Advanced Synthesis of Nifuratel Related Substance C for Pharmaceutical Quality Control
Advanced Synthesis of Nifuratel Related Substance C for Pharmaceutical Quality Control
The pharmaceutical industry faces relentless pressure to ensure the absolute purity and safety of active pharmaceutical ingredients (APIs), particularly for broad-spectrum antibiotics like Nifuratel. As detailed in patent CN112724098A, a groundbreaking preparation method has been developed for Nifuratel Related Substance C, a critical impurity that must be strictly monitored to meet global regulatory standards. This innovative synthetic route addresses the historical challenges of low yield and difficult separation associated with isolating this specific compound from reaction mixtures. By shifting from passive isolation to active, directed synthesis, manufacturers can now secure high-purity reference standards essential for quality control laboratories. The structural complexity of Nifuratel, featuring a nitrofuran moiety and an oxazolidinone ring, necessitates precise impurity profiling to prevent toxic side effects in patients. 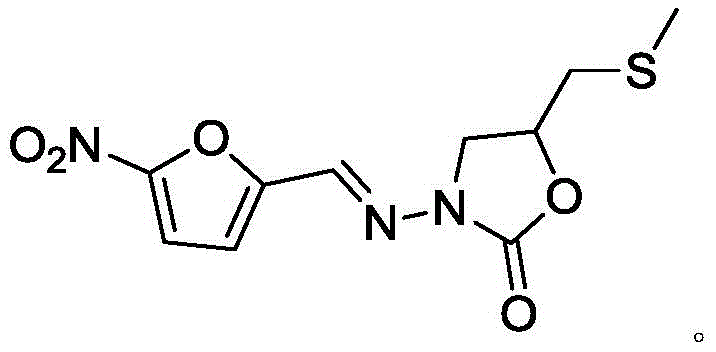
Nifuratel is a vital therapeutic agent used globally for treating vaginal mixed infections, exhibiting potent activity against Trichomonas, Candida, and various bacterial pathogens. However, its chemical instability during synthesis can lead to the formation of related substances that compromise drug safety. Regulatory bodies mandate that known impurities be controlled below 0.15% and unknown impurities below 0.10%, making the availability of authentic impurity standards non-negotiable for drug registration. The traditional reliance on preparative liquid chromatography to isolate Related Substance C from bulk reaction solutions was inefficient, costly, and yielded insufficient quantities for comprehensive toxicological studies. This new patent introduces a robust, two-step chemical synthesis that transforms simple, commercially available feedstocks into the target oxazolidinone derivative with exceptional efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Nifuratel Related Substance C was a bottleneck in the quality assurance workflow for this antibiotic. Conventional methods relied entirely on the isolation of the impurity from the main Nifuratel reaction liquid using preparative liquid phase chromatography. This approach suffered from inherently low yields because the impurity is formed only in trace amounts as a byproduct, meaning vast quantities of reaction mixture had to be processed to obtain milligrams of the target substance. Furthermore, the separation difficulty was high due to the structural similarity between the API and its degradation products, often requiring multiple purification cycles that degraded the sample further. The high cost of preparative HPLC solvents and the time-intensive nature of the process made it economically unviable for large-scale production of reference standards. Consequently, research into the toxicology and stability of this specific related substance was severely limited, creating a gap in the safety data required for regulatory filings in major markets like the US and Europe.
The Novel Approach
The novel approach disclosed in the patent fundamentally reimagines the supply chain for this critical intermediate by synthesizing it directly rather than isolating it. The process utilizes a straightforward condensation and cyclization strategy starting from epichlorohydrin and hydrazine hydrate, bypassing the need for the complex Nifuratel synthesis entirely. This direct route allows for the deliberate construction of the 3-amino-5-chloromethyl oxazoline-2-ketone skeleton with high atom economy. By controlling the stoichiometry and reaction conditions precisely, the method achieves yields ranging from 88.3% to 91.2%, which is a dramatic improvement over the negligible recovery rates of isolation techniques. The resulting product exhibits purity levels exceeding 99.9%, ensuring that it serves as a reliable benchmark for HPLC calibration and impurity quantification. This shift from extraction to synthesis not only secures the supply of the reference material but also drastically reduces the cost per gram, making routine quality control testing more affordable for generic manufacturers.
Mechanistic Insights into Epichlorohydrin-Based Cyclization
The core of this technological breakthrough lies in the efficient ring-opening and subsequent cyclization mechanism involving epichlorohydrin. In the first stage, hydrazine hydrate acts as a nucleophile, attacking the less hindered carbon of the epoxide ring in epichlorohydrin under thermal conditions of 70-90°C. This ring-opening reaction generates 3-chloro-2-hydroxy-propylhydrazine, a key intermediate that retains the chloromethyl group necessary for the final structure. The reaction is driven to completion by using a slight molar excess of hydrazine hydrate (1.05 to 1.3 equivalents), ensuring full conversion of the epoxide while minimizing polymerization side reactions. Following the initial coupling, water and unreacted hydrazine are distilled off, simplifying the reaction matrix before the cyclization step begins. This careful management of the intermediate prevents hydrolysis of the chloromethyl group, which is a common degradation pathway in aqueous acidic or basic environments.
The second stage involves the formation of the oxazolidinone ring through reaction with diethyl carbonate in the presence of an alkaline catalyst. 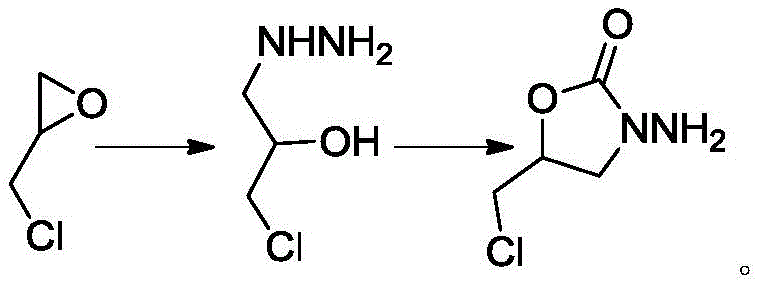
Diethyl carbonate serves as both a carbonyl source and a solvent component, reacting with the hydrazine and hydroxyl groups to close the five-membered heterocyclic ring. The use of bases such as sodium methoxide, sodium ethoxide, or sodium hydroxide facilitates the deprotonation of the hydroxyl group, enabling it to attack the carbonyl carbon of the carbonate. The reaction is conducted at moderate temperatures between 50-70°C, which provides sufficient energy for cyclization without promoting thermal decomposition of the sensitive chloromethyl side chain. The final workup involves adjusting the pH to a neutral range of 4-8 using hydrochloric acid, which quenches the base and precipitates inorganic salts, allowing for clean extraction into ethyl acetate. This mechanistic precision ensures that the chlorine atom remains intact, distinguishing this synthesis from other routes that might inadvertently substitute the halogen.
How to Synthesize 3-Amino-5-chloromethyl Oxazoline-2-Ketone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters regarding temperature and molar ratios to maximize yield and purity. The process is designed to be operationally simple, utilizing standard glassware and heating mantles without the need for high-pressure reactors or cryogenic cooling. Operators should focus on the precise control of the hydrazine addition rate in the first step to manage the exotherm, followed by careful monitoring of the pH during the final acidification to prevent product degradation. The detailed standardized synthesis steps, including specific solvent volumes and stirring times, are outlined in the procedural guide below to ensure reproducibility across different batches.
- React epichlorohydrin with hydrazine hydrate at 70-90°C for 2-3 hours to form 3-chloro-2-hydroxy-propylhydrazine, then distill off excess reagents.
- Add solvent (ethanol or dichloromethane) and diethyl carbonate to the intermediate, then slowly introduce an alkaline catalyst like sodium hydroxide.
- Maintain reaction temperature at 50-70°C until completion, adjust pH to 4-8 with hydrochloric acid, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift to a direct synthesis model decouples the supply of this critical reference standard from the volatility of the main Nifuratel production lines. Previously, if the main API batch failed or had low impurity levels, the supply of the reference standard would be interrupted; now, it can be produced on demand independently. This reliability is crucial for maintaining continuous quality control operations in GMP facilities, where the absence of a validated impurity standard can halt the release of finished drug products. Furthermore, the use of commodity chemicals like epichlorohydrin and diethyl carbonate ensures that raw material costs remain stable and predictable, shielding the budget from the price fluctuations often seen with specialized fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the elimination of expensive preparative chromatography processes. Traditional isolation required massive volumes of HPLC-grade solvents and significant instrument time, both of which are cost-prohibitive at scale. By contrast, this synthesis relies on bulk solvents like ethanol or dichloromethane and simple filtration techniques, leading to a drastic reduction in operational expenditure. The high yield of over 88% means that less raw material is wasted, directly lowering the cost of goods sold (COGS) for the reference standard. Additionally, the removal of transition metal catalysts or exotic reagents simplifies the waste stream treatment, further reducing disposal costs associated with hazardous chemical waste management.
- Enhanced Supply Chain Reliability: Sourcing high-purity impurities is often a logistical challenge, with long lead times from specialized niche suppliers. This method empowers manufacturers to produce the substance in-house or source it from general fine chemical suppliers who have the capacity to run batch reactions. The raw materials, specifically epichlorohydrin and hydrazine hydrate, are produced globally in massive quantities for the epoxy resin and agrochemical industries, ensuring a robust and uninterrupted supply chain. This abundance mitigates the risk of shortages that could delay regulatory submissions or quality audits. The simplicity of the process also means that technology transfer to contract manufacturing organizations (CMOs) is rapid, allowing for flexible scaling of production volume based on demand without lengthy qualification periods.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is highly favorable for scale-up. The reaction conditions are mild, operating at atmospheric pressure and temperatures below 100°C, which reduces energy consumption and safety risks associated with high-pressure vessels. The solvents used, such as ethanol and ethyl acetate, are relatively green and easily recoverable through distillation, aligning with modern sustainability goals in pharmaceutical manufacturing. The absence of heavy metals or persistent organic pollutants in the reaction scheme simplifies the environmental impact assessment, facilitating faster regulatory approval for the manufacturing site. This compliance readiness is a significant asset for companies aiming to expand their production capacity while adhering to increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Nifuratel Related Substance C. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps R&D and procurement teams evaluate the feasibility of integrating this intermediate into their quality control workflows. For further specific technical data or custom synthesis requirements, direct consultation with our technical team is recommended.
Q: What is the primary advantage of this new synthesis method for Nifuratel Related Substance C?
A: The primary advantage is the significant improvement in yield and purity compared to traditional isolation methods. While conventional methods rely on extracting trace amounts from reaction liquids with low efficiency, this novel direct synthesis route achieves yields exceeding 88% and purity levels greater than 99.9%, making it ideal for generating reference standards.
Q: Why is controlling Nifuratel Related Substance C critical for pharmaceutical manufacturers?
A: Regulatory agencies such as the FDA and EMA impose strict limits on unknown drug impurities, typically requiring identification and quantification for any species present above 0.10%. Since Nifuratel can degrade or form harmful byproducts during synthesis, having access to high-purity Related Substance C is essential for validating analytical methods and ensuring patient safety.
Q: Is this synthesis process scalable for industrial production of reference materials?
A: Yes, the process is designed for industrial scalability. It utilizes readily available bulk chemicals like epichlorohydrin and diethyl carbonate, operates at moderate temperatures (50-90°C), and employs standard workup procedures like extraction and filtration, eliminating the need for complex or hazardous specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-5-chloromethyl Oxazoline-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the quality of every single component, including the reference standards used to test them. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for early-stage R&D or full-scale QC operations. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3-amino-5-chloromethyl oxazoline-2-ketone meets the >99.9% purity benchmark established by the latest patent technologies. Our commitment to quality ensures that your analytical methods are validated against the most accurate standards available in the market.
We invite you to optimize your supply chain by partnering with us for the sourcing of this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific annual volume needs, demonstrating how our efficient synthesis route can lower your overall quality control costs. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project timelines are met with precision and reliability. Let us handle the complexity of synthesis so you can focus on delivering safe and effective medicines to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
