Advanced Synthesis of Optically Active Homophenylalanine Derivatives for Pharmaceutical Applications
Advanced Synthesis of Optically Active Homophenylalanine Derivatives for Pharmaceutical Applications
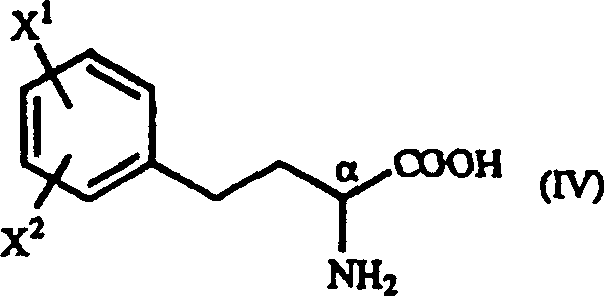
The pharmaceutical industry continuously seeks efficient pathways to produce high-value amino acid derivatives, particularly those serving as critical building blocks for Angiotensin-Converting Enzyme (ACE) inhibitors. Patent CN1321636A discloses a groundbreaking methodology for the preparation of optically active homophenylalanine derivatives, specifically targeting the general formula (IV) structures that are indispensable in modern cardiovascular therapeutics. This innovation represents a significant departure from traditional biocatalytic routes, offering a purely chemical synthesis that leverages Michael addition followed by catalytic reduction. The strategic advantage lies in the ability to generate these complex chiral molecules from inexpensive, commercially available starting materials without the need for costly enzymes or toxic cyanide reagents. By establishing a robust protocol for diastereoselective synthesis, this technology enables manufacturers to achieve high optical purity while maintaining economic viability. For R&D teams and procurement specialists alike, understanding this pathway is crucial for optimizing the supply chain of next-generation antihypertensive agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
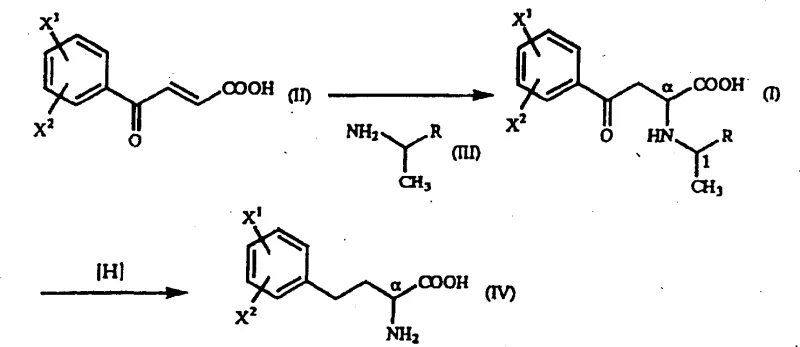
Historically, the production of optically active homophenylalanine derivatives has been plagued by significant technical and economic bottlenecks inherent to earlier methodologies. Traditional approaches often relied heavily on biological catalysts, such as hydantoinase or nitrilase, which necessitated the use of hazardous raw materials like virulent prussic acid compounds, posing severe safety and environmental risks. Furthermore, enzymatic transamination methods required expensive ketone acid substrates that drastically inflated the cost of goods sold, making them less attractive for large-scale commodity production. Another prevalent strategy involved racemic resolution techniques, which theoretically cap the maximum yield at 50% since half of the produced material is the unwanted enantiomer. These resolution processes also introduced complex operational steps, including multiple crystallization and separation stages, which increased processing time and reduced overall throughput. Consequently, the industry has long suffered from a lack of cost-effective, high-yield synthetic routes that can meet the rigorous purity standards of modern pharmaceutical manufacturing without compromising on safety or scalability.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined two-step chemical sequence that maximizes atom economy and minimizes waste generation. The core of this innovation involves the Michael addition reaction between beta-benzoyl acrylic acid derivatives and optically active 1-arylethylamine derivatives to form gamma-oxo-homophenylalanine intermediates. This reaction is remarkable for its ability to proceed under mild conditions using common alcoholic solvents, eliminating the need for exotic reagents or extreme temperatures. Following the formation of the intermediate, a subsequent catalytic reduction step efficiently converts the gamma-oxo group into the desired amine functionality while preserving the stereochemical integrity established in the first step. This methodology not only bypasses the 50% yield ceiling of racemic resolution but also avoids the supply chain vulnerabilities associated with biological enzymes. The result is a versatile platform technology capable of producing various substituted homophenylalanine derivatives with high efficiency.
Mechanistic Insights into Diastereoselective Michael Addition
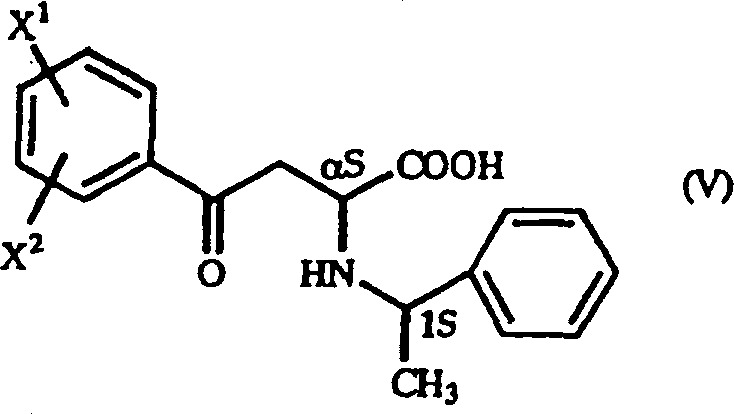
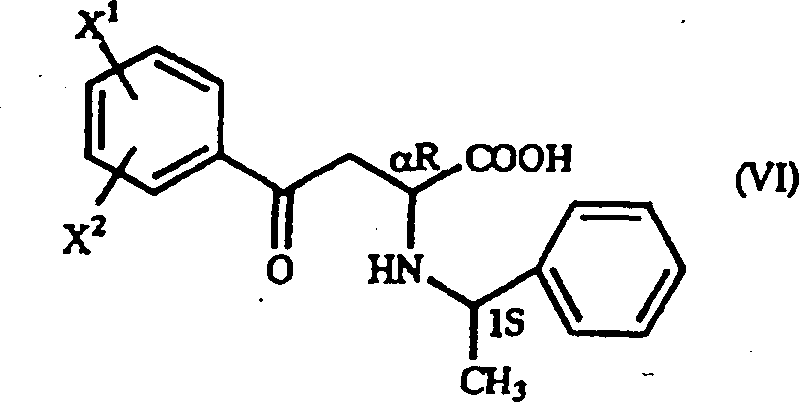
The success of this synthesis hinges on the precise control of stereochemistry during the initial Michael addition phase, where the interaction between the beta-benzoyl acrylic acid derivative and the chiral amine dictates the final optical purity. When an optically active 1-arylethylamine, such as (S)-1-phenylethylamine, reacts with the acrylic acid derivative, it generates a mixture of diastereomers, specifically the (alpha S, 1S) and (alpha R, 1S) configurations. The patent data reveals that the solubility differences between these diastereomers in specific solvent systems, particularly alcoholic solvents like ethanol, can be exploited to isolate the desired isomer with high selectivity. By carefully tuning reaction parameters such as temperature and concentration, manufacturers can drive the diastereomeric ratio to favor the target configuration, often achieving ratios exceeding 95:5. This level of control is critical because it ensures that the subsequent reduction step yields the final homophenylalanine derivative with the correct absolute configuration required for biological activity in ACE inhibitors.
Furthermore, the mechanistic pathway allows for flexibility in the choice of substituents on the aromatic ring, enabling the synthesis of a diverse library of derivatives including methoxy, chloro, methyl, and nitro variants. The reduction step typically employs a palladium catalyst, such as palladium-on-carbon, in the presence of hydrogen or formic acid, which effectively reduces the ketone moiety without racemizing the chiral center at the alpha position. This retention of configuration is paramount for maintaining the pharmacological efficacy of the final drug substance. The robustness of this catalytic system means that it can tolerate various functional groups, making it a universally applicable method for producing a wide range of homophenylalanine analogues. Understanding these mechanistic nuances allows process chemists to troubleshoot potential impurities and optimize the crystallization process for maximum recovery.
How to Synthesize Homophenylalanine Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to ensure optimal yield and purity, particularly during the isolation of the gamma-oxo intermediate. The process begins by dissolving the trans-beta-benzoyl acrylic acid derivative in a suitable solvent, with ethanol being the preferred choice due to its ability to facilitate high diastereoselectivity. The optically active amine is then added, and the mixture is maintained at a controlled temperature, typically between 30 and 60 degrees Celsius, to promote the formation of the precipitate. Detailed standard operating procedures regarding stoichiometry, agitation rates, and filtration techniques are essential for reproducibility. For a comprehensive breakdown of the specific experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Dissolve trans-beta-benzoyl acrylic acid derivatives in an alcoholic solvent such as ethanol and adjust the temperature to between 30 and 60 degrees Celsius.
- Add optically active 1-arylethylamine derivatives dropwise to the solution and maintain the reaction temperature to facilitate the formation of gamma-oxo-homophenylalanine intermediates.
- Filter the resulting precipitate to isolate the diastereomerically enriched intermediate, then perform catalytic hydrogenation using a palladium catalyst to obtain the final homophenylalanine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers transformative benefits for procurement managers and supply chain directors looking to optimize the cost structure of API manufacturing. The primary driver of cost reduction is the substitution of expensive, specialized raw materials with commodity chemicals that are readily available in the global market. By eliminating the dependency on biological catalysts and toxic cyanide precursors, the process significantly lowers both material costs and the expenses associated with hazardous waste disposal. Additionally, the high yield and selectivity of the reaction mean that less raw material is wasted, further enhancing the overall economic efficiency of the production line. This shift towards a more chemical-centric process also reduces the risk of supply disruptions often associated with biologically derived reagents, ensuring a more stable and predictable supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive enzymatic catalysts and the use of low-cost starting materials like beta-benzoyl acrylic acid derivatives lead to substantial savings in direct material costs. Furthermore, the simplified downstream processing, which relies on straightforward filtration and crystallization rather than complex chromatographic separations, reduces operational expenditures related to labor and equipment usage. The ability to recycle solvents like ethanol adds another layer of economic benefit, minimizing the total cost of ownership for the manufacturing process. These factors combined create a highly competitive cost profile that allows for better margin management in the final drug product.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the key reagents are bulk chemicals with multiple global suppliers, reducing the risk of single-source dependency. The robustness of the chemical synthesis means that production can be scaled up rapidly to meet surges in demand without the lead times associated with fermenting biological catalysts. This reliability is crucial for maintaining continuous manufacturing operations and meeting strict delivery schedules for downstream API producers. The stability of the intermediates also allows for safer storage and transportation, further de-risking the logistics network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram scale to multi-kilogram batches without loss of efficiency. From an environmental standpoint, the avoidance of heavy metals and toxic cyanides simplifies regulatory compliance and reduces the burden on wastewater treatment facilities. The use of green solvents like ethanol aligns with modern sustainability goals, making the process more attractive to environmentally conscious stakeholders. This combination of scalability and eco-friendliness positions the technology as a future-proof solution for long-term manufacturing needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on yield expectations, purity profiles, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production requirements. The information provided here serves as a foundational guide for further discussions with our technical sales team.
Q: What is the primary advantage of this synthesis method over enzymatic approaches?
A: Unlike enzymatic methods that often require expensive ketone acids or toxic cyanide compounds, this chemical synthesis utilizes readily available beta-benzoyl acrylic acid derivatives and 1-arylethylamines, significantly reducing raw material costs and simplifying the supply chain.
Q: How is optical purity controlled during the Michael addition step?
A: Optical purity is achieved through diastereoselective crystallization. By using optically active 1-arylethylamine and selecting appropriate solvents like ethanol, the reaction preferentially forms specific diastereomers (such as the alpha S, 1S configuration) which can be isolated via filtration with high selectivity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable as it avoids complex biological catalysts and uses standard chemical reactors. The use of common solvents like ethanol and robust metal catalysts like palladium-on-carbon ensures that the method can be easily transferred from laboratory scale to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homophenylalanine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific homophenylalanine analogues or large-scale supply of standard intermediates, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to validate the suitability of our materials for your process. Let us partner with you to drive innovation and efficiency in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
