Advanced Catalytic Aromatization For Commercial Scale Benzo-Fused Heterocycle Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients. Patent CN1214023C introduces a groundbreaking production method for benzo-fused heterocycles of general formula I, which addresses significant limitations found in prior art synthesis routes. This technology leverages a catalytic aromatization process using noble metal catalysts in the presence of hydrogen acceptors, followed by a seamless deacylation step. The innovation lies in the ability to convert tetrahydrobenzoannulated heterocycles of general formula II into valuable aromatic systems without the ecological and safety hazards associated with traditional elemental sulfur methods. For R&D directors and procurement managers alike, this patent represents a pivotal shift towards greener, more cost-effective manufacturing protocols that maintain high purity standards. The process is particularly relevant for the synthesis of PDE-V inhibitors and other high-value therapeutic intermediates where supply chain continuity is paramount. By adopting this catalytic approach, manufacturers can achieve substantial operational efficiencies while adhering to stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the aromatization of tetrahydrobenzo-fused compounds has relied heavily on reactions with elemental sulfur at elevated temperatures ranging from 200°C to 220°C. This conventional methodology presents severe drawbacks including high energy consumption due to the extreme thermal conditions required for the reaction to proceed efficiently. Furthermore, the process generates hydrogen sulfide gas, which possesses a noxious odor and poses significant safety risks to personnel and surrounding communities if not meticulously managed. Another critical issue is the solubility profile of elemental sulfur, which is only soluble in highly flammable solvents like carbon disulfide, thereby introducing substantial fire hazards during purification stages. The separation of by-products and unreacted sulfur often requires complex workup procedures that increase waste generation and reduce overall process yield. Additionally, the use of stoichiometric amounts of hydrogenation catalysts in alternative older methods leads to excessive costs due to the difficulty in recovering expensive noble metals from heterogeneous reaction mixtures. These factors collectively render traditional methods less viable for modern large-scale industrial applications where sustainability and cost control are decisive factors.
The Novel Approach
The novel approach described in the patent utilizes a catalytic amount of a noble metal catalyst in conjunction with organic hydrogen acceptors to drive the aromatization process under much milder and controlled conditions. This method eliminates the need for elemental sulfur entirely, thereby removing the associated risks of hydrogen sulfide release and flammable solvent usage. The reaction operates effectively within a temperature range of 140°C to 200°C for dehydrogenation, which significantly reduces energy expenditure compared to legacy techniques. A key feature of this innovation is the one-pot nature of the synthesis, where aromatization and subsequent deacylation occur consecutively without the isolation of intermediates. This streamlined workflow minimizes solvent consumption and reduces the time required for product isolation, leading to enhanced throughput capabilities. The use of hydrogen acceptors such as alpha-methylstyrene or oxygen gas mixtures facilitates the removal of hydrogen atoms from the substrate, driving the equilibrium towards the desired aromatic product. This catalytic transfer hydrogenation strategy ensures that the noble metal catalyst can be recovered easily via simple filtration, preserving its value for potential reuse and lowering material costs.
Mechanistic Insights into Pd-Catalyzed Aromatization and Deacylation
The core mechanism involves the activation of the tetrahydrobenzo-fused substrate by the noble metal catalyst, typically palladium on activated carbon, which facilitates the removal of hydrogen atoms from the saturated rings. The presence of a hydrogen acceptor is crucial as it scavenges the liberated hydrogen, preventing the reverse reaction and ensuring high conversion rates towards the aromatic system. This catalytic cycle allows for the use of only catalytic quantities of the expensive metal, contrasting sharply with older methods that required equimolar amounts. The reaction conditions are optimized to maintain the catalyst in its active state while preventing aggregation or deactivation over the course of the reaction. Solvent selection plays a vital role, with high-boiling inert solvents like xylene providing the necessary thermal stability and solubility for both reactants and products. The precise control of temperature and atmosphere ensures that side reactions are minimized, leading to a cleaner crude product profile that simplifies downstream purification efforts.
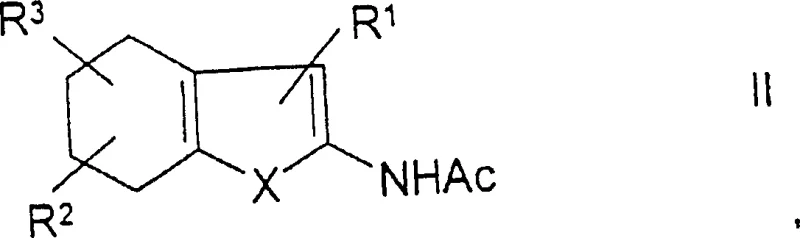
Following the aromatization step, the process incorporates an immediate deacylation of the acylated amino group at the 2-position of the heterocycle by the addition of amines such as pyrrolidine. This in-situ deacylation transforms the intermediate into a readily soluble product, which prevents precipitation issues that often complicate catalyst recovery in heterogeneous systems. The solubility of the final product allows for the noble metal catalyst to be isolated by simple hot filtration, ensuring minimal loss of precious metal and high purity of the filtrate. This mechanistic design effectively decouples the aromatization and deprotection steps into a single continuous operation, reducing the number of unit operations required. The choice of amine is critical, with primary or secondary amines boiling between 50°C and 200°C being preferred to ensure efficient reaction kinetics without decomposing the sensitive heterocyclic core. This integrated approach significantly enhances the overall yield, which generally ranges from 65% to 80% depending on the specific substrate and reaction parameters employed.
How to Synthesize 2-Aminobenzo[b]thiophene Efficiently
The synthesis of 2-aminobenzo[b]thiophene derivatives using this patented methodology offers a robust pathway for producing key pharmaceutical intermediates with high consistency. The process begins with the preparation of the tetrahydrobenzo-fused precursor, which is then subjected to the catalytic aromatization conditions described previously. Operators must ensure strict control over the addition rate of the hydrogen acceptor to manage exotherms and maintain reaction stability throughout the extended reaction time. The detailed standardized synthesis steps involve specific ratios of catalyst to substrate and precise temperature ramping profiles to maximize efficiency. For a comprehensive breakdown of the exact operational parameters and safety protocols required for implementation, please refer to the technical guide injected below.
- React tetrahydrobenzoannulated heterocycle with catalytic noble metal catalyst in the presence of a hydrogen acceptor.
- Maintain reaction temperature between 140°C and 200°C to ensure efficient dehydrogenation and aromatization.
- Add amine to deacylate the acylated amino group and isolate the final product via filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this catalytic technology offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex heterocyclic intermediates. The elimination of hazardous reagents like elemental sulfur and flammable solvents reduces the regulatory burden and insurance costs associated with chemical manufacturing facilities. This shift towards safer chemistry translates into more reliable supply chains as production is less susceptible to shutdowns caused by environmental compliance issues or safety incidents. The ability to recover and reuse noble metal catalysts significantly lowers the raw material cost base, providing a competitive edge in pricing negotiations for long-term contracts. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials and utilities, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. The scalability of this one-pot process ensures that production can be ramped up from laboratory scale to multi-ton commercial volumes without significant re-engineering of the process infrastructure.
- Cost Reduction in Manufacturing: The use of catalytic amounts of noble metals instead of stoichiometric quantities drastically reduces the consumption of expensive precious metal resources. By enabling easy filtration and recovery of the catalyst, the process minimizes waste and lowers the effective cost per kilogram of the final product. The removal of hazardous waste streams associated with sulfur-based methods further reduces disposal costs and environmental levies. These cumulative efficiencies result in substantial cost savings that can be passed down to customers or reinvested into process optimization initiatives.
- Enhanced Supply Chain Reliability: The robustness of the catalytic method ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The reduced reliance on hazardous materials minimizes the risk of logistical delays caused by strict transportation regulations for dangerous goods. Additionally, the simpler purification process shortens the production cycle time, allowing for faster turnaround on purchase orders and improved responsiveness to market demand fluctuations. This reliability makes the supplier a more strategic partner for global pharmaceutical companies seeking stability in their raw material sourcing.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing common solvents and equipment that are readily available in standard chemical manufacturing plants. The absence of toxic gas emissions aligns with increasingly stringent global environmental regulations, ensuring long-term operational viability without the need for costly abatement technologies. The reduced solvent usage and energy consumption contribute to a lower carbon footprint, supporting corporate sustainability goals. This environmental compatibility facilitates easier permitting and expansion of production capacity to meet growing market needs for high-purity intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic aromatization technology. These answers are derived directly from the patent specifications and practical experience with similar chemical transformations. Understanding these details helps stakeholders assess the feasibility and benefits of adopting this method for their specific production needs. The information provided here serves as a foundational guide for further technical discussions and feasibility studies.
Q: What are the advantages of using catalytic aromatization over elemental sulfur?
A: Catalytic aromatization eliminates the release of toxic hydrogen sulfide gas and avoids the use of flammable carbon disulfide, significantly improving safety and environmental compliance.
Q: Which noble metal catalysts are suitable for this synthesis?
A: Palladium on activated carbon, platinum on carbon, or rhodium on carbon are suitable, with 5% Pd/C being particularly preferred for cost and efficiency.
Q: How does this method impact supply chain reliability?
A: The one-pot process simplifies purification and reduces solvent usage, leading to faster turnaround times and more consistent batch-to-batch quality for procurement teams.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzo[b]thiophene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, leveraging advanced catalytic technologies like the one described in patent CN1214023C to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of benzo-fused heterocycle intermediates meets the exacting standards required for pharmaceutical synthesis. Our team of expert chemists is dedicated to optimizing these processes further to enhance yield and reduce environmental impact continuously. By choosing us as your partner, you gain access to a supply chain that is both resilient and compliant with international regulatory frameworks.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this catalytic route for your supply chain. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you engineer a more efficient and sustainable supply chain for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
