Advanced Rh(III)-Catalyzed Synthesis of Benzo[a]carbazoles for Commercial Pharmaceutical Applications
Introduction to Patent CN108929262B
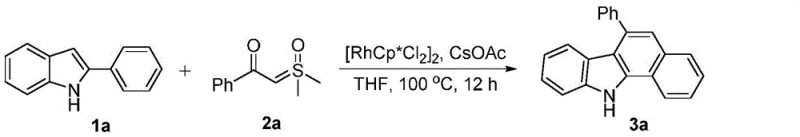
The pharmaceutical and functional material industries are constantly seeking more efficient pathways to access complex nitrogen-containing heterocycles, particularly the benzo[a]carbazole scaffold which is pivotal in drug discovery and organic electronics. Patent CN108929262B introduces a groundbreaking synthetic methodology that addresses long-standing challenges in constructing this privileged structure. By leveraging a rhodium(III)-catalyzed cascade reaction between readily available 2-arylindole compounds and sulfur ylides, this technology enables the direct assembly of benzo[a]carbazole derivatives under remarkably mild conditions. This innovation represents a significant leap forward for manufacturers of high-purity pharmaceutical intermediates and OLED materials, offering a streamlined alternative to traditional multi-step syntheses that often suffer from poor atom economy and harsh operational parameters.
The strategic value of this patent lies in its ability to transform simple starting materials into high-value fused heterocyclic systems with exceptional efficiency. For R&D directors and process chemists, the implication is clear: the barrier to entry for synthesizing these complex scaffolds is significantly lowered. The method operates effectively in common solvents like tetrahydrofuran and does not require stringent exclusion of moisture or oxygen, which drastically simplifies the engineering controls needed for scale-up. As we delve deeper into the technical specifics, it becomes evident that this approach is not merely an academic curiosity but a robust platform suitable for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply chain for critical downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzo[a]carbazole core has been fraught with synthetic inefficiencies that hinder large-scale production. Traditional routes often rely on multi-step sequences involving tedious protection and deprotection strategies, which inherently reduce the overall yield and increase waste generation. Many existing methods necessitate the use of highly reactive or unstable precursors that are difficult to source commercially, leading to supply chain bottlenecks and inflated raw material costs. Furthermore, conventional cyclization protocols frequently demand extreme temperatures, strong acids, or stoichiometric amounts of toxic heavy metal oxidants, creating significant environmental and safety liabilities for manufacturing facilities. These factors collectively result in a high cost of goods sold and extended lead times, making it challenging for procurement managers to secure consistent volumes of high-purity intermediates required for clinical trials and commercial drug launches.
The Novel Approach
In stark contrast, the methodology disclosed in CN108929262B utilizes a direct C-H functionalization strategy that bypasses the need for pre-functionalized substrates. By employing a sulfur ylide as a unique carbon source and a rhodium catalyst to drive the transformation, this novel approach achieves the formation of two new carbon-carbon bonds in a single operational step. The reaction proceeds with high atom economy, minimizing the generation of byproducts and simplifying the purification process to a standard silica gel column chromatography. This one-pot tandem reaction not only accelerates the synthesis timeline but also enhances the overall sustainability profile of the manufacturing process. The versatility of this method is further demonstrated by its compatibility with a broad spectrum of functional groups, allowing for the rapid generation of diverse analog libraries essential for structure-activity relationship studies in drug discovery.
![General reaction scheme for benzo[a]carbazole synthesis using 2-arylindoles and sulfur ylides](/insights/img/benzocarbazole-synthesis-rh-catalysis-pharma-supplier-20260302220548-01.webp)
The visual representation of this transformation highlights the elegance of the design, where the indole nitrogen and the aryl ring are seamlessly integrated into the larger carbazole framework. This structural convergence is achieved without the need for isolating sensitive intermediates, thereby reducing material loss and handling risks. For supply chain heads, this translates to a more resilient production process that is less susceptible to disruptions caused by the instability of intermediate species. The ability to run this reaction under air conditions further reduces the capital expenditure associated with specialized inert atmosphere equipment, making it an economically attractive option for both pilot-scale development and full commercial production of fine chemical intermediates.
Mechanistic Insights into Rh(III)-Catalyzed Cascade Reaction
The success of this synthetic route hinges on the precise orchestration of organometallic steps facilitated by the pentamethylcyclopentadienyl rhodium(III) catalyst. The mechanism initiates with the coordination of the rhodium center to the nitrogen atom of the 2-arylindole substrate, directing the activation of the proximal C-H bond on the aryl ring. This C-H metallation step is critical as it generates a reactive rhodacycle intermediate that is poised for subsequent insertion reactions. The sulfur ylide then acts as a carbene precursor or an electrophilic partner, inserting into the rhodium-carbon bond to extend the carbon skeleton. This insertion is followed by a series of migratory insertions and reductive elimination events that ultimately forge the new six-membered ring characteristic of the benzo[a]carbazole system. Understanding this catalytic cycle is vital for process optimization, as it explains the high regioselectivity observed in the formation of the final product.
Furthermore, the role of the cesium acetate additive cannot be overstated, as it serves as a crucial base to facilitate the C-H activation step and stabilize the cationic rhodium species generated during the catalytic turnover. The tolerance of the catalytic system to various electronic environments on the substrate allows for the incorporation of electron-withdrawing groups like fluorine and chlorine, as well as electron-donating groups like methyl and methoxy, without significant erosion of yield. This mechanistic robustness ensures that impurity profiles remain manageable, a key concern for R&D directors focused on regulatory compliance. The detailed characterization data provided in the patent examples, including NMR and HRMS, confirms the structural integrity of the products, validating the proposed mechanistic pathway and providing confidence in the reproducibility of the method across different batches of high-purity OLED material precursors.
How to Synthesize Benzo[a]carbazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocol parameters to maximize yield and purity. The general procedure involves dissolving the 2-arylindole starting material and the sulfur ylide reagent in a polar aprotic solvent, with tetrahydrofuran being the preferred medium based on the optimization data. The catalyst loading is kept relatively low, typically around 5 mol%, which helps in minimizing the residual metal content in the final API intermediate. The reaction mixture is then heated to a moderate temperature of 100°C, which provides sufficient thermal energy to overcome the activation barrier for C-H cleavage without promoting decomposition of the sensitive ylide reagent. Following the reaction period, a standard aqueous workup followed by chromatographic purification yields the target benzo[a]carbazole derivative in high purity.
- Dissolve 2-arylindole compound and sulfur ylide in a suitable solvent such as tetrahydrofuran.
- Add the Rh(III) catalyst [RhCp*Cl2]2 and cesium acetate additive to the reaction mixture.
- Heat the sealed reaction vessel to 100°C under air atmosphere for 12 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Rh(III)-catalyzed methodology offers substantial advantages that directly impact the bottom line and operational efficiency. The primary driver for cost reduction lies in the simplification of the synthetic route; by condensing multiple steps into a single pot, manufacturers can significantly reduce labor costs, solvent consumption, and reactor occupancy time. The elimination of cryogenic conditions or high-pressure hydrogenation steps further lowers the energy footprint of the process. Additionally, the use of commercially available 2-arylindoles and sulfur ylides ensures a stable supply of raw materials, mitigating the risk of shortages that often plague custom synthesis projects. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot cascade reaction eliminates the need for intermediate isolation and purification, which are traditionally resource-intensive operations. By avoiding the use of expensive protecting groups and stoichiometric oxidants, the overall material cost per kilogram of product is drastically lowered. The catalytic system operates efficiently at low loadings, reducing the expense associated with precious metal catalysts. Furthermore, the simplified workup procedure minimizes solvent waste disposal costs, contributing to a more sustainable and economically viable manufacturing process for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the ability to operate under air without strict moisture exclusion, greatly enhances process reliability. This reduces the dependency on specialized infrastructure and allows for production in a wider range of facilities, thereby diversifying the supply base. The broad substrate scope means that a single platform technology can be used to produce a variety of analogs, allowing for flexible inventory management and rapid response to changing market demands. This adaptability ensures that procurement managers can secure a consistent supply of high-quality intermediates even in volatile market conditions.
- Scalability and Environmental Compliance: The mild thermal conditions and lack of hazardous reagents make this process highly amenable to scale-up from gram to ton quantities. The reduced generation of chemical waste aligns with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing sites. The high atom economy of the transformation ensures that a maximum proportion of raw materials ends up in the final product, minimizing the environmental impact. This green chemistry profile is increasingly valued by end-users who are committed to sustainable sourcing practices, adding a competitive edge to suppliers who adopt this technology for the commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis for their specific projects, we have compiled answers to common inquiries regarding the operational details and scope of the technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for process engineers and quality assurance personnel who are responsible for validating new manufacturing routes. The following questions address key aspects such as reaction conditions, catalyst performance, and product purification, providing a clear roadmap for implementation.
Q: What are the optimal reaction conditions for this synthesis?
A: The patent specifies using THF as a solvent at 100°C with [RhCp*Cl2]2 catalyst and CsOAc additive under air conditions for 12 hours.
Q: Is an inert atmosphere required for this Rh(III) catalyzed reaction?
A: No, the methodology is robust enough to proceed efficiently under standard air conditions, simplifying operational requirements.
Q: What is the substrate scope for this benzo[a]carbazole synthesis?
A: The method tolerates a wide range of substituents including halogens, alkyl groups, and alkoxy groups on both the indole and aryl rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[a]carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN108929262B for accelerating drug development pipelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are equipped with state-of-the-art rigorous QC labs and analytical capabilities to meet stringent purity specifications required for GMP manufacturing. Our commitment to technical excellence allows us to offer customized solutions for the synthesis of benzo[a]carbazole derivatives, guaranteeing consistent quality and supply continuity for our global partners in the pharmaceutical and electronic materials sectors.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving efficiency and innovation in the production of high-value chemical intermediates.
