Advanced Green Synthesis of Nervonic Acid for High-Purity Pharmaceutical Applications
Advanced Green Synthesis of Nervonic Acid for High-Purity Pharmaceutical Applications
The global demand for high-purity nervonic acid, a critical monounsaturated fatty acid essential for brain health and myelin sheath formation, has historically been constrained by reliance on limited natural sources such as shark oil or specific plant seeds. However, a groundbreaking technological shift is emerging through the innovations detailed in patent CN111423320A, which outlines a robust, scalable, and environmentally superior chemical synthesis pathway. This patent introduces a novel methodology that leverages erucic acid as a cost-effective starting material, utilizing a key Grignard reaction coupled with ethylene oxide ring-opening to efficiently construct the C24 carbon backbone. For R&D directors and procurement strategists in the pharmaceutical and nutraceutical sectors, this represents a pivotal opportunity to secure a reliable nervonic acid supplier capable of delivering consistent quality without the ecological baggage of traditional extraction methods. The process is designed not only for high yield but also for operational safety, replacing hazardous reagents with greener alternatives while maintaining the critical cis-configuration of the double bond.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of long-chain unsaturated fatty acids like nervonic acid has been plagued by significant technical and environmental hurdles that compromise both cost-efficiency and product integrity. Traditional routes often rely on phosphorus halides, such as phosphorus trichloride (PCl3) or phosphorus tribromide (PBr3), for the halogenation of alcohol intermediates. These reagents are notoriously aggressive, frequently inducing unwanted cis-trans isomerization of the delicate double bonds, which degrades the biological activity of the final product. Furthermore, the carbon chain extension steps in legacy processes typically utilize diethyl malonate, a method characterized by poor atom economy and the generation of substantial stoichiometric waste. Perhaps most critically, the final oxidation steps in conventional synthesis have depended heavily on chromium-based oxidants like pyridinium chlorochromate (PCC) or Jones reagent. These hexavalent chromium compounds are known carcinogens that create severe disposal challenges, requiring complex and expensive wastewater treatment protocols to meet modern environmental compliance standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111423320A presents a paradigm shift towards green chemistry principles without sacrificing synthetic efficiency. This innovative route substitutes hazardous phosphorus halides with lithium bromide monohydrate (LiBr·H2O) for the bromination step, a milder reagent system that effectively preserves the stereochemistry of the cis-double bond while eliminating phosphorus contamination. The carbon chain elongation is achieved through a highly atom-economical Grignard reaction with ethylene oxide, catalyzed by cuprous iodide, which drastically reduces waste compared to malonate condensation. Finally, the oxidation of the primary alcohol to the carboxylic acid is executed using a TEMPO (2,2,6,6-tetramethylpiperidin-1-yl)oxyl and iodobenzene diacetate system. This metal-free oxidation strategy operates under mild conditions, avoiding the generation of toxic heavy metal sludge and ensuring a cleaner impurity profile suitable for sensitive pharmaceutical applications.
Mechanistic Insights into Grignard-Mediated Chain Elongation
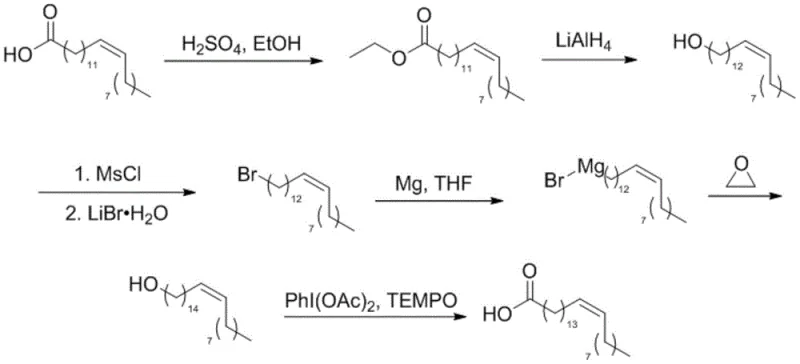
The core of this synthetic breakthrough lies in the precise execution of the Grignard reaction and the subsequent epoxide ring-opening, which serves as the critical carbon-carbon bond-forming event. The process begins with the formation of the Grignard reagent from 1-bromo-cis-13-docosene using activated magnesium turnings and a catalytic amount of iodine in anhydrous tetrahydrofuran (THF). The presence of cuprous iodide (CuI) during the subsequent addition of ethylene oxide is mechanistically vital; it acts as a catalyst to facilitate the nucleophilic attack of the organomagnesium species on the strained epoxide ring. This copper-catalyzed variation allows the reaction to proceed at lower temperatures (initially at -40°C), which is essential for preventing side reactions such as Wurtz coupling or proton abstraction that could lower the yield. The ring-opening of ethylene oxide extends the carbon chain by exactly two units, transforming the C22 precursor into the C24 backbone required for nervonic acid with high regioselectivity.
Furthermore, the choice of reagents in the preceding and succeeding steps is meticulously engineered to control the impurity profile and protect the olefinic functionality. The conversion of erucyl alcohol to the corresponding bromide utilizes methanesulfonyl chloride (MsCl) followed by displacement with lithium bromide. This two-step "activation-displacement" sequence avoids the strong acidic conditions of direct HBr substitution, which is a common cause of double bond migration or isomerization in long-chain alkenes. Similarly, the final oxidation utilizing the TEMPO/PhI(OAc)2 system proceeds via a radical mechanism that selectively targets the primary alcohol without affecting the internal cis-alkene. This chemoselectivity ensures that the final product retains the specific 15Z configuration necessary for its biological function in nerve tissue repair and development, resulting in a high-purity intermediate that meets stringent pharmacopeial standards.
How to Synthesize Nervonic Acid Efficiently
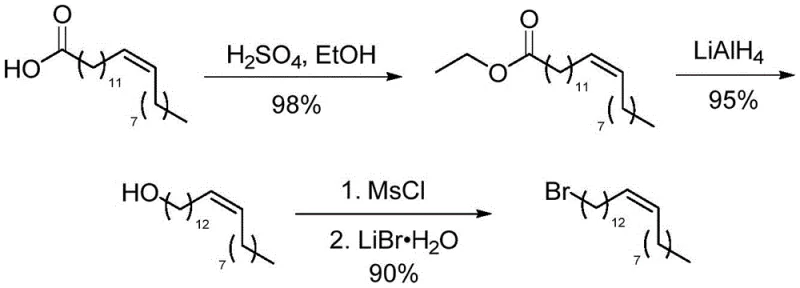
The synthesis of nervonic acid described in this patent offers a streamlined pathway that is amenable to kilogram-scale production, provided that strict anhydrous conditions and temperature controls are maintained during the Grignard formation. The process integrates standard unit operations such as esterification, reduction, and oxidation, making it compatible with existing multipurpose chemical manufacturing facilities. The following guide outlines the critical operational phases derived from the patent data, emphasizing the specific stoichiometric ratios and conditions required to maximize yield and purity. For detailed standard operating procedures (SOPs) regarding reactor setup, quenching protocols, and purification parameters, please refer to the technical documentation below.
- Convert erucic acid to ethyl erucate via acid-catalyzed esterification, followed by reduction to erucyl alcohol using LiAlH4.
- Transform erucyl alcohol into 1-bromo-cis-13-docosene using mesyl chloride and lithium bromide to preserve cis-geometry.
- Form the Grignard reagent and react with ethylene oxide in the presence of CuI to extend the carbon chain, followed by TEMPO oxidation to yield nervonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic benefits beyond mere technical feasibility. The shift away from extraction-dependent sourcing mitigates the risks associated with seasonal variability, geopolitical instability in raw material sourcing regions, and the ethical concerns surrounding shark-derived products. By establishing a fully synthetic supply chain, manufacturers can guarantee continuity of supply and stabilize pricing structures, insulating the business from the volatility of agricultural commodity markets. Moreover, the simplified purification workflows resulting from the use of cleaner reagents mean faster batch turnover times and reduced inventory holding costs for work-in-progress materials.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as chromium oxidants and phosphorus halides leads to a significant reduction in raw material procurement costs. More importantly, the removal of heavy metals from the process flow drastically lowers the operational expenditure associated with waste treatment and environmental compliance, as there is no need for specialized heavy metal scavenging resins or complex wastewater remediation systems. The high atom utilization of the ethylene oxide coupling step further ensures that a greater proportion of input mass is converted into saleable product, minimizing raw material waste and maximizing overall process efficiency.
- Enhanced Supply Chain Reliability: The starting material, erucic acid, is derived from widely available rapeseed or mustard seed oils, providing a robust and diversified supply base that is not subject to the same bottlenecks as exotic botanical extracts. The synthetic nature of the process allows for production to be scaled up or down rapidly in response to market demand without being constrained by harvest cycles. This flexibility ensures that downstream pharmaceutical customers can rely on consistent delivery schedules, reducing the need for excessive safety stock and enabling leaner inventory management strategies across the value chain.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the room temperature bromination and mild TEMPO oxidation, are inherently safer and easier to control on a large industrial scale compared to exothermic or high-pressure alternatives. The absence of toxic byproducts simplifies the regulatory approval process for the manufacturing site, facilitating faster audits and certifications. This environmental stewardship aligns with the increasing corporate sustainability goals of major multinational buyers, making the product more attractive for inclusion in green supply chains and eco-labeled final formulations.
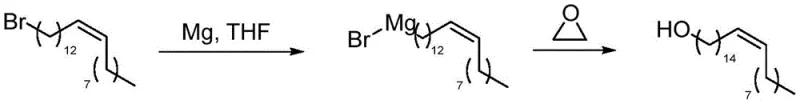
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of nervonic acid synthesized via this advanced Grignard-based protocol. These insights are derived directly from the experimental data and comparative analysis provided in the underlying patent literature, aiming to clarify the advantages of this method over traditional extraction or synthesis techniques. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this intermediate into their own drug substance or nutraceutical manufacturing processes.
Q: Why is the Grignard-Ethylene Oxide route preferred over traditional malonate synthesis for nervonic acid?
A: The Grignard-ethylene oxide route offers significantly higher atom utilization compared to the traditional diethyl malonate method, which generates substantial stoichiometric waste. Additionally, the mild conditions prevent isomerization of the sensitive cis-double bond.
Q: How does this synthesis method address environmental concerns regarding heavy metals?
A: This protocol replaces toxic chromium-based oxidants (like PCC or Jones reagent) with a TEMPO/PhI(OAc)2 system, eliminating carcinogenic heavy metal waste and simplifying downstream purification and wastewater treatment.
Q: What ensures the preservation of the cis-configuration at the C15 position?
A: The use of lithium bromide monohydrate for bromination avoids the harsh acidic conditions and radical mechanisms associated with phosphorus halides (PCl3/PBr3), thereby preventing thermal or acid-catalyzed cis-trans isomerization of the double bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nervonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN111423320A can be translated into a reliable industrial process. Our state-of-the-art facilities are equipped to handle moisture-sensitive Grignard reactions and low-temperature oxidations with precision, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of nervonic acid meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for neurological health products. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green synthesis route can improve your margins. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation studies, and let us help you secure a sustainable future for your nervonic acid supply.
