Optimized Chiral Synthesis of Pregabalin Intermediates for Commercial Scale Manufacturing
Introduction to Advanced Pregabalin Synthesis Technology
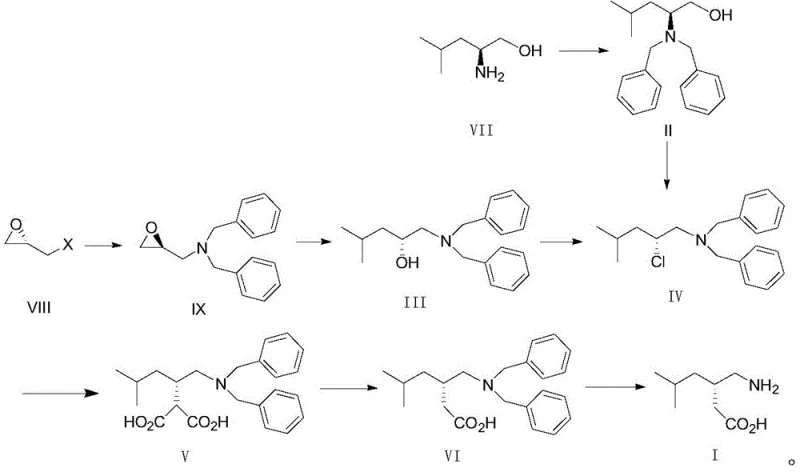
The pharmaceutical industry continuously seeks robust, scalable, and safe methodologies for the production of high-volume active pharmaceutical ingredients (APIs) such as pregabalin. Patent CN109942446B introduces a significant technological advancement in this domain by disclosing a novel preparation method for pregabalin (Compound I) utilizing R-1-(dibenzylamino)-4-methyl-2-chloropentane (Compound IV) as a pivotal intermediate. This approach represents a strategic shift from traditional chiral resolution or enzymatic methods towards a more direct chiral pool synthesis that leverages configuration retention. The core innovation lies in the streamlined reaction sequence which minimizes step count while maintaining high stereochemical integrity, addressing critical pain points in process chemistry regarding yield optimization and impurity control. By employing a dibenzylamine protecting group strategy, the process circumvents the use of hazardous deprotection reagents often associated with older synthetic routes.
Furthermore, the disclosed methodology emphasizes the use of mild reaction conditions and commercially accessible starting materials, which are essential factors for ensuring supply chain stability and cost-effectiveness in large-scale manufacturing. The transition from the chloro-intermediate to the final amino acid involves well-understood organic transformations such as nucleophilic substitution and catalytic hydrogenation, allowing for precise control over reaction parameters. This patent not only provides a specific chemical pathway but also offers a comprehensive framework for quality control, detailing specific solvent systems, molar ratios, and temperature ranges that are critical for reproducibility. For procurement and technical teams, understanding this specific pathway is vital as it offers a viable alternative to established but potentially problematic legacy processes, promising enhanced operational efficiency and reduced regulatory burden during the drug master file (DMF) registration phase.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pregabalin has relied heavily on routes involving phthalimide intermediates, as illustrated in prior art such as Scheme 1 found in patent IN201403517. In these conventional pathways, S-epichlorohydrin is reacted with a Grignard reagent followed by coupling with potassium phthalimide to introduce the nitrogen functionality. While chemically feasible, this approach suffers from significant drawbacks that impact both process safety and product quality. A major issue is the instability of the phthalimide side chain, which frequently leads to the formation of difficult-to-remove byproducts, specifically identified as Impurity C in related literature. The presence of such impurities necessitates rigorous and costly purification steps, thereby reducing overall process yield and increasing production costs. Moreover, the final deprotection step in phthalimide routes typically requires the use of hydrazine hydrate.
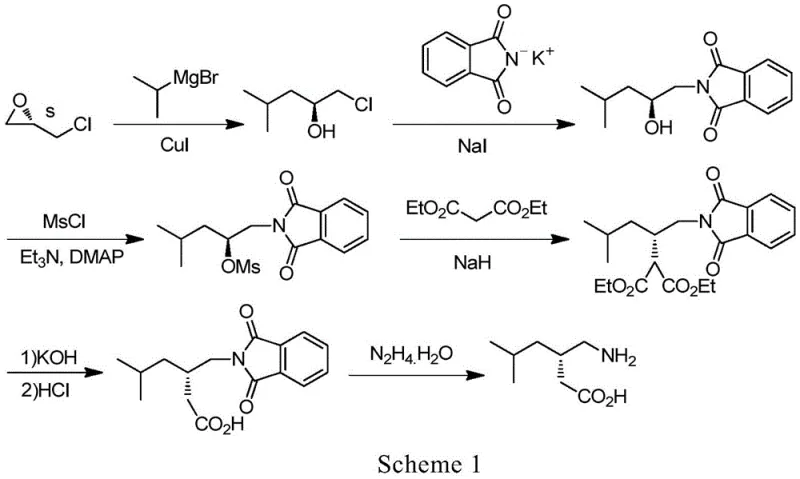
Hydrazine hydrate is a substance of very high concern due to its genotoxic and mutagenic properties, posing severe health risks to operators and creating complex waste disposal challenges for manufacturing facilities. The handling of hydrazine requires specialized equipment and strict safety protocols, which adds a layer of operational complexity and capital expenditure. Additionally, the reactivity of the phthalimide anion with diethyl malonate can be poor, leading to incomplete conversions and further complicating the impurity profile. These cumulative factors make the phthalimide route less attractive for modern, green chemistry-focused manufacturing environments where safety and environmental compliance are paramount. Consequently, there is a pressing industrial need for alternative synthetic strategies that eliminate genotoxic reagents while improving the robustness of the carbon-nitrogen bond formation.
The Novel Approach
The novel approach detailed in CN109942446B effectively addresses these limitations by substituting the phthalimide moiety with a dibenzylamine group. This strategic modification fundamentally changes the deprotection landscape, replacing the hazardous hydrazine treatment with a clean catalytic hydrogenation step. The use of dibenzylamine allows for the stable introduction of the amine functionality early in the synthesis, either via alkylation of S-leucinol or ring-opening of S-epichlorohydrin derivatives. This protecting group is robust enough to withstand the subsequent alkylation and decarboxylation conditions but can be easily removed under mild hydrogenation pressures (0.4 to 0.8 MPa) using palladium on carbon. This switch not only eliminates the genotoxic risk associated with hydrazine but also simplifies the downstream processing, as the byproducts of hydrogenolysis (toluene) are volatile and easily separated from the product. Furthermore, the new route demonstrates improved reactivity in the key alkylation step, reducing the formation of stubborn impurities and enhancing the overall purity profile of the intermediate compounds.
Mechanistic Insights into Nucleophilic Substitution and Decarboxylation
The core chemical transformation in this novel synthesis involves the nucleophilic substitution of the chloride in Compound IV by an active methylene compound, followed by hydrolysis and decarboxylation. Mechanistically, the reaction begins with the generation of a nucleophilic enolate from diethyl malonate using a strong base such as sodium hydride or metallic sodium in an aprotic polar solvent like N,N-dimethylformamide (DMF). The chloride leaving group in Compound IV is positioned at a chiral center, and the reaction proceeds with inversion or retention depending on the specific mechanistic pathway, though the patent indicates a process designed to maintain the desired stereochemistry for the (S)-enantiomer of pregabalin. The steric environment around the reacting center is managed by the bulky dibenzylamine group, which helps direct the incoming nucleophile and minimize side reactions. The reaction temperature is carefully controlled between 70°C and 110°C to provide sufficient activation energy for the substitution without promoting elimination side reactions that could lead to olefinic impurities.
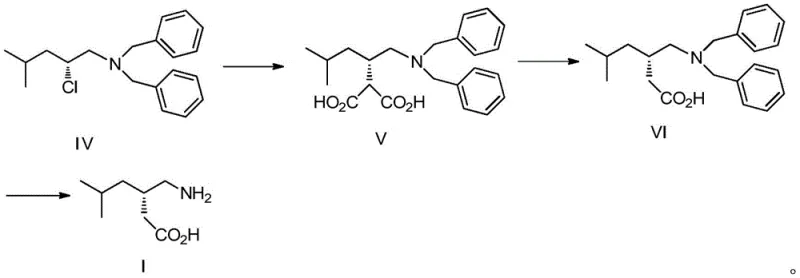
Following the alkylation, the resulting diester undergoes hydrolysis to form the dicarboxylic acid (Compound V), which is then subjected to thermal decarboxylation in an acidic medium. This step is critical for establishing the final carbon skeleton of the gabapentinoid structure. The decarboxylation mechanism involves the protonation of one carboxyl group, facilitating the loss of carbon dioxide through a cyclic transition state, ultimately yielding the mono-acid Compound VI. The choice of acid, preferably sulfuric acid in an aqueous acetic acid mixture, ensures efficient protonation while maintaining the solubility of the organic intermediate. Finally, the removal of the dibenzyl protecting groups via hydrogenolysis is a surface-catalyzed reaction where hydrogen atoms adsorbed on the palladium surface cleave the benzylic carbon-nitrogen bonds. This step is highly selective and does not affect the carboxylic acid moiety or the saturated alkyl chain, ensuring the structural integrity of the final pregabalin molecule. Understanding these mechanistic nuances is essential for R&D teams to optimize reaction kinetics and troubleshoot potential scale-up issues related to heat transfer or mass transfer limitations.
How to Synthesize Pregabalin Efficiently
The synthesis of pregabalin via this optimized route is structured around three primary operational stages that transform the chiral chloro-intermediate into the final active pharmaceutical ingredient. The process is designed to be modular, allowing for the isolation and characterization of key intermediates such as the malonic acid derivative and the protected amino acid, which facilitates rigorous quality control at each stage. The initial alkylation step requires precise control of stoichiometry, with a preferred molar ratio of Compound IV to diethyl malonate of 1:1.2, ensuring complete consumption of the valuable chiral chloride while minimizing excess reagent waste. Subsequent hydrolysis and decarboxylation are performed in a telescoped or sequential manner under reflux conditions, leveraging the thermal stability of the dibenzylamine group to drive the reaction to completion. The final hydrogenation step is conducted in alcoholic solvents like methanol or ethanol, which serve both as reaction media and crystallization anti-solvents, streamlining the isolation of the final product.
- React R-1-(dibenzylamino)-4-methyl-2-chloropentane (Compound IV) with diethyl malonate under basic conditions to form the malonic acid derivative (Compound V).
- Perform acidic decarboxylation of Compound V at reflux temperature to yield S-3-[(dibenzylamino)methyl]-5-methylhexanoic acid (Compound VI).
- Execute catalytic hydrogenation of Compound VI using Pd/C to remove benzyl protecting groups and obtain final Pregabalin (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial and supply chain perspective, the adoption of this synthetic route offers substantial strategic advantages over legacy methods, particularly concerning cost structure and regulatory compliance. The elimination of hydrazine hydrate from the process removes a significant safety liability and reduces the costs associated with hazardous material handling, storage, and waste treatment. This translates directly into lower operational expenditures and a reduced environmental footprint, aligning with the sustainability goals of modern pharmaceutical manufacturers. Additionally, the starting materials for this route, such as S-leucinol or S-epichlorohydrin, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and ensuring supply continuity even during market fluctuations. The robustness of the dibenzylamine protection strategy also implies higher yields and fewer batch failures, which enhances overall production capacity and reliability.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the simplification of the purification workflow and the avoidance of expensive or hazardous reagents. By replacing the phthalimide route, manufacturers eliminate the need for complex impurity profiling related to hydrazine residues and the associated analytical testing costs. The use of common solvents like DMF, ethanol, and methanol further reduces raw material expenses compared to specialized solvents required in other chiral synthesis methods. Moreover, the high selectivity of the catalytic hydrogenation step minimizes product loss during the final deprotection, maximizing the yield of the high-value API from the chiral precursor. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), providing a competitive edge in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available chiral building blocks and standard chemical reagents. Unlike proprietary enzymatic processes that may rely on specific biocatalysts with limited availability, this chemical synthesis route utilizes off-the-shelf reagents such as sodium hydride, palladium on carbon, and diethyl malonate. This universality ensures that procurement teams can source materials from a broad vendor base, reducing lead times and preventing production bottlenecks. The stability of the intermediates, particularly the dibenzyl-protected species, also allows for flexible inventory management, enabling manufacturers to stockpile key intermediates if necessary without significant degradation risks. This flexibility is crucial for meeting fluctuating market demands for pregabalin formulations globally.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, operating at atmospheric or moderate pressures and temperatures that are easily manageable in standard stainless steel reactors. The absence of cryogenic conditions or high-pressure hydrogenation (pressures are kept below 0.8 MPa) reduces the capital investment required for specialized equipment. From an environmental standpoint, the process generates less hazardous waste; the primary byproducts are benign salts and toluene, which can be recovered and recycled. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the burden of environmental compliance reporting. The streamlined nature of the synthesis also shortens the overall production cycle time, allowing for faster turnaround from raw material intake to finished API release.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pregabalin synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding the specific reaction parameters and safety profiles is essential for technical teams evaluating the feasibility of technology transfer. The answers below highlight the key differentiators of this method, focusing on safety, efficiency, and scalability.
Q: How does this method avoid genotoxic impurities compared to phthalimide routes?
A: Unlike traditional phthalimide routes that require hydrazine hydrate for deprotection—a known genotoxic reagent—this method utilizes dibenzylamine protection which is cleanly removed via catalytic hydrogenation, eliminating genotoxic risk profiles.
Q: What are the critical reaction conditions for the malonate alkylation step?
A: The alkylation of Compound IV requires an inert solvent like DMF, a strong base such as sodium hydride or metallic sodium, and temperatures ranging from 70°C to 110°C to ensure complete conversion to the malonic acid derivative.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions, readily available chiral starting materials like leucinol or epichlorohydrin, and avoids hazardous reagents, making it highly scalable and compliant with modern environmental safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregabalin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN109942446B to maintain competitiveness in the global pharmaceutical market. As a dedicated CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with state-of-the-art hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of pregabalin intermediate meets the highest international standards. We understand that consistency and quality are non-negotiable for our partners, and our technical team is prepared to support the full lifecycle of your product development.
We invite you to collaborate with us to leverage this superior synthesis technology for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your pregabalin supply chain with a solution that balances cost, quality, and safety, ensuring your continued success in the therapeutic landscape.
