Revolutionizing Diaryl Sulfone Production With High-Yield Catalytic Coupling Technology
Introduction to Advanced Diaryl Sulfone Manufacturing
The global demand for high-performance polymers and pharmaceutical intermediates relies heavily on the consistent supply of diaryl sulfones, particularly 4,4'-dichlorodiphenyl sulfone (DCDPS). A significant technological breakthrough in this sector is detailed in Chinese Patent CN107922325B, which outlines an improved method for preparing diaryl sulfones through a highly efficient catalytic coupling process. This innovation addresses long-standing challenges in the industry, specifically the trade-off between yield, selectivity, and the economic burden of stoichiometric dehydrating agents. By shifting from traditional stoichiometric dehydration to a catalytic system with continuous water removal, this technology offers a pathway to substantially lower production costs while maintaining rigorous purity standards required by top-tier R&D departments.
The core of this invention lies in a two-stage sequence: first, the sulfonation of an aryl compound using sulfur trioxide to form a sulfonic acid intermediate, and second, the coupling of this intermediate with an additional aryl compound in the presence of a catalyst. Crucially, the process mandates the continuous removal of water generated during the coupling step, which drives the equilibrium forward without the need for excessive chemical dehydrators. This approach not only simplifies the downstream purification workflow but also enhances the overall atom economy of the reaction, positioning it as a superior alternative for reliable diaryl sulfone suppliers seeking to optimize their manufacturing footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl sulfones has been plagued by inefficiencies that impact both cost and environmental compliance. Early methods, such as those described in U.S. Patent 4,937,387, utilized a two-step process starting from monochlorobenzene and sulfur trioxide but suffered from low conversion rates, often averaging only around 20.8% based on the sulfonic acid feed. Other approaches, like the one found in U.S. Patent 4,983,773, attempted to improve yields by using boric acid but still faced significant hurdles in purification, often resulting in tarry by-products that were difficult to separate. Furthermore, more modern attempts described in WO 2012/143281 achieved high yields by employing trifluoroacetic anhydride (TFAA) as a dehydrating agent; however, this introduced a severe economic bottleneck due to the requirement for stoichiometric quantities of expensive reagents, generating large volumes of trifluoroacetic acid waste that necessitates complex disposal protocols.
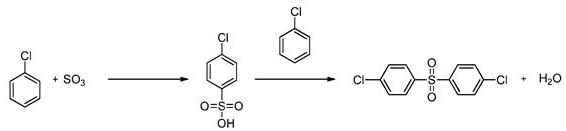
The Novel Approach
The methodology disclosed in CN107922325B represents a paradigm shift by eliminating the dependency on stoichiometric dehydrating agents entirely. Instead, it leverages a catalytic amount of inexpensive materials—such as boric acid, metal oxides, or Lewis acids—combined with physical water removal techniques like distillation. This novel approach allows the reaction to proceed under anhydrous conditions where the water produced is continuously stripped from the reaction mixture, effectively driving the equilibrium toward the desired sulfone product. Experimental data within the patent demonstrates that this method can achieve crude yields ranging from 82% to 93% relative to the sulfonic acid, with purified yields consistently exceeding 70%. Moreover, the process exhibits exceptional selectivity, minimizing the formation of unwanted 2,4' and 3,4' isomers to less than 20%, thereby drastically reducing the burden on crystallization and purification units.
Mechanistic Insights into Catalytic Sulfonation and Coupling
The chemical elegance of this process is rooted in its precise control over reaction thermodynamics and kinetics. The first stage involves the electrophilic aromatic substitution of an aryl halide, typically monochlorobenzene, with sulfur trioxide. This sulfonation step is exothermic and must be carefully managed, typically occurring between 30°C and 100°C, to prevent polysulfonation or degradation. The resulting 4-chlorobenzenesulfonic acid serves as the key electrophile for the subsequent coupling. Unlike traditional Friedel-Crafts type reactions that might struggle with reversibility in the presence of water, this system is engineered to operate under strictly anhydrous initiation conditions, ensuring that the sulfonic acid remains active and ready for nucleophilic attack by the second aryl molecule.
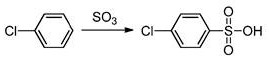
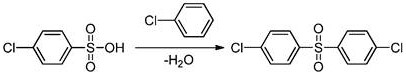
In the critical coupling phase, the presence of a catalyst facilitates the formation of the carbon-sulfur bond between the sulfonic acid and the aryl halide. The mechanism likely involves the activation of the sulfonic acid group by the catalyst, making the sulfur atom more susceptible to nucleophilic attack by the electron-rich aromatic ring of the chlorobenzene. As the reaction proceeds at elevated temperatures between 180°C and 240°C, water is generated as a byproduct. The continuous removal of this water is the linchpin of the mechanism; by preventing water accumulation, the reverse hydrolysis reaction is suppressed, and the equilibrium is forced toward the formation of the diaryl sulfone. This dynamic control allows for the use of catalytic rather than stoichiometric promoters, fundamentally altering the cost structure of the synthesis.
How to Synthesize 4,4'-Dichlorodiphenyl Sulfone Efficiently
Implementing this advanced synthesis route requires careful attention to process parameters, particularly regarding temperature gradients and water management. The patent outlines a robust protocol where the sulfonic acid mixture is first prepared and then transferred to a coupling reactor equipped with a Dean-Stark trap or similar distillation apparatus. The addition of the aryl halide is performed continuously over a period of 7 to 13 hours, allowing for precise thermal control and preventing localized hot spots that could degrade the product. For R&D teams looking to replicate or adapt this chemistry, the key lies in maintaining the water concentration below 10 wt.% throughout the coupling step, ideally keeping it under 1 wt.% to maximize yield. Detailed standardized synthetic steps for laboratory and pilot-scale execution are provided in the guide below.
- React monochlorobenzene with sulfur trioxide at controlled temperatures (30-100°C) to generate 4-chlorobenzenesulfonic acid.
- Initiate the coupling step by adding a catalyst (e.g., boric acid or metal oxide) to the sulfonic acid mixture under anhydrous conditions.
- Continuously add excess monochlorobenzene while removing generated water via distillation at 180-240°C to drive the reaction to completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this catalytic process offers compelling strategic benefits beyond mere technical performance. The most immediate impact is seen in raw material cost optimization. By replacing stoichiometric amounts of expensive dehydrating agents like TFAA with catalytic quantities of commodity chemicals such as boric acid or iron oxide, the direct material cost per kilogram of product is significantly reduced. Additionally, the elimination of massive volumes of acidic waste streams simplifies environmental compliance and reduces the overhead associated with waste treatment facilities. This streamlined workflow translates into a more resilient supply chain, as the reliance on specialized, high-cost reagents is minimized in favor of widely available industrial chemicals.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it decouples yield from the consumption of expensive reagents. In traditional methods, achieving high conversion often required molar equivalents of dehydrating agents, which not only increased material costs but also created downstream separation challenges. By utilizing a catalytic system where water is physically removed, the process achieves high conversion with minimal chemical input. This reduction in reagent intensity directly lowers the variable cost of goods sold (COGS), allowing manufacturers to offer more competitive pricing for high-purity diaryl sulfones without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of niche reagents. This method relies on monochlorobenzene and sulfur trioxide, which are bulk commodities with stable global supply chains. The catalysts employed, such as metal oxides or boric acid, are also readily sourced from multiple vendors, mitigating the risk of single-source bottlenecks. Furthermore, the process flexibility allows for both batch and continuous operation modes, giving production planners the agility to scale output up or down based on market demand without requiring extensive retooling or new equipment investments.
- Scalability and Environmental Compliance: From an operational perspective, the continuous removal of water and the ability to recover unreacted starting materials make this process highly scalable. The patent data indicates that unreacted sulfonic acid can be recovered and recycled, further enhancing atom economy. Environmentally, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical manufacturing. The process produces fewer by-products and avoids the generation of fluorinated waste associated with TFAA methods, facilitating easier permitting and long-term sustainability goals for large-scale production facilities.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is essential for stakeholders evaluating its adoption. The following questions address common concerns regarding purity, scalability, and catalyst selection based on the technical disclosures in the patent. These insights are derived directly from the experimental data and embodiment descriptions provided in CN107922325B, ensuring that the information reflects the actual capabilities of the process rather than theoretical projections.
Q: What are the primary advantages of this catalytic method over traditional stoichiometric dehydration?
A: Unlike methods requiring stoichiometric amounts of expensive dehydrating agents like trifluoroacetic anhydride (TFAA), this process uses catalytic amounts of inexpensive materials like boric acid or metal oxides, significantly reducing raw material costs and waste generation.
Q: How does the process control isomer formation to ensure high 4,4'-selectivity?
A: By maintaining strict anhydrous conditions during the initiation of the coupling step and utilizing specific catalysts such as boric acid or iron oxide, the process minimizes the formation of 2,4' and 3,4' isomers, achieving isomeric purity greater than 85% in the crude product.
Q: Is this synthesis method scalable for industrial production volumes?
A: Yes, the patent explicitly describes both batch and continuous process embodiments. The ability to continuously remove water and add reactants allows for efficient heat management and reaction control, making it highly suitable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dichlorodiphenyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify isomeric purity and trace impurity profiles. Whether your application requires polymer-grade DCDPS for high-performance plastics or pharmaceutical-grade intermediates for drug synthesis, our infrastructure is designed to support your specific technical requirements with consistency and reliability.
We invite you to explore how this innovative catalytic technology can enhance your product portfolio and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production metrics. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to validate the performance of our materials against your internal standards. Let us collaborate to build a more efficient and sustainable supply chain for your critical chemical needs.
