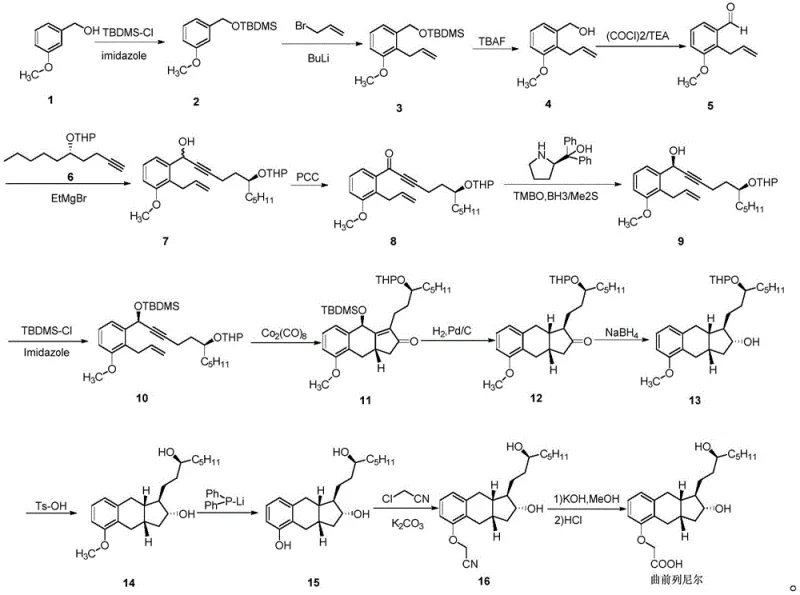
The Novel Approach
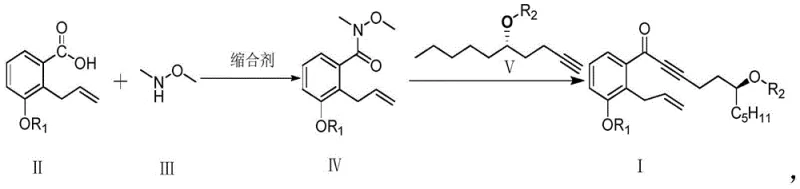
In stark contrast, the methodology disclosed in CN111116419A represents a paradigm shift towards green chemistry and process intensification. The core innovation lies in the utilization of a Weinreb amide intermediate (Formula IV) which reacts directly with a chiral alkyne anion (Formula V) to form the target ketone (Formula I). This approach completely bypasses the need for PCC oxidation, thereby eliminating the associated heavy metal contamination risks and simplifying the downstream purification workflow. Instead of cryogenic lithiation, the new process employs Grignard reagents, such as ethylmagnesium bromide, which facilitate the coupling reaction under mild thermal conditions between room temperature and 45°C. This moderation of reaction parameters not only reduces energy costs but also enhances operational safety by removing the hazards associated with handling pyrophoric reagents at ultra-low temperatures. Moreover, the resulting intermediates in this new pathway are solids that can be effectively purified through recrystallization, offering a robust solution for achieving high purity levels exceeding 98% without the scalability issues of chromatography.
Mechanistic Insights into Weinreb Amide Coupling
The success of this novel synthetic strategy hinges on the unique reactivity of the Weinreb amide functional group. Mechanistically, the reaction involves the nucleophilic attack of the alkyne anion, generated in situ from the protected side chain, onto the carbonyl carbon of the Weinreb amide. Unlike standard esters or acid chlorides which might undergo double addition to form tertiary alcohols, the Weinreb amide forms a stable tetrahedral intermediate chelated by the methoxy-methyl-amino group. This chelation prevents over-addition of the Grignard reagent, ensuring that the reaction stops cleanly at the ketone stage upon acidic workup. This selectivity is critical for maintaining the integrity of the complex molecular architecture required for Treprostinil activity. From an impurity control perspective, this mechanism significantly reduces the formation of over-alkylated by-products, which are common in direct ester additions. The ability to control the stoichiometry and reaction temperature precisely allows for the minimization of side reactions, resulting in a cleaner crude profile that facilitates easier downstream processing.
Furthermore, the preparation of the chiral side chain (Formula V) from (S)-1,2-epoxyheptane demonstrates excellent stereochemical control. The ring-opening of the epoxide by the acetylide species proceeds with high regioselectivity, preserving the chiral center essential for the biological activity of the final drug substance. The subsequent protection of the hydroxyl group with a tetrahydropyranyl (THP) moiety provides the necessary stability for the coupling step while remaining orthogonal to other functional groups present in the molecule. This strategic use of protecting groups ensures that the sensitive alkyne and aromatic moieties remain intact throughout the synthesis. The combination of these mechanistic advantages results in a process that is not only chemically elegant but also practically superior for industrial application, offering a reliable pathway to high-purity Treprostinil intermediates.
How to Synthesize Treprostinil Intermediate Efficiently
The implementation of this synthesis requires careful attention to reagent quality and reaction monitoring to maximize yield and purity. The process begins with the activation of the benzoic acid derivative to form the Weinreb amide, followed by the separate preparation of the chiral alkyne side chain. These two fragments are then united in the key coupling step. Detailed procedural specifics, including solvent choices, molar ratios, and workup procedures, are critical for reproducibility. For laboratory and pilot plant teams looking to adopt this technology, the following guide outlines the standardized operational framework derived from the patent examples.
- Prepare the Weinreb amide precursor (Formula IV) by reacting 2-allyl-3-protected-hydroxy-benzoic acid (Formula II) with N,O-dimethylhydroxylamine using a condensing agent like EDCI or HATU.
- Synthesize the chiral alkyne side chain (Formula V) from (S)-1,2-epoxyheptane via ring-opening with an acetylide species followed by hydroxyl protection.
- Couple Formula IV and Formula V using a Grignard reagent (e.g., Ethylmagnesium bromide) at mild temperatures (25°C to 45°C) to yield the ketone intermediate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents and the simplification of purification steps translate directly into a more resilient and cost-effective supply chain. By moving away from processes that require specialized waste treatment and extreme temperature control, manufacturers can reduce their operational overhead and mitigate the risk of production delays caused by equipment failure or regulatory scrutiny. This stability is crucial for maintaining the continuous supply of life-saving medications to the global market.
- Cost Reduction in Manufacturing: The removal of PCC oxidants and cryogenic equipment leads to substantial cost savings in both raw materials and utilities. Without the need for expensive heavy metal scavengers or energy-intensive cooling systems, the overall cost of goods sold is significantly reduced. Furthermore, the ability to purify intermediates via recrystallization rather than column chromatography drastically cuts solvent consumption and waste disposal fees, enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as (S)-1,2-epoxyheptane and common Grignard reagents ensures a stable supply of inputs. Unlike specialized lithiation reagents that may have long lead times or supply constraints, the reagents used in this process are commodity chemicals available from multiple global suppliers. This diversification of the supply base reduces the risk of shortages and allows for more flexible procurement strategies.
- Scalability and Environmental Compliance: The generation of solid intermediates that can be crystallized makes this process inherently scalable. Chromatography is a major bottleneck in scaling pharmaceutical synthesis, and its elimination allows for larger batch sizes and higher throughput. Additionally, the absence of chromium waste simplifies environmental compliance, reducing the regulatory burden and potential fines associated with hazardous waste management, thus ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new process is vital for stakeholders evaluating its adoption. The following questions address common concerns regarding safety, purity, and scalability, providing clarity on how this method compares to established industry standards. These insights are derived directly from the experimental data and claims presented in the patent documentation.
Q: How does this new process improve environmental compliance compared to traditional methods?
A: The novel process eliminates the use of Pyridinium Chlorochromate (PCC), a toxic heavy metal oxidant used in prior art, thereby removing the need for complex heavy metal waste treatment and reducing environmental hazards significantly.
Q: What are the temperature requirements for the key coupling reaction?
A: Unlike conventional methods requiring cryogenic conditions (-40°C to -50°C) with n-butyllithium, this invention utilizes Grignard reagents allowing the reaction to proceed under mild conditions ranging from room temperature to 45°C.
Q: How is product purity managed without column chromatography?
A: The process generates solid intermediates that can be purified via recrystallization rather than column chromatography, which is difficult to scale. This ensures high purity (>98%) suitable for industrial GMP production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Treprostinil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes in the pharmaceutical industry. Our team of expert chemists has extensively evaluated the methodology described in CN111116419A and possesses the technical capability to implement this advanced route at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Treprostinil intermediate meets the highest quality standards required for API synthesis.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available.
