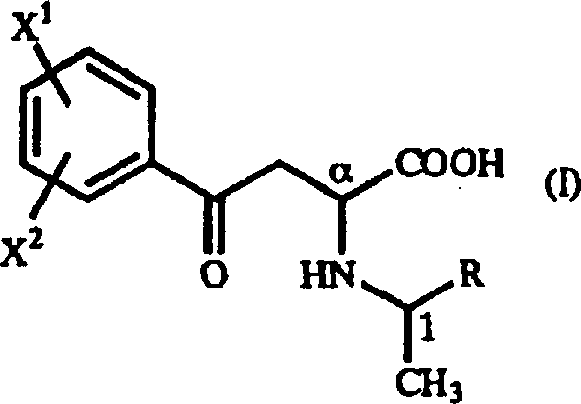
Advanced Synthesis of Homophenylalanine Derivatives for Commercial ACE Inhibitor Production
The pharmaceutical industry continuously seeks robust and cost-efficient pathways for producing critical amino acid intermediates, particularly those serving as key building blocks for Angiotensin-Converting Enzyme (ACE) inhibitors. Patent CN1217710A introduces a groundbreaking methodology for the preparation of optically active homophenylalanine derivatives, addressing long-standing challenges in yield, purity, and economic feasibility. This technical insight report analyzes the novel synthetic route disclosed in the patent, highlighting its potential to revolutionize the supply chain for high-purity pharmaceutical intermediates. By leveraging a strategic combination of Michael addition and catalytic reduction, this method bypasses the limitations of traditional biocatalytic and resolution-based approaches. For R&D directors and procurement managers, understanding the mechanistic advantages of this process is essential for optimizing manufacturing costs and ensuring a reliable supply of complex amino acid derivatives. The following analysis details how this technology aligns with modern demands for sustainable and scalable chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active homophenylalanine derivatives has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional methods often rely on biocatalysts such as enzymes, which, while specific, require expensive raw materials like ketoacids or toxic hydrocyanic acid compounds for precursor synthesis. Furthermore, enzymatic processes can be sensitive to reaction conditions, leading to inconsistent batch quality and difficulties in scale-up. Another prevalent approach involves the optical resolution of racemic mixtures, which theoretically limits the maximum yield to fifty percent of the starting material, inherently doubling the waste and cost associated with the discarded enantiomer. These conventional pathways also frequently involve complex operational procedures, such as multiple purification steps and the handling of hazardous reagents, which increase the environmental footprint and regulatory burden. For supply chain heads, these inefficiencies translate into volatile pricing and potential disruptions in the availability of critical ACE inhibitor intermediates. The reliance on scarce biological agents or the generation of substantial chemical waste makes these older methods unsustainable for modern, high-volume pharmaceutical production requirements.
The Novel Approach
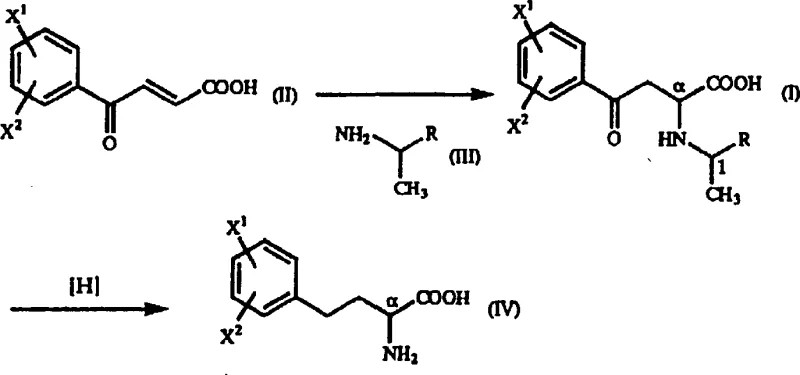
In stark contrast, the method disclosed in patent CN1217710A offers a streamlined, cost-effective solution that fundamentally alters the production landscape for these valuable intermediates. This novel approach utilizes a chemical synthesis strategy centered on the reaction of beta-benzoyl acrylic acid derivatives with optically active 1-arylethylamine derivatives. By employing a Michael addition reaction, the process constructs the carbon skeleton with high diastereoselectivity, effectively bypassing the need for expensive enzymes or toxic cyanide sources. The key innovation lies in the ability to isolate the desired intermediate as a precipitate directly from the reaction mixture, leveraging differences in solubility between diastereomers to achieve high optical purity without complex chromatographic separations. This chemical route uses readily available and inexpensive starting materials, such as 1-phenylethylamine, which significantly lowers the raw material cost basis. Moreover, the subsequent reduction step employs standard catalytic hydrogenation, a well-understood and scalable unit operation in the fine chemical industry. This shift from biological or resolution-based methods to a direct asymmetric synthesis provides a robust foundation for cost reduction in pharmaceutical intermediate manufacturing, ensuring a more stable and economical supply chain for downstream drug producers.
Mechanistic Insights into Michael Addition and Catalytic Reduction
The core of this innovative synthesis lies in the precise control of stereochemistry during the Michael addition phase, which sets the stage for the final optical purity of the homophenylalanine derivative. The reaction involves the nucleophilic attack of an optically active 1-arylethylamine on a beta-benzoyl acrylic acid derivative, forming a gamma-oxo-homophenylalanine intermediate. The chiral information from the amine induces diastereoselectivity, resulting in a mixture of diastereomers where one isomer is heavily favored. Crucially, the patent data indicates that by selecting appropriate protic solvents like ethanol or isopropanol, the desired diastereomer can be induced to precipitate out of the solution, allowing for physical separation from the minor isomer. This crystallization-induced diastereomeric enrichment is a powerful tool for R&D teams, as it achieves high optical purity (often exceeding 95% diastereomeric ratio) through simple filtration rather than energy-intensive distillation or chromatography. The structural integrity of the intermediate is maintained throughout this process, ensuring that the chiral center at the alpha position remains intact for the subsequent transformation.
Following the isolation of the high-purity gamma-oxo intermediate, the process proceeds to a catalytic reduction step to yield the final homophenylalanine derivative. This reduction is typically carried out in the presence of a metal catalyst, such as palladium-carbon, under a hydrogen atmosphere or using chemical reducing agents like formic acid. The reaction conditions are mild, often conducted in acidic alcohol solvents at moderate temperatures, which preserves the stereochemical configuration established in the previous step. The mechanism involves the hydrogenolysis of the benzyl-amine bond and the reduction of the ketone group to a methylene group, effectively removing the chiral auxiliary and revealing the free amino acid structure. This step is highly efficient and can be driven to near-quantitative conversion, minimizing the formation of by-products and simplifying the final workup. The ability to maintain asymmetry at the alpha position during this reductive cleavage is critical for producing the specific enantiomers required for ACE inhibitor activity. Understanding this mechanistic pathway allows technical teams to optimize reaction parameters for maximum yield and purity.
How to Synthesize Homophenylalanine Derivatives Efficiently
Implementing this synthesis route requires careful attention to solvent selection and reaction stoichiometry to maximize the diastereomeric ratio and overall yield. The process begins with the dissolution of the beta-benzoyl acrylic acid derivative in a suitable alcohol solvent, followed by the controlled addition of the optically active amine. Temperature control is vital, with ranges between 30°C and 60°C often providing the best balance between reaction rate and selectivity. Once the intermediate is formed and isolated, the reduction step necessitates the use of a robust catalyst system and acidic conditions to facilitate the cleavage of the chiral auxiliary. Detailed standard operating procedures for scaling this chemistry from laboratory to production scale are essential for ensuring consistency and safety. The following guide outlines the critical operational parameters derived from the patent data to assist process engineers in technology transfer.
- React beta-benzoyl acrylic acid derivatives with optically active 1-arylethylamine derivatives in a protic solvent to form gamma-oxo-homophenylalanine intermediates.
- Isolate the diastereomeric intermediate precipitate through filtration and recrystallization to ensure high optical purity.
- Perform catalytic hydrogenation using a palladium catalyst in an acidic alcohol solvent to reduce the ketone group and yield the final homophenylalanine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. The primary advantage is the substantial cost savings achieved through the elimination of expensive biocatalysts and the avoidance of yield-limiting resolution steps. By utilizing commodity chemicals as starting materials, the raw material cost profile is drastically improved, making the final intermediate more competitive in the global market. Furthermore, the simplified purification process, which relies on precipitation and filtration rather than complex chromatography, reduces solvent consumption and waste disposal costs, contributing to a more sustainable manufacturing footprint. This efficiency translates directly into improved margins and the ability to offer more stable pricing to downstream pharmaceutical partners. The robustness of the chemical steps also ensures a reliable supply of high-purity pharmaceutical intermediates, mitigating the risk of production delays associated with sensitive enzymatic processes.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the use of inexpensive, commercially available starting materials such as 1-phenylethylamine and beta-benzoyl acrylic acid derivatives. Unlike enzymatic routes that require costly ketoacids or specialized enzymes, this chemical pathway leverages standard organic synthesis reagents that are sourced easily at scale. Additionally, the high diastereoselectivity achieved through crystallization minimizes the loss of material associated with racemic resolution, effectively doubling the theoretical yield compared to traditional splitting methods. The reduction in processing steps and the ability to isolate intermediates via simple filtration further lower operational expenditures related to energy and labor. These factors combine to create a significantly leaner cost structure for the production of complex amino acid derivatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on robust chemical reactions that are less susceptible to the variability inherent in biological systems. The starting materials are stable and have long shelf lives, reducing the risk of raw material degradation and inventory loss. The process scalability ensures that production volumes can be ramped up quickly to meet surging demand for ACE inhibitor intermediates without the need for specialized bioreactor infrastructure. By reducing lead time for high-purity pharmaceutical intermediates, manufacturers can respond more agilely to market fluctuations and regulatory requirements. This reliability is crucial for maintaining uninterrupted drug production schedules and meeting the stringent delivery expectations of global pharmaceutical clients.
- Scalability and Environmental Compliance: The commercial scale-up of complex pharmaceutical intermediates is facilitated by the use of standard unit operations such as mixing, filtration, and hydrogenation, which are well-established in multi-purpose chemical plants. The process generates less chemical waste compared to resolution methods, as the majority of the starting material is converted into the desired product rather than discarded enantiomers. The use of common solvents like ethanol simplifies solvent recovery and recycling systems, aligning with green chemistry principles and environmental regulations. This environmental efficiency not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturer. The ability to scale from kilogram to multi-ton production with consistent quality makes this route ideal for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of homophenylalanine derivatives. These answers are derived from the specific technical disclosures and experimental data found in the patent literature, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains. The responses cover aspects of purity control, scalability, and comparative advantages over legacy methods.
Q: What are the primary advantages of this synthesis route over enzymatic methods?
A: This chemical synthesis route avoids the use of expensive biocatalysts and toxic hydrocyanic acid compounds required in enzymatic methods. It utilizes readily available starting materials and achieves high diastereoselectivity through simple crystallization, significantly reducing raw material costs and operational complexity.
Q: How is optical purity controlled during the manufacturing process?
A: Optical purity is controlled by using optically active 1-arylethylamine as a chiral auxiliary during the Michael addition step. The resulting diastereomers exhibit different solubilities, allowing the desired isomer to be isolated with high purity (often exceeding 95% diastereomeric ratio) through selective precipitation and recrystallization before the final reduction step.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It employs standard chemical reactions like Michael addition and catalytic hydrogenation which are well-established in industrial settings. The use of common solvents like ethanol and robust catalysts like palladium-carbon ensures that the process can be safely scaled from kilogram to multi-ton production without significant technical barriers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homophenylalanine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our technical team has extensively evaluated the synthetic route described in patent CN1217710A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of homophenylalanine derivatives meets the exacting standards required for pharmaceutical applications. We are committed to delivering consistent quality and reliability, serving as a strategic partner for your long-term drug development needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical synthesis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By leveraging our expertise in process chemistry and scale-up, we can help you secure a stable supply of high-purity pharmaceutical intermediates while maximizing operational efficiency. Let us engineer the solution for your production bottlenecks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
