Advanced Solid Acid Catalysis for Commercial Scale-Up of 4,4'-Dichlorodiphenyl Sulfone
Introduction to Next-Generation Sulfone Synthesis
The global demand for high-performance engineering plastics, particularly polysulfones, has driven an urgent need for more efficient and environmentally sustainable manufacturing routes for their key monomers. Patent CN114507165B introduces a groundbreaking method for synthesizing 4,4'-dichlorodiphenyl sulfone (DCPS) utilizing a novel solid acid catalysis system. This technology represents a significant paradigm shift from traditional liquid acid oxidation processes, addressing critical pain points regarding equipment corrosion, waste generation, and product purity. By leveraging a specially designed polymeric solid acid catalyst containing both carboxylic and sulfonic acid functional groups, this process facilitates the oxidation of 4,4'-dichlorodiphenyl sulfoxide with hydrogen peroxide under mild conditions. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: a pathway to ultra-high purity intermediates that simplifies downstream processing and aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-dichlorodiphenyl sulfone has relied heavily on homogeneous catalytic systems involving concentrated sulfuric acid or small molecular carboxylic acids like acetic acid. While these methods are chemically feasible, they present severe operational drawbacks that impact both capital expenditure and operational efficiency. The use of concentrated sulfuric acid creates a highly corrosive environment that necessitates expensive Hastelloy or glass-lined reactors, significantly increasing infrastructure costs. Furthermore, the separation of the catalyst from the reaction mixture is notoriously difficult; small molecule acids are often soluble in the reaction medium or form azeotropes with solvents, requiring energy-intensive distillation processes to recover valuable solvents. Additionally, the strong acidity can promote side reactions, such as the sulfonation of the aromatic ring, leading to the formation of difficult-to-remove impurities that compromise the chromaticity and thermal stability of the final polymer.
The Novel Approach
In stark contrast, the methodology disclosed in CN114507165B employs a heterogeneous solid acid catalyst that fundamentally alters the separation dynamics of the synthesis. This polymeric catalyst is insoluble in the organic reaction media, such as chlorobenzene or 1,2-dichloropropane, allowing for physical separation via simple filtration rather than complex chemical workups. This heterogeneity not only protects reactor hardware from corrosion but also enables the direct recycling of the catalyst after a straightforward washing and drying protocol. The process operates at moderate temperatures between 50°C and 80°C, reducing thermal stress on the materials and minimizing energy consumption. By avoiding the introduction of metal ions or strong mineral acids, the process inherently reduces the impurity profile, yielding a product that requires less rigorous purification to meet the exacting standards of the electronics and aerospace industries.
Mechanistic Insights into Solid Acid-Catalyzed Oxidation
The efficacy of this synthesis relies on the unique dual-functional nature of the solid acid catalyst, which acts as both a proton donor and a carrier for active oxidizing species. The polymer backbone, synthesized from monomers containing olefin, carboxylic acid, and sulfonic acid groups, creates a micro-environment that stabilizes hydrogen peroxide. In the presence of the acidic protons ionized from the catalyst surface, hydrogen peroxide is activated to form peroxyacid species in situ. This mechanism avoids the bulk decomposition of hydrogen peroxide, ensuring that the oxidizing power is directed specifically toward the sulfoxide substrate. The carboxylic acid groups on the polymer chain react with hydrogen peroxide to generate peroxyacid groups, which possess significantly stronger oxidizability than hydrogen peroxide alone, thereby accelerating the conversion of the sulfoxide to the sulfone.
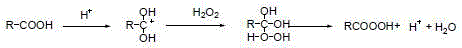
Following the formation of the active peroxyacid species on the solid support, the oxidation of the 4,4'-dichlorodiphenyl sulfoxide proceeds through a nucleophilic attack on the oxygen atom of the peroxy group. The solid acid catalyst participates directly in this cycle, where the peroxy carboxylic acid moiety transfers an oxygen atom to the sulfur center of the substrate. Once the oxidation is complete, the peroxy group is reduced back to a carboxylic acid group, regenerating the active site on the polymer for subsequent cycles. This cyclic regeneration is crucial for the economic viability of the process, as it allows a single batch of catalyst to be used repeatedly without significant loss of activity. The structural integrity of the catalyst, maintained by its cross-linked network, ensures that it remains mechanically stable even after prolonged exposure to oxidative conditions and elevated temperatures.
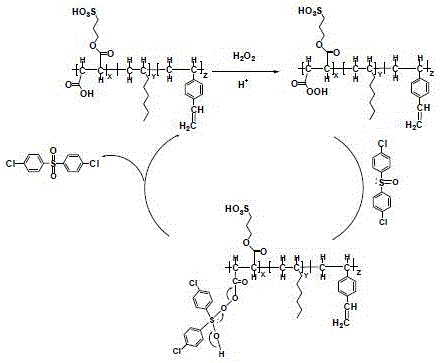
The synthesis of the catalyst itself is a testament to the precision of modern polymer chemistry, involving a multi-step process that begins with the esterification of 3-hydroxypropanesulfonic acid with fumaric acid. This reaction yields a monoester compound containing the necessary olefinic and acidic functionalities, which is subsequently polymerized with divinylbenzene and 1-octene to form a porous microsphere structure. The inclusion of pore-forming agents during polymerization creates a high specific surface area, maximizing the exposure of active acid sites to the reactants. This tailored porosity is essential for mass transfer, ensuring that the bulky diphenyl sulfoxide molecules can access the internal catalytic sites efficiently. The resulting material exhibits a defined acid strength and particle size distribution, parameters that are critical for optimizing the reaction kinetics and facilitating the final filtration step.
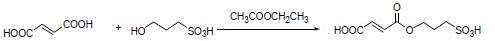
How to Synthesize 4,4'-Dichlorodiphenyl Sulfone Efficiently
Implementing this synthesis route in a pilot or production setting requires strict adherence to the optimized parameters regarding catalyst loading, solvent ratios, and temperature control to maximize yield and purity. The process is designed to be robust, tolerating slight variations in reagent quality while maintaining consistent output, which is vital for commercial reliability. Operators should focus on the efficient recovery of the solvent phase from the water-oil two-phase mixture post-reaction, as this directly impacts the overall cost structure of the manufacturing campaign. The following guide outlines the standardized operational procedure derived from the patent examples to ensure reproducible results.
- Mix 4,4'-dichlorodiphenyl sulfoxide with a porous solid acid catalyst and an organic solvent such as chlorobenzene or 1,2-dichloropropane in a reaction vessel.
- Heat the mixture to 50-80°C and dropwise add hydrogen peroxide while stirring to initiate the oxidation reaction.
- Filter the hot reaction mixture to recover the solid catalyst, treat the filtrate with activated carbon, and crystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid acid catalytic process offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of corrosive mineral acids translates directly into reduced maintenance costs and extended equipment lifespan, lowering the total cost of ownership for production facilities. Furthermore, the ability to recycle the catalyst multiple times without significant degradation in performance drastically reduces the consumption of auxiliary materials, contributing to a leaner and more cost-effective supply chain. The simplified workup procedure, which replaces complex distillation columns with basic filtration units, reduces the energy footprint of the plant and shortens the batch cycle time, thereby enhancing overall throughput capacity.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive neutralization and waste treatment steps associated with liquid acid catalysts. By utilizing a solid acid that can be filtered and reused, the facility eliminates the continuous purchase of fresh catalyst and the disposal costs of spent acidic waste streams. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which reduces the load on purification units and increases the effective yield of saleable product per batch. This efficiency gain allows for a more competitive pricing structure in the market for high-purity phenyl sulfone intermediates.
- Enhanced Supply Chain Reliability: The robustness of the solid acid catalyst ensures consistent production schedules, as there is less risk of batch failure due to catalyst deactivation or contamination. The raw materials required for the catalyst synthesis, such as fumaric acid and sulfonic acid derivatives, are widely available commodity chemicals, mitigating the risk of supply bottlenecks. Moreover, the solvent recovery system, which allows for the direct reuse of the organic phase after simple phase separation, insulates the production line from volatility in solvent markets. This self-sufficiency in key process inputs strengthens the resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the heterogeneous nature of the reaction, which avoids the heat transfer limitations often encountered in highly exothermic liquid acid oxidations. The absence of heavy metal contaminants and strong mineral acids simplifies wastewater treatment, making it easier to meet increasingly stringent environmental regulations. The process generates significantly less hazardous waste, aligning with corporate sustainability goals and reducing the regulatory burden on the manufacturing site. This environmental compatibility is a critical factor for securing long-term operating permits and maintaining a social license to operate in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid acid catalytic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this method into existing production lines.
Q: What are the advantages of using a solid acid catalyst over concentrated sulfuric acid for DCPS synthesis?
A: Solid acid catalysts eliminate equipment corrosion caused by strong mineral acids, simplify separation via filtration instead of complex distillation, and prevent the formation of aromatic sulfonic acid by-products, ensuring higher product purity.
Q: Can the solid acid catalyst be reused in industrial production?
A: Yes, the polymeric solid acid catalyst is insoluble in organic solvents and can be recovered by simple hot filtration. Data indicates it maintains high catalytic activity and product purity even after five consecutive recycling cycles.
Q: What purity levels can be achieved with this novel oxidation method?
A: The method consistently achieves product purities exceeding 99.8%, with specific examples reaching 99.89%, which meets the stringent requirements for high-performance engineering plastics like polysulfone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dichlorodiphenyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of downstream engineering plastics and pharmaceutical applications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We are committed to delivering 4,4'-dichlorodiphenyl sulfone with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for polysulfone synthesis. Our capability to implement advanced catalytic technologies like the one described in CN114507165B underscores our dedication to providing superior chemical solutions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and our proven track record in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
