Scalable Nervonic Acid Production via Erucic Acid Redox Active Ester Technology
Scalable Nervonic Acid Production via Erucic Acid Redox Active Ester Technology
Introduction: A Paradigm Shift in Nervonic Acid Manufacturing
The global demand for nervonic acid (CAS 506-37-6), a critical monounsaturated fatty acid essential for brain health and myelin repair, has historically been constrained by inefficient production methods. Recent advancements detailed in patent CN115417759A introduce a revolutionary synthetic pathway that leverages erucic acid redox active esters to overcome these limitations. This technology represents a significant leap forward for the pharmaceutical intermediates industry, offering a robust alternative to resource-intensive plant extraction. By transitioning from biological extraction to a concise chemical synthesis, manufacturers can now access a more reliable nervonic acid supplier channel that is not subject to agricultural harvest cycles. The method utilizes a novel decarboxylative conjugate addition strategy, enabling the construction of the carbon skeleton with high precision and minimal waste. For R&D directors and procurement specialists, this patent signals a new era of cost reduction in pharmaceutical intermediates manufacturing, where complex fatty acids can be produced with unprecedented efficiency. The ability to synthesize high-purity nervonic acid in just three steps from cheap starting materials fundamentally alters the economic landscape of neurotherapeutic production. This report analyzes the technical merits and commercial implications of adopting this redox-active ester methodology for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of nervonic acid has been plagued by severe supply chain vulnerabilities inherent to natural extraction processes. Traditional methods rely heavily on the seed oils of specific plants such as Xanthoceras sorbifolium or Malania oleifera, which are geographically limited and subject to seasonal fluctuations. The extraction process itself is chemically inefficient, often requiring complex separation techniques to isolate the target fatty acid from a mixture of similar lipids, resulting in low overall yields and exorbitant costs. Furthermore, existing chemical synthesis routes described in prior art, such as those involving Grignard reagents or phosphorus trichloride, are operationally hazardous and environmentally burdensome. These conventional pathways typically involve lengthy multi-step sequences that accumulate impurities at each stage, necessitating rigorous and costly purification protocols to meet pharmaceutical standards. The reliance on moisture-sensitive reagents and strict inert atmosphere conditions further complicates the commercial scale-up of complex fatty acids, creating barriers for widespread industrial adoption. Consequently, the market has suffered from inconsistent availability and premium pricing, hindering the development of affordable neurological treatments.
The Novel Approach
In stark contrast, the methodology disclosed in CN115417759A streamlines the entire production workflow into a highly efficient three-step sequence that begins with readily available erucic acid. This innovative approach bypasses the need for hazardous halogenating agents and moisture-sensitive organometallics, instead utilizing stable redox active esters as key intermediates. The process is designed to be green and environmentally friendly, operating under mild conditions that do not require nitrogen or inert gas protection for the critical coupling step. This simplification of reaction conditions translates directly into substantial cost savings by reducing energy consumption and eliminating the need for specialized anhydrous infrastructure. The robustness of the redox active ester chemistry allows for high product yield and purity of more than 95 percent, ensuring that the final material meets the stringent requirements for API intermediates. By shortening the synthetic route and utilizing inexpensive feedstocks, this technology effectively addresses the core pain points of cost and scalability that have long hindered the nervonic acid market. It provides a viable pathway for reducing lead time for high-purity nervonic acid derivatives, ensuring a steady flow of materials for downstream drug formulation.
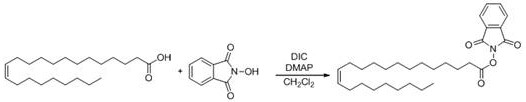
Mechanistic Insights into Erucic Acid Redox Active Ester Decarboxylation
The cornerstone of this synthetic breakthrough lies in the formation and subsequent reactivity of the erucic acid redox active ester, which serves as a potent radical precursor. In the first stage, erucic acid is activated through condensation with an N-hydroxy compound, such as N-hydroxyphthalimide (NHPI), using a carbodiimide coupling agent like DIC in the presence of a catalytic amount of DMAP. This transformation converts the relatively inert carboxylic acid into a highly electrophilic species capable of undergoing single-electron transfer (SET) processes under mild conditions. The resulting redox active ester is stable enough to be isolated or used in situ, providing flexibility in process design and inventory management for manufacturing teams. The choice of the N-hydroxy moiety is critical, as it dictates the reduction potential of the ester and influences the efficiency of the subsequent radical generation step. This activation strategy avoids the use of toxic thionyl chloride or oxalyl chloride, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. The structural integrity of the long-chain unsaturated fatty acid is preserved during this activation, preventing isomerization of the cis-double bond which is crucial for the biological activity of the final nervonic acid product.
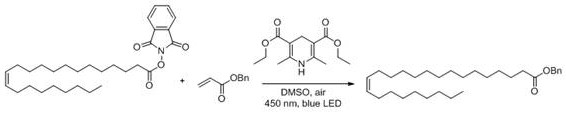
Following activation, the redox active ester undergoes a decarboxylative conjugate addition with an acrylate derivative, driven by visible light photocatalysis or metal catalysis. This step involves the generation of an alkyl radical via the fragmentation of the redox active ester, which then adds across the double bond of the acrylate acceptor to extend the carbon chain. The use of a Hantzsch ester as a terminal reductant facilitates the regeneration of the catalyst and ensures the turnover of the radical cycle without the need for external stoichiometric metals. Conducting this reaction under air conditions is a remarkable feat of chemical engineering, as it eliminates the operational complexity associated with oxygen-sensitive radical chemistry. The regioselectivity of the addition is controlled by the electronic properties of the acrylate and the stability of the intermediate radical species, ensuring the formation of the desired 1,5-dicarbonyl skeleton precursor. This mechanistic pathway allows for the precise installation of the requisite carbon atoms while maintaining the stereochemistry of the existing double bond in the erucic acid chain. The versatility of this system allows for variations in the acrylate component, enabling the synthesis of diverse nervonic acid derivatives for different therapeutic applications.
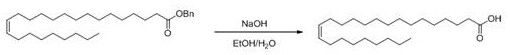
The final transformation involves the hydrolysis or oxidation of the nervonic acid derivative to reveal the free carboxylic acid functionality of the target molecule. In the hydrolysis pathway, the ester intermediate is treated with a base such as sodium hydroxide in a mixed solvent system of ethanol and water at elevated temperatures. This saponification reaction cleaves the protecting group efficiently, and subsequent acidification precipitates the crude nervonic acid for isolation. Alternatively, if an aldehyde intermediate is generated, a mild oxidation using sodium chlorite can be employed to convert the aldehyde to the carboxylic acid without affecting the sensitive alkene moiety. The purification process is straightforward, often requiring only extraction and crystallization to achieve purity levels exceeding 95 percent. This final step demonstrates the robustness of the overall synthetic design, as it tolerates the functional groups present in the long lipid chain without degradation. The ability to switch between hydrolysis and oxidation provides process chemists with options to optimize the workup based on the specific derivative synthesized in the previous step. This modularity enhances the adaptability of the process for different manufacturing scales and equipment configurations.
How to Synthesize Nervonic Acid Efficiently
To implement this cutting-edge technology in a production environment, operators must follow a standardized protocol that ensures safety and reproducibility across batches. The detailed standardized synthesis steps见下方的指南 outline the precise molar ratios, solvent choices, and reaction times required to maximize yield and minimize impurity formation. Adhering to these parameters is essential for maintaining the high purity specifications demanded by regulatory bodies for pharmaceutical ingredients.
- Synthesize erucic acid redox active ester by condensing erucic acid with N-hydroxy compounds using DIC and DMAP in dichloromethane.
- Perform decarboxylative conjugate addition with acrylate derivatives under light, electrochemical, or metal-catalyzed conditions to form the nervonic acid skeleton.
- Execute hydrolysis or oxidation of the nervonic acid derivative followed by acidification and purification to isolate the final nervonic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this redox active ester technology offers transformative benefits that extend beyond simple chemical efficiency. The shift from extraction to synthesis mitigates the risks associated with agricultural variability, ensuring a consistent and predictable supply of raw materials regardless of seasonal changes. This stability is crucial for long-term production planning and inventory management, allowing companies to secure their supply chains against external disruptions. The simplification of the synthetic route directly correlates to a reduction in manufacturing overhead, as fewer unit operations mean lower labor costs and reduced equipment occupancy time. Furthermore, the elimination of inert gas requirements lowers the barrier to entry for contract manufacturing organizations, increasing the pool of potential production partners. These factors combine to create a more resilient and cost-effective supply network for nervonic acid and its derivatives.
- Cost Reduction in Manufacturing: The streamlined three-step process significantly reduces the consumption of reagents and solvents compared to traditional multi-step syntheses. By avoiding expensive and hazardous reagents like Grignard reagents or phosphorus halides, the material costs are drastically lowered while improving workplace safety. The ability to run reactions under air conditions eliminates the capital expenditure associated with nitrogen generation and distribution systems. Additionally, the high yields reported in the patent minimize waste disposal costs and maximize the throughput of existing reactor volumes. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested into R&D initiatives.
- Enhanced Supply Chain Reliability: Sourcing erucic acid as a starting material provides a much more stable foundation than relying on niche plant extracts subject to geopolitical and climatic factors. Erucic acid is a commodity chemical available in large quantities, ensuring that production can be scaled up rapidly to meet surging market demand without raw material bottlenecks. The robustness of the chemistry allows for production in diverse geographic locations, reducing dependency on single-source suppliers and enhancing overall supply security. This reliability is paramount for pharmaceutical companies that require guaranteed continuity of supply to maintain their own drug manufacturing schedules. Consequently, partnering with a supplier utilizing this technology reduces the risk of stockouts and production delays.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent environmental regulations governing chemical manufacturing. The reduction in hazardous waste and the use of milder conditions simplify the permitting process for new production facilities. Scalability is inherently built into the design, as the reaction conditions are not dependent on specialized laboratory equipment like high-pressure photoreactors that are difficult to scale. The process can be adapted to standard stirred-tank reactors, facilitating a smooth transition from pilot plant to commercial scale production. This ease of scale-up ensures that the technology can meet the growing global demand for nervonic acid without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nervonic acid synthesis technology. These answers are derived directly from the patent specifications and practical considerations for industrial application. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their supply chain.
Q: How does the redox active ester method improve production costs compared to traditional extraction?
A: Traditional extraction relies on limited plant resources like Xanthoceras sorbifolium, leading to volatile pricing and supply bottlenecks. The redox active ester method utilizes abundant erucic acid as a starting material and reduces the synthesis to only 3 steps, significantly lowering raw material and operational expenses while ensuring consistent supply continuity.
Q: Does this synthesis route require inert gas protection for large-scale manufacturing?
A: No, one of the critical advantages of this patented technology is that the key decarboxylative conjugate addition step can be conducted under air conditions. This eliminates the need for expensive nitrogen or argon blanketing systems, simplifying reactor requirements and drastically reducing infrastructure costs for commercial scale-up.
Q: What purity levels can be achieved using this 3-step synthetic pathway?
A: The patent data indicates that the final nervonic acid product can achieve a purity of more than 95 percent. The use of specific redox active esters minimizes side reactions and byproduct formation, allowing for simpler purification processes and meeting stringent quality specifications required for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nervonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of nervonic acid in the development of next-generation neurological therapeutics and are committed to supporting your production needs with advanced synthetic capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of nervonic acid meets the highest industry standards. Our facility is equipped to handle the specific requirements of redox active ester chemistry, providing a safe and efficient environment for the manufacture of high-value pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative technology for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can enhance your supply chain efficiency and reduce your overall production costs.
