Advanced Catalytic Synthesis of Benzo-fused Heterocycles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical scaffolds, particularly those serving as key building blocks for potent therapeutic agents. Patent CN1422265A introduces a transformative production method for benzo-fused heterocycles of general formula I, which are indispensable intermediates in the synthesis of PDE-V inhibitors and other fine chemicals. This technology addresses long-standing challenges in heterocyclic chemistry by replacing hazardous stoichiometric reagents with a sophisticated catalytic system. The core innovation lies in the ability to aromatize tetrahydrobenzo-fused precursors using catalytic amounts of noble metals in the presence of hydrogen acceptors, followed by a seamless one-pot deacylation. This approach not only enhances the safety profile of the manufacturing process but also significantly streamlines the purification workflow, making it an attractive option for a reliable pharmaceutical intermediate supplier aiming to optimize their portfolio.
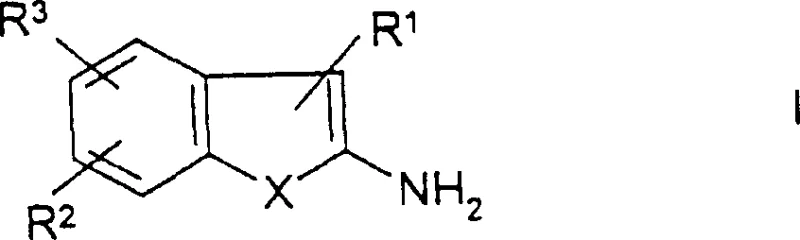
For R&D directors evaluating process viability, the structural versatility offered by this patent is paramount. The general formula encompasses variations where X can be sulfur, oxygen, or NH, allowing for the synthesis of thiophenes, furans, and pyrroles respectively. Specifically, the embodiment where X is sulfur yields 2-aminobenzo[b]thiophene derivatives, which are crucial precursors for blockbuster drugs. The ability to tune substituents R1 through R5 provides a flexible platform for generating diverse libraries of compounds, facilitating rapid structure-activity relationship studies without necessitating entirely new process development for each analog. This flexibility ensures that the technology remains relevant across multiple drug discovery programs, supporting the continuous demand for high-purity OLED material and specialty chemical precursors beyond just pharmaceuticals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the aromatization of tetrahydrobenzo-fused compounds has been plagued by severe operational and environmental drawbacks that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical intermediates. Traditional protocols often rely on reacting precursors with elemental sulfur at excessively high temperatures ranging from 200°C to 220°C. This harsh thermal requirement not only incurs substantial energy costs but also leads to the evolution of hydrogen sulfide, a toxic gas with a noxious odor that poses significant workplace safety risks and requires expensive scrubbing systems. Furthermore, elemental sulfur exhibits poor solubility in most benign solvents, frequently necessitating the use of carbon disulfide, which is highly flammable and presents acute fire hazards. Alternative methods utilizing stoichiometric amounts of hydrogenation catalysts like palladium on carbon suffer from economic inefficiency due to the high loading of precious metals required, alongside difficult separation processes where the product precipitates and traps the catalyst.
The Novel Approach
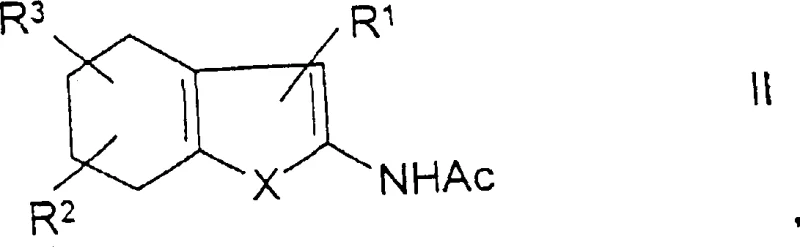
In stark contrast, the methodology disclosed in the patent utilizes a catalytic transfer hydrogenation strategy that fundamentally redefines the efficiency of benzo-fused heterocycle production. By employing a catalytic quantity of supported noble metals, such as 5% palladium on activated carbon, in conjunction with organic hydrogen acceptors like alpha-methylstyrene, the reaction proceeds under much milder conditions. This novel approach eliminates the need for elemental sulfur entirely, thereby eradicating the generation of hydrogen sulfide and removing the dependency on hazardous solvents. The process is designed as a one-pot sequence where aromatization is immediately followed by deacylation upon the addition of an amine, ensuring the final product remains soluble in the reaction medium. This solubility characteristic is a game-changer for cost reduction in electronic chemical manufacturing and pharma, as it allows for the simple filtration of the heterogeneous catalyst, preserving its activity for potential recycling and drastically simplifying downstream processing.
Mechanistic Insights into Pd-Catalyzed Transfer Hydrogenation
The mechanistic elegance of this synthesis lies in the synergistic interaction between the noble metal surface and the hydrogen acceptor molecule. During the reaction, the tetrahydrobenzo-fused substrate adsorbs onto the palladium active sites, where dehydrogenation occurs to form the aromatic system. Instead of releasing molecular hydrogen, which could pose explosion risks or require high-pressure equipment, the abstracted hydrogen atoms are immediately transferred to the hydrogen acceptor, such as alpha-methylstyrene, converting it to its saturated counterpart. This transfer hydrogenation mechanism drives the equilibrium towards the desired aromatic product without the need for external hydrogen gas sources or stoichiometric oxidants. The use of supported catalysts ensures that the metal remains heterogeneous, facilitating easy separation while maintaining high surface area for efficient catalysis. This controlled environment minimizes side reactions and ensures high selectivity, which is critical for maintaining the integrity of sensitive functional groups present on the heterocyclic ring.
Furthermore, the subsequent deacylation step is ingeniously integrated to solve solubility issues inherent in intermediate isolation. The acylated amino group on the newly formed aromatic ring is susceptible to nucleophilic attack by amines like pyrrolidine or piperidine at elevated temperatures. By adding the amine directly to the reaction mixture after aromatization is complete, the acyl group is cleaved in situ, yielding the free amine product. Crucially, while the acylated intermediate might have limited solubility leading to catalyst fouling, the final deacylated product exhibits excellent solubility in high-boiling solvents like xylene. This distinction allows the reaction mixture to remain homogeneous regarding the product, enabling the solid catalyst to be filtered off hot without product loss. This mechanism effectively decouples the catalyst recovery from product isolation, a significant advancement for reducing lead time for high-purity thiophenes in industrial settings.
How to Synthesize 2-Aminobenzo[b]thiophene Efficiently
Implementing this synthesis requires careful control of reaction parameters to maximize yield and purity, leveraging the specific conditions outlined in the patent data. The process begins with the suspension of the supported catalyst in an inert high-boiling solvent, followed by the addition of the tetrahydro precursor and the hydrogen acceptor. Temperature control is vital, with the aromatization phase typically conducted between 140°C and 200°C to ensure sufficient kinetic energy for dehydrogenation without degrading the sensitive heterocyclic core. Once the aromatization is deemed complete, usually monitored by the cessation of water separation or reaction time, the temperature is adjusted, and the amine is introduced to trigger deacylation. Detailed standardized synthesis steps see the guide below.
- React tetrahydrobenzo-fused heterocycle with catalytic noble metal (e.g., 5% Pd/C) and hydrogen acceptor (e.g., alpha-methylstyrene) at 140-200°C.
- Maintain reaction until aromatization is complete, ensuring the hydrogen acceptor drives the equilibrium forward effectively.
- Add amine (e.g., pyrrolidine) directly to the mixture for in-situ deacylation, then filter catalyst and isolate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this catalytic methodology offers profound advantages by restructuring the cost drivers associated with heterocycle manufacturing. The shift from stoichiometric reagents to catalytic systems dramatically reduces the consumption of expensive noble metals, directly impacting the bill of materials. Moreover, the elimination of elemental sulfur and carbon disulfide removes significant costs related to hazardous waste disposal, environmental compliance, and specialized safety infrastructure. The one-pot nature of the reaction reduces the number of unit operations, minimizing solvent usage and labor hours required for intermediate isolation and purification. These factors collectively contribute to substantial cost savings, making the final intermediate more price-competitive in the global market without compromising on quality or regulatory standards.
- Cost Reduction in Manufacturing: The transition to a catalytic process using supported palladium significantly lowers the raw material costs compared to traditional stoichiometric methods. By utilizing hydrogen acceptors like alpha-methylstyrene, which are inexpensive commodity chemicals, the process avoids the need for costly oxidants or high-pressure hydrogenation equipment. The ability to filter and potentially recycle the heterogeneous catalyst further enhances the economic viability, as the precious metal content is retained within the solid phase rather than being lost in the mother liquor. Additionally, the use of common solvents like xylene reduces procurement complexity and storage costs, streamlining the overall supply chain expenditure.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including supported palladium catalysts and organic hydrogen acceptors, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require stringent anhydrous environments, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream API manufacturers. The simplified workup procedure also reduces the likelihood of bottlenecks in the production line, ensuring a steady flow of high-quality intermediates to the market.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of toxic gas evolution and the use of standard reactor configurations. The elimination of hydrogen sulfide generation simplifies the environmental permitting process and reduces the burden on废气 treatment systems. The high boiling point solvents used allow for operation at atmospheric pressure, reducing the engineering complexity and capital expenditure required for pressure-rated vessels. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific advantages and operational details described in the patent documentation, providing clarity for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing frameworks.
Q: What are the safety advantages of this catalytic method over traditional sulfur aromatization?
A: Unlike conventional methods that generate toxic hydrogen sulfide gas and require flammable carbon disulfide solvents, this catalytic process operates in inert high-boiling solvents like xylene without hazardous gas evolution.
Q: How does the one-pot deacylation improve process efficiency?
A: By performing deacylation immediately after aromatization in the same vessel, the intermediate remains soluble, allowing for simple filtration of the noble metal catalyst and eliminating complex isolation steps.
Q: Is this method scalable for industrial production of PDE-V inhibitor intermediates?
A: Yes, the use of supported catalysts like Pd/C and common hydrogen acceptors like alpha-methylstyrene makes the process highly suitable for commercial scale-up with robust yield profiles between 65% and 80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzo[b]thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic routes like the one described in CN1422265A for securing a competitive edge in the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-aminobenzo[b]thiophene meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of catalytic heterocyclic synthesis, delivering products that empower our clients' drug development pipelines.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this catalytic route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain resilience and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
