Advanced Synthesis of Esomeprazole Sodium: High Purity and Scalable Manufacturing Solutions
The pharmaceutical industry continuously demands more efficient, scalable, and environmentally benign processes for the production of critical proton pump inhibitors. Patent CN102887885A introduces a transformative preparation method for Esomeprazole Sodium, the S-isomer of omeprazole, which addresses long-standing challenges in chiral resolution and salt formation. This technology leverages a sophisticated inclusion complexation strategy followed by a direct crystallization protocol that eliminates the need for aqueous workups and toxic solvents. By strictly controlling temperature gradients during the resolution phase and utilizing a specific binary solvent system for salt formation, the method achieves exceptional optical purity exceeding 99.9% e.e. while maintaining a robust yield suitable for industrial application. For procurement and technical leaders, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, reducing both operational complexity and environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of esomeprazole from its racemic mixture has been plagued by inefficient and hazardous processing conditions. Prior art, such as WO2007/013743A1, relies heavily on aqueous solvent systems where the inclusion complex is formed and subsequently treated with sodium hydroxide. This approach necessitates multiple extraction steps to separate the resolving agent, leading to excessive solvent consumption and complex wastewater treatment requirements. Furthermore, the necessity to evaporate aqueous solutions to induce crystallization exposes the thermally unstable esomeprazole sodium to high temperatures, often resulting in significant impurity generation and reduced product quality. Other methods, like those described in CN1087739C, utilize highly toxic solvents such as benzene, posing severe safety risks and regulatory hurdles for modern manufacturing facilities. These conventional routes often suffer from low optical purity, difficult post-treatment procedures involving acidification and re-salification, and poor color profiles in the final product, making them unsuitable for cost-effective, large-scale commercial production.
The Novel Approach
The methodology disclosed in CN102887885A fundamentally reengineers the synthesis pathway to overcome these deficiencies through an anhydrous, direct crystallization strategy. Instead of relying on water-based extractions, the novel process forms an inclusion complex between omeprazole and (S)-(-)-[1,1'-dinaphthalene]-2,2'-diphenol in a controlled organic environment. The innovation lies in the precise thermal management: dissolving reactants at 65-75°C, maintaining reaction at 45-65°C, and crystallizing at 30-45°C. This specific window ensures that only the desired S-enantiomer complex precipitates, leaving the R-enantiomer in solution. Subsequently, the conversion to the sodium salt is performed in a mixture of organic solvents (Solvent B and Solvent C) where the resolving agent remains soluble while the Esomeprazole Sodium crystallizes directly. This eliminates the need for extraction, concentration, and evaporation steps, drastically simplifying the workflow and preserving the thermal integrity of the active pharmaceutical ingredient.
Mechanistic Insights into Chiral Resolution and Salt Formation
The core of this technological advancement is the stereoselective formation of the inclusion complex, which acts as the primary purification engine. The interaction between the racemic omeprazole and the chiral resolving agent, (S)-(-)-[1,1'-dinaphthalene]-2,2'-diphenol, is governed by specific host-guest chemistry that favors the S-enantiomer under the defined thermal conditions. By heating the mixture to 65-75°C, the system ensures complete dissolution and molecular mobility, allowing the thermodynamically stable diastereomeric complex to form. As the temperature is lowered to the crystallization range of 30-45°C, the solubility product of the S-complex is exceeded, prompting nucleation. Crucially, the temperature is kept high enough to prevent the co-precipitation of the R-enantiomer or the free resolving agent, which would otherwise contaminate the crystal lattice. This results in an off-white solid inclusion complex with high optical purity, setting the stage for the final conversion.
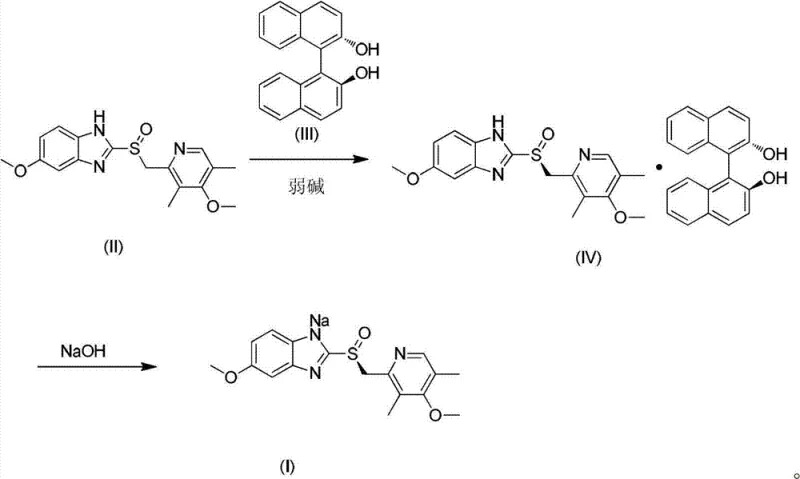
Following the isolation of the high-purity inclusion complex, the mechanism shifts to a displacement reaction facilitated by sodium hydroxide in a tailored solvent system. The choice of Solvent B (e.g., ethanol, acetone) and Solvent C (e.g., ethyl acetate, methyl tert-butyl ether) is critical for the differential solubility that drives the purification. Upon addition of NaOH, the benzimidazole nitrogen is deprotonated to form the sodium salt. In this specific binary solvent mixture, the Esomeprazole Sodium exhibits low solubility and precipitates out of the solution, whereas the resolving agent ((S)-(-)-[1,1'-dinaphthalene]-2,2'-diphenol) remains highly soluble. This phenomenon allows for a simple filtration step to isolate the pure product, effectively separating the chiral auxiliary without the need for chromatographic or extractive separation. The result is a product with impurity levels of the resolving agent below 0.1% and optical purity greater than 99.9% e.e., demonstrating the efficacy of the solubility-driven purification mechanism.
How to Synthesize Esomeprazole Sodium Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and temperature profiling to maximize yield and purity. The process begins with the preparation of the inclusion complex using alcohols or nitriles as the primary solvent, followed by the salt formation step using ketones or esters mixed with alcohols. Operators must adhere to the specified molar ratios, particularly keeping the resolving agent between 0.5 to 1.0 equivalents relative to omeprazole to optimize cost efficiency without sacrificing resolution power. The detailed standardized synthetic steps, including specific reagent quantities and stirring times derived from the patent embodiments, are outlined in the guide below to ensure reproducible results in a pilot or production setting.
- Dissolve omeprazole and the chiral resolving agent in an organic solvent (e.g., ethanol) at 65-75°C, then cool to 30-45°C to crystallize the high-purity inclusion complex.
- Treat the isolated inclusion complex with sodium hydroxide in a mixture of organic solvents (Solvent B and Solvent C) at 30-70°C.
- Cool the reaction mixture to 0-15°C to directly crystallize Esomeprazole Sodium, leaving the resolving agent in the mother liquor for filtration and recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic benefits beyond mere technical superiority. The elimination of aqueous extraction and evaporation steps translates directly into reduced utility consumption and shorter batch cycle times. By avoiding the use of high-boiling point aqueous mixtures that require significant energy to remove, the process lowers the overall thermal load on the manufacturing facility. Furthermore, the ability to recover the expensive chiral resolving agent from the mother liquor provides a clear pathway for raw material cost optimization, reducing the dependency on fresh inputs for every batch. This efficiency makes the supply of high-purity pharmaceutical intermediates more resilient against raw material price fluctuations.
- Cost Reduction in Manufacturing: The streamlined workflow significantly reduces operational expenditures by removing unit operations such as liquid-liquid extraction and rotary evaporation of water. Traditional methods often require multiple extraction cycles to remove the resolving agent, consuming vast amounts of organic solvents and generating large volumes of waste. In contrast, this method achieves purification through direct crystallization, minimizing solvent usage and waste disposal costs. Additionally, the avoidance of thermal degradation means higher effective yields of saleable product, further driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures associated with complex workups. Processes that rely on precise pH adjustments and multiple phase separations are prone to emulsions and yield losses, which can delay shipments. By simplifying the isolation to a filtration step, the process becomes more predictable and easier to scale. The use of common, non-restricted organic solvents like ethanol and ethyl acetate also mitigates supply risks associated with regulated or toxic chemicals like benzene, ensuring a smoother procurement process for raw materials.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns perfectly with modern green chemistry principles and regulatory standards. The exclusion of carcinogenic solvents like benzene and the reduction of aqueous waste streams simplify the environmental compliance burden for manufacturing sites. The process is inherently safer for operators due to the absence of high-temperature evaporation of unstable intermediates. This facilitates easier scale-up from pilot plants to multi-ton commercial production, as the heat transfer and mixing requirements are less demanding compared to heterogeneous extraction processes, ensuring consistent quality across large batches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Esomeprazole Sodium preparation method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity specifications, solvent recovery, and process scalability for potential partners and technical evaluators.
Q: How does this method achieve >99.9% optical purity?
A: The process utilizes precise temperature control during the inclusion complex formation (dissolving at 65-75°C, reacting at 45-65°C, and crystallizing at 30-45°C). This specific thermal profile prevents the co-crystallization of the R-enantiomer, ensuring the S-enantiomer is selectively isolated with high optical purity before the final salt formation step.
Q: What are the advantages regarding solvent usage compared to traditional methods?
A: Unlike prior art methods that rely on aqueous systems requiring energy-intensive evaporation or toxic solvents like benzene, this method operates entirely in organic phases. It eliminates the need for liquid-liquid extraction and aqueous evaporation, significantly reducing solvent load and energy consumption while avoiding thermal degradation of the product.
Q: Is the chiral resolving agent recoverable in this process?
A: Yes, the process is designed for sustainability. During the final crystallization of Esomeprazole Sodium, the resolving agent ((S)-(-)-[1,1'-dinaphthalene]-2,2'-diphenol) remains dissolved in the mother liquor due to the specific solvent combination (Solvent B + Solvent C). This allows the filtrate to be processed for the recovery and recycling of the expensive resolving agent, further lowering production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology presented in CN102887885A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of Esomeprazole Sodium meets the highest international standards for optical purity and chemical integrity. Our commitment to process excellence allows us to deliver high-purity pharmaceutical intermediates that support the development of next-generation generic and branded medications.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your total landed cost. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can serve as a reliable extension of your own R&D and production operations.
