Advanced Iodine-Catalyzed Synthesis of Substituted Pyrazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for constructing nitrogen-containing heterocycles, particularly pyrazole scaffolds which serve as critical building blocks for numerous active pharmaceutical ingredients. Patent CN1154637C presents a significant technological advancement in this domain by disclosing a highly efficient preparation method for substituted pyrazole compounds. This innovation leverages a unique combination of sulfuric acid acting simultaneously as a solvent, condensing agent, and oxidant, facilitated by catalytic quantities of iodine or iodine-releasing compounds. Unlike traditional routes that often rely on stoichiometric amounts of hazardous oxidants or expensive transition metal catalysts, this process offers a streamlined approach that aligns perfectly with modern green chemistry principles while maintaining high industrial viability. For R&D directors and procurement specialists alike, understanding the nuances of this iodine-catalyzed cyclization is essential for optimizing supply chains and reducing the cost of goods sold for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole derivatives has been plagued by significant operational and safety challenges that hinder large-scale commercial adoption. Conventional methodologies frequently employ aggressive oxidizing agents such as chlorine, alkali metal hypochlorites, or even toxic elements like selenium to effect the necessary dehydrogenation of pyrazoline intermediates. These reagents not only pose severe handling risks due to their corrosive and toxic nature but also generate substantial quantities of hazardous waste, including hydrogen sulfide and selenides, which complicate downstream waste treatment and increase environmental compliance costs. Furthermore, alternative gas-phase dehydrogenation methods utilizing palladium or platinum catalysts, while effective, introduce prohibitive capital expenditures due to the high cost of noble metals and the specialized reactor engineering required to manage high-temperature gas flows. These legacy processes often suffer from poor atom economy and require multi-step sequences that erode overall yield and extend production lead times, making them less attractive for cost-sensitive bulk manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a liquid-phase reaction system where sulfuric acid serves a triple function, drastically simplifying the reactor setup and reagent inventory. By employing catalytic amounts of iodine or readily available iodide salts, the process achieves efficient oxidative cyclization under relatively moderate thermal conditions, typically ranging from 80°C to 200°C. This method eliminates the need for external stoichiometric oxidants, as the sulfuric acid itself participates in the redox cycle, thereby reducing the chemical footprint of the reaction. The ability to use simple, commodity-grade starting materials such as carbonyl compounds and hydrazine hydrate, combined with the recyclability of the iodine catalyst through distillation, creates a closed-loop system that minimizes raw material loss. This streamlined protocol not only enhances safety by avoiding toxic by-products but also significantly improves the economic feasibility of producing high-purity pyrazole derivatives on a multi-ton scale.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the intricate interplay between the carbonyl substrate, hydrazine, and the iodine-sulfuric acid matrix. The reaction initiates with the condensation of the carbonyl compound and hydrazine to form a hydrazone intermediate, a standard step in pyrazole synthesis. However, the subsequent dehydrogenation and aromatization are where the iodine catalyst plays a pivotal role. In the presence of strong sulfuric acid, the iodine species facilitate the removal of hydrogen atoms from the dihydropyrazole ring, driving the equilibrium towards the fully aromatic pyrazole structure. The sulfuric acid acts as the terminal oxidant, being reduced to sulfur dioxide in the process, which can be visually monitored as a gas evolution indicating reaction progress. This mechanism avoids the radical pathways often seen with chlorine-based oxidants, which can lead to unwanted halogenation of the aromatic ring or side-chain degradation, thus ensuring a cleaner impurity profile.
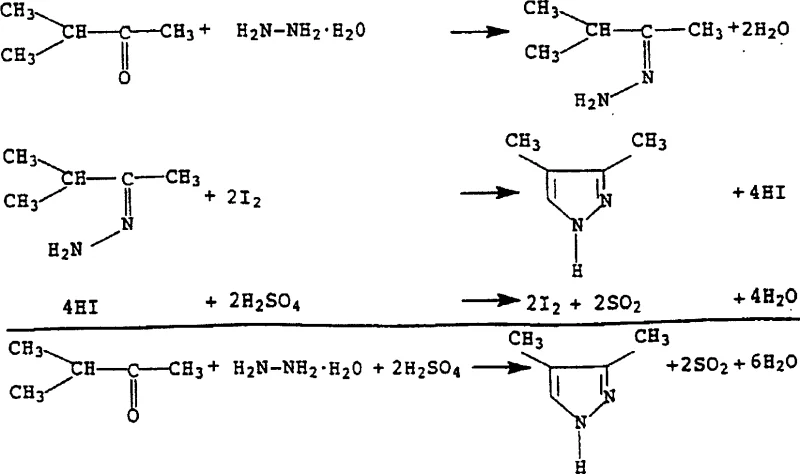
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical grade manufacturing. The mild nature of the iodine catalyst prevents over-oxidation of sensitive functional groups that might be present on the R1, R2, or R3 substituents of the carbonyl starting material. For instance, when synthesizing derivatives with electron-rich aryl groups, traditional strong oxidants might attack the ring system, whereas the iodine-mediated pathway preserves the structural integrity of the substrate. Additionally, the reaction conditions allow for the continuous removal of water via distillation, which shifts the equilibrium towards product formation according to Le Chatelier's principle, thereby maximizing conversion rates. The resulting crude product typically exhibits high purity, often exceeding 90% prior to final purification, which reduces the burden on downstream crystallization or distillation units and lowers the overall energy consumption of the plant.
How to Synthesize Substituted Pyrazoles Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and safety. The process generally involves preparing a suspension of sulfuric acid and the iodine catalyst, followed by the controlled addition of the carbonyl and hydrazine components at elevated temperatures. Water formed during the condensation is continuously distilled off to drive the reaction to completion, after which the mixture is neutralized and the product is extracted.
- Prepare a reaction suspension containing 30-100% sulfuric acid and a catalytic amount of iodine or iodide salt (0.01-10 mol%).
- Add the carbonyl compound (e.g., methyl isopropyl ketone) and hydrazine hydrate to the mixture while maintaining a temperature between 80-200°C.
- Distill off water to drive the reaction, then neutralize the mixture with base and extract the pyrazole product using organic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iodine-catalyzed process represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of expensive noble metal catalysts like palladium or platinum removes a significant variable cost component and mitigates the risk associated with the volatility of precious metal markets. Furthermore, the reliance on commodity chemicals such as sulfuric acid and simple ketones ensures that raw material sourcing remains stable and unaffected by the supply chain bottlenecks often seen with specialized reagents. The simplified work-up procedure, which avoids complex filtration steps for catalyst recovery, translates directly into reduced labor hours and faster batch turnover times, allowing manufacturing facilities to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the substitution of stoichiometric oxidants with catalytic iodine and sulfuric acid. By removing the need for costly reagents and the associated waste disposal fees for toxic by-products like selenium or heavy metal sludge, the overall variable cost per kilogram of product is significantly lowered. Additionally, the potential for recycling the iodine catalyst from the distillate further enhances the economic efficiency, creating a sustainable loop that minimizes raw material expenditure. This economic model is particularly advantageous for high-volume production runs where marginal savings per unit compound into substantial annual financial benefits.
- Enhanced Supply Chain Reliability: The robustness of this chemical route contributes directly to supply chain resilience. Since the key reagents are widely available bulk chemicals with established global supply networks, the risk of production stoppages due to raw material shortages is minimized. The process tolerance for various carbonyl substrates also provides flexibility; if a specific ketone becomes temporarily unavailable, the chemistry can often be adapted to use structurally similar alternatives without requiring a complete process re-validation. This adaptability ensures continuous production capability, which is critical for maintaining just-in-time delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the use of standard stainless steel or glass-lined reactors compatible with sulfuric acid. The absence of high-pressure hydrogenation steps or cryogenic conditions simplifies the engineering requirements and reduces capital investment barriers. From an environmental standpoint, the generation of sulfur dioxide, while requiring scrubbing, allows for the potential recovery of sulfuric acid, aligning with circular economy goals. The reduction in hazardous waste streams simplifies regulatory compliance and lowers the long-term liability associated with environmental remediation, making this a future-proof manufacturing solution.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams.
Q: How does the iodine catalyst improve selectivity compared to traditional oxidants?
A: The iodine catalyst facilitates a mild dehydrogenation process that avoids the harsh conditions associated with chlorine or hypochlorites, significantly reducing the formation of chlorinated by-products and improving the overall impurity profile of the pyrazole derivative.
Q: Can the sulfuric acid be recycled in this process?
A: Yes, the process design allows for the potential recovery of sulfuric acid. The reaction generates sulfur dioxide which can be converted back to sulfuric acid via SO3, and the spent acid residue can be processed to recover valuable components, enhancing the environmental sustainability of the manufacturing cycle.
Q: What is the typical purity achievable for 3,4-dimethylpyrazole using this method?
A: According to the patent examples, this method can achieve purities exceeding 99% for specific derivatives like 3,4-dimethylpyrazole after standard purification steps such as distillation, demonstrating its suitability for high-specification pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our technical team has extensively evaluated the iodine-catalyzed pyrazole synthesis route described in CN1154637C and confirmed its viability for large-scale operations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyrazole intermediate delivered meets the exacting standards required for API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your specific supply chain. By leveraging our expertise, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, allowing you to quantify the economic benefits of switching to this greener, more efficient process. We encourage you to contact us to obtain specific COA data for our pyrazole derivatives and to receive detailed route feasibility assessments that demonstrate our commitment to quality and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
