Revolutionizing Progesterone Manufacturing: A Strategic Shift to 4-Androstenedione Feedstocks
Revolutionizing Progesterone Manufacturing: A Strategic Shift to 4-Androstenedione Feedstocks
The pharmaceutical landscape for steroid hormone production is undergoing a significant transformation, driven by the urgent need to decouple synthesis from volatile agricultural supply chains. Patent CN103524588A introduces a groundbreaking methodology for the preparation of progesterone, a critical natural progestogen essential for maintaining pregnancy and a key precursor for corticosteroids and contraceptives. Historically, the industry has relied heavily on diosgenin extracted from wild yams, a dependency that has led to skyrocketing raw material costs and supply instability. This patent proposes a robust alternative by utilizing 4-androstenedione (4AD) as the primary starting material. By leveraging microbial fermentation technologies to produce 4AD, manufacturers can bypass the limitations of plant extraction, securing a more consistent and economically viable feedstock. This shift represents not merely a chemical optimization but a strategic supply chain realignment that promises to stabilize the global availability of high-purity hormonal intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial standard for synthesizing progesterone has depended on the Marker degradation of diosgenin, typically sourced from plants like Dioscorea zingiberensis. As illustrated in the traditional reaction pathway, this process begins with the extraction of diosgenin, followed by multiple chemical transformations to reach dienolone acetate, which is then converted into progesterone. However, this agricultural reliance creates a fragile supply chain vulnerable to weather patterns, land use changes, and increasing labor costs in cultivation regions. The patent explicitly notes that the price of dienolone acetate has shown a straight-line upward trend, directly inflating the production cost of the final API. Furthermore, the multi-step nature of extracting and purifying diosgenin introduces significant variability in raw material quality, complicating downstream processing and quality control. These structural inefficiencies make the conventional route increasingly unsustainable for large-scale commercial manufacturing in a cost-sensitive market.
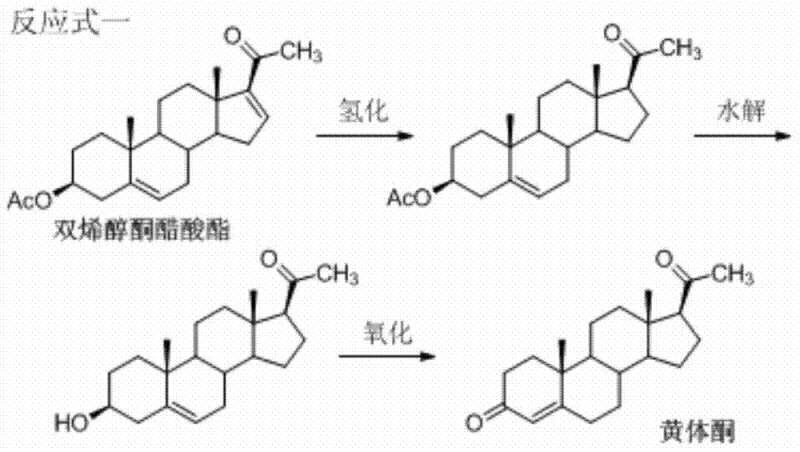
The Novel Approach
In stark contrast to the plant-dependent legacy methods, the novel approach detailed in CN103524588A establishes a streamlined synthetic route originating from 4-androstenedione (4AD). This strategy capitalizes on the emerging biotechnology sector's ability to produce 4AD via microbial degradation of phytosterols, a process that is scalable, reproducible, and largely immune to agricultural volatility. The new pathway involves a logical sequence of hydroxycyanation, dehydration, ketal protection, selective hydrogenation, and final Grignard addition. By starting with 4AD, the synthesis bypasses the early, cumbersome steps of sapogenin manipulation, effectively shortening the overall process timeline. The reaction conditions are designed to be mild and controllable, utilizing common reagents like acetone cyanohydrin and phosphorus oxychloride. This modernization of the synthetic backbone offers a compelling value proposition for procurement teams seeking to reduce exposure to raw material price shocks while maintaining rigorous quality standards for steroid intermediates.
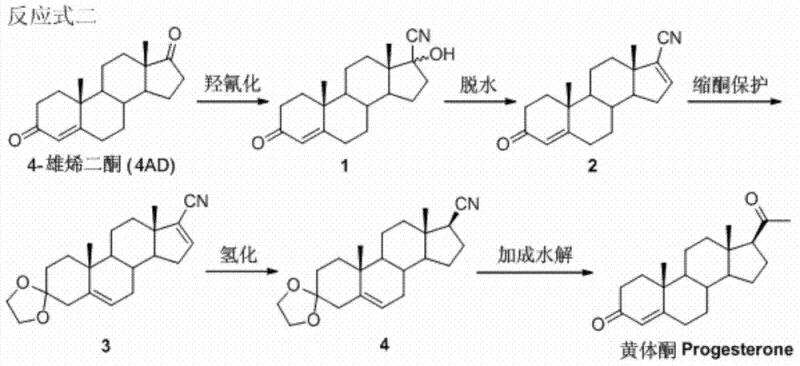
Mechanistic Insights into the 4AD-to-Progesterone Conversion
The chemical elegance of this process lies in its precise control over stereochemistry and functional group tolerance, particularly during the critical hydrogenation and addition steps. The synthesis initiates with the hydroxycyanation of the C17 ketone of 4AD using acetone cyanohydrin under alkaline conditions, yielding the 17-hydroxycyano intermediate with exceptional efficiency, reported at approximately 95%. Following dehydration with phosphorus oxychloride in pyridine, the molecule undergoes a strategic ketal protection at the C3 position. This protection is vital, as it shields the conjugated enone system during the subsequent catalytic hydrogenation. The hydrogenation step employs active nickel or palladium catalysts to selectively reduce the 16,17-double bond introduced during dehydration. Crucially, the reaction parameters—specifically the use of ethyl acetate as a solvent and room temperature conditions—are tuned to preserve the Δ4-3-keto structure, which is indispensable for the biological activity of the final progesterone molecule. This selectivity prevents the formation of inactive saturated byproducts, thereby simplifying purification.
Impurity control is further enhanced by the final conversion step, where the nitrile group is transformed into the acetyl side chain characteristic of progesterone. This is achieved through the addition of a methyl Grignard or methyllithium reagent, followed by acidic hydrolysis. The patent specifies that conducting this reaction at low temperatures (0°C) minimizes side reactions and ensures the formation of the desired 20-keto functionality. The use of weak acid solutions for the final hydrolysis allows for the simultaneous removal of the C3 ketal protecting group, delivering the final API in a single operational sequence. This telescoping of deprotection and ketone formation reduces the number of isolation steps, minimizing material loss and solvent consumption. For R&D directors, this mechanistic clarity offers a clear path to optimizing reaction parameters for even higher purity profiles, ensuring that the final product meets stringent pharmacopeial specifications for hormonal therapies.
How to Synthesize Progesterone Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and environmental controls, particularly during the moisture-sensitive Grignard addition and the exothermic hydroxycyanation phases. The patent outlines a robust protocol that balances yield with operational safety, making it highly suitable for transfer from laboratory to pilot plant scales. Operators must ensure strict temperature regulation during the dehydration step to prevent polymerization, and maintain an inert atmosphere during hydrogenation to guarantee catalyst activity. The following guide summarizes the critical operational milestones required to execute this pathway successfully, providing a framework for process engineers to establish standard operating procedures.
- Perform hydroxycyanation on 4-androstenedione using acetone cyanohydrin under alkaline conditions to form the 17-hydroxycyano intermediate.
- Execute dehydration of the hydroxyl group using phosphorus oxychloride and pyridine to generate the 16,17-double bond.
- Protect the C3 carbonyl group as a ketal using ethylene glycol and an acid catalyst.
- Conduct selective catalytic hydrogenation to reduce the 16,17-double bond while preserving the 4,5-double bond.
- React the nitrile group with a methyl Grignard or lithium reagent followed by acidic hydrolysis to yield progesterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to a 4AD-based synthesis route offers profound strategic benefits beyond simple chemical yield. The primary advantage is the decoupling of production costs from the volatile agricultural markets that dictate diosgenin prices. By sourcing 4AD from microbial fermentation, manufacturers gain access to a feedstock with a predictable cost structure and consistent quality, eliminating the seasonal bottlenecks associated with crop harvesting. This stability allows for more accurate long-term budgeting and contract negotiation with downstream pharmaceutical clients. Furthermore, the simplified synthetic sequence reduces the overall consumption of solvents and reagents per kilogram of finished product, contributing to a leaner manufacturing footprint. The elimination of complex plant extraction processes also reduces the environmental compliance burden, aligning production with increasingly strict green chemistry regulations.
- Cost Reduction in Manufacturing: The substitution of expensive dienolone acetate with cost-effective 4-androstenedione fundamentally alters the cost basis of progesterone production. Since 4AD is produced via fermentation, its pricing is driven by industrial biotechnology efficiencies rather than agricultural scarcity, leading to substantial potential savings in raw material expenditure. Additionally, the high yield of the initial hydroxycyanation step minimizes waste generation, further lowering the cost of goods sold. The process avoids the use of precious metal catalysts in favor of active nickel or standard palladium on carbon, which are more economically sustainable for large-scale operations. These factors combine to create a significantly more competitive cost structure without compromising product quality.
- Enhanced Supply Chain Reliability: Relying on microbial fermentation for the starting material ensures a continuous, year-round supply that is unaffected by droughts, pests, or geopolitical issues impacting crop exports. This reliability is critical for maintaining uninterrupted production schedules for essential hormonal medications. The synthetic route itself uses commodity chemicals like acetone cyanohydrin and ethylene glycol, which are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of the supply base enhances resilience against market disruptions, ensuring that delivery commitments to pharmaceutical partners can be met consistently.
- Scalability and Environmental Compliance: The reaction conditions described, such as atmospheric pressure hydrogenation and ambient temperature additions, are inherently safer and easier to scale than high-pressure or cryogenic alternatives. This facilitates rapid capacity expansion to meet surging market demand without requiring specialized, high-cost infrastructure. Moreover, the streamlined workflow reduces the total volume of organic waste generated per unit of product, simplifying wastewater treatment and disposal. The ability to operate with fewer isolation steps also lowers energy consumption for heating and cooling, supporting corporate sustainability goals and reducing the overall carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 4AD-based progesterone synthesis. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this technology for industrial production. Understanding these details is essential for stakeholders evaluating the transition from traditional plant-based routes to this modern fermentation-derived pathway.
Q: Why is 4-androstenedione preferred over traditional diosgenin-based raw materials?
A: Traditional routes rely on plant extraction (diosgenin), which is subject to agricultural fluctuations and rising labor costs. 4-androstenedione can be produced via microbial fermentation, offering a more stable, scalable, and potentially lower-cost supply chain independent of seasonal harvests.
Q: How does this process ensure selectivity during the hydrogenation step?
A: The process utilizes specific metal catalysts, such as active nickel or palladium on carbon, under controlled conditions (room temperature, specific solvent systems like ethyl acetate) to selectively reduce the 16,17-double bond without affecting the conjugated 4,5-double bond essential for biological activity.
Q: What are the yield expectations for the key intermediates in this pathway?
A: According to the patent data, the initial hydroxycyanation step achieves yields around 95%, while subsequent steps like dehydration and protection maintain robust yields between 65% and 87%, demonstrating a chemically efficient pathway suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Progesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 4-androstenedione synthesis route for the global steroid market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN103524588A are translated into reliable commercial reality. Our facilities are equipped with state-of-the-art hydrogenation reactors and cryogenic capabilities necessary for the Grignard steps, supported by rigorous QC labs that enforce stringent purity specifications for every batch. We are committed to helping our partners navigate the complexities of steroid synthesis, offering a seamless bridge between process development and full-scale manufacturing.
We invite pharmaceutical companies and chemical distributors to explore how this optimized route can enhance their supply chain resilience and cost efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can secure your supply of high-purity progesterone intermediates for the future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
