Advanced Ionic Liquid Catalysis for High-Purity 4,4'-Dinitrodiphenyl Ether Production
Advanced Ionic Liquid Catalysis for High-Purity 4,4'-Dinitrodiphenyl Ether Production
The global demand for high-performance polymers, particularly polyimides used in flexible electronics and aerospace applications, has necessitated a re-evaluation of precursor synthesis routes. Patent CN112724021A introduces a groundbreaking preparation method for 4,4'-dinitrodiphenyl ether, a critical intermediate for 4,4'-diaminodiphenyl ether. This technology leverages advanced ionic liquid catalysis to overcome the longstanding limitations of traditional etherification processes. By utilizing pyridinium or imidazolium salts supported on silica gel, the method achieves a remarkable yield of up to 98.8% while drastically minimizing environmental impact. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier or fine chemical partner, this patent represents a paradigm shift towards greener, more efficient manufacturing. The process not only ensures high purity but also addresses the critical supply chain need for sustainable production methodologies that reduce waste disposal costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,4'-dinitrodiphenyl ether has been plagued by significant technical and environmental hurdles. Traditional routes, such as the direct nitration of diphenyl ether, suffer from poor regioselectivity, yielding mixtures of 2,4-isomers alongside the desired 4,4'-product, which complicates purification and lowers overall yield. Alternative condensation methods involving p-dinitrobenzene generate hazardous nitrogen oxide byproducts, imposing stringent tail gas treatment requirements. Furthermore, the widely used condensation of p-nitrochlorobenzene with nitrophenol relies heavily on strong bases like sodium hydroxide. This necessitates subsequent acid neutralization, resulting in massive volumes of high-salinity wastewater that pose severe environmental discharge pressures. Other methods utilizing phosphorus phase transfer catalysts are consumable and non-recyclable, further inflating raw material costs and contributing to the accumulation of chemical waste.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally alters the reaction landscape by replacing consumable strong bases and phase transfer catalysts with recyclable ionic liquids. This novel approach utilizes p-nitrohalogenated benzene and an organic salt auxiliary, such as potassium acetate, within a solvent system comprising xylenes or dichlorobenzene. The key differentiator is the use of silica gel-supported ionic liquids, which act as highly efficient, recoverable catalysts. This structural design eliminates the generation of high-concentration salt wastewater and avoids the use of strong acids in post-treatment. Consequently, the process achieves superior atom utilization and simplifies the workflow, making it ideally suited for continuous flow line production and automatic control, thereby enhancing operational safety and consistency for large-scale manufacturing.
Mechanistic Insights into Ionic Liquid-Catalyzed Etherification
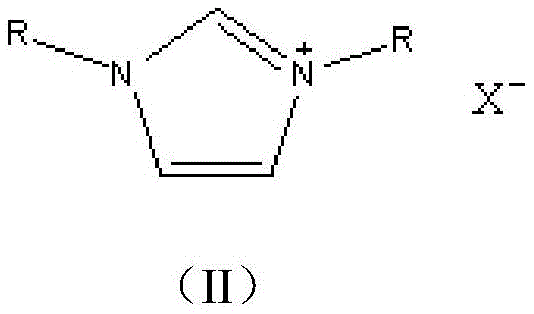
The core of this technological advancement lies in the specific catalytic activity of the ionic liquids, which function through a nucleophilic catalysis mechanism distinct from traditional base-promoted reactions. The cationic nature of the pyridinium or imidazolium salts stabilizes the transition state during the nucleophilic aromatic substitution, facilitating the displacement of the halogen atom on the p-nitrohalogenated benzene by the phenoxide species generated in situ. This stabilization lowers the activation energy required for the etherification, allowing the reaction to proceed efficiently at moderate temperatures between 90°C and 140°C. The choice of anion, ranging from chloride to trifluoroacetate, fine-tunes the Lewis acidity and solubility properties of the catalyst, optimizing the interaction between the organic substrates and the catalytic active sites.
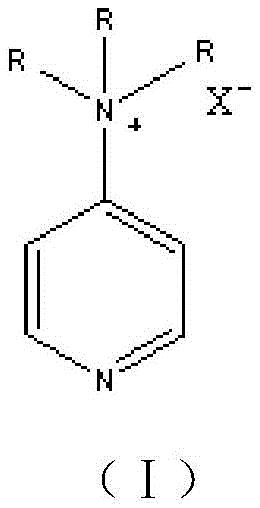
To further enhance performance and address the issue of high viscosity common in pure ionic liquids, the patent describes supporting these catalysts on a silica gel matrix. This heterogenization strategy creates a pseudo-homogeneous environment that combines the high activity of homogeneous catalysis with the ease of separation associated with heterogeneous systems. The silica support prevents the aggregation of ionic liquid molecules, ensuring maximum exposure of active sites to the reactants. Additionally, the support structure aids in the physical separation of the catalyst from the product mixture, allowing for effective regeneration through simple alcohol washing and distillation. This mechanism ensures that even after multiple cycles, the catalyst maintains a selectivity exceeding 96%, proving its robustness for industrial application.
How to Synthesize 4,4'-Dinitrodiphenyl Ether Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this high-efficiency route in a commercial setting. The process begins with the meticulous pretreatment of the ionic liquid catalyst, where it is loaded onto silica gel via a sol-gel process involving silicate esters and alcohols, followed by acidification and drying to activate the surface. This preparation step is crucial for ensuring the long-term stability and reusability of the catalyst system. Once prepared, the catalyst is introduced to the reaction vessel containing the p-nitrohalogenated benzene substrate and the organic salt auxiliary in a suitable aromatic solvent. The reaction proceeds under mild pressure and temperature conditions, significantly reducing energy consumption compared to high-temperature fusion methods.
- Prepare the silica gel-supported ionic liquid catalyst by mixing silicate ester, alcohol, and ionic liquid, followed by acidification and drying.
- Conduct the etherification reaction by mixing p-nitrohalogenated benzene, organic salt auxiliary, and solvent under the catalysis of the prepared ionic liquid at 90-140°C.
- Separate the mixture, regenerate the lower ionic liquid layer via washing and distillation, and evaporate the upper layer to crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid technology translates into tangible strategic benefits beyond mere chemical yield. The elimination of consumable strong bases and phase transfer catalysts directly correlates to a substantial reduction in raw material procurement costs. By removing the need for expensive heavy metal catalysts or stoichiometric amounts of base, the variable cost per kilogram of the final product is significantly optimized. Furthermore, the drastic reduction in wastewater salinity and volume alleviates the burden on effluent treatment plants, leading to lower operational expenditures related to environmental compliance and waste disposal fees. This streamlined waste profile enhances the overall sustainability rating of the supply chain, a critical metric for modern multinational corporations.
- Cost Reduction in Manufacturing: The ability to recycle the ionic liquid catalyst multiple times without significant loss of activity means that the catalyst cost is amortized over many batches, effectively rendering it a fixed asset rather than a recurring consumable expense. This contrasts sharply with traditional methods where catalysts are lost in the aqueous waste stream. Additionally, the simplified post-treatment process, which avoids complex neutralization and salt removal steps, reduces utility consumption and labor hours, driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the silica-supported catalyst ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by off-spec material. The use of common, commercially available solvents like xylenes and toluene ensures that raw material sourcing remains stable and unaffected by niche supply constraints. This reliability is paramount for maintaining continuous production schedules for downstream polyimide manufacturers who depend on a steady flow of high-purity intermediates to meet their own delivery commitments.
- Scalability and Environmental Compliance: The process is explicitly designed for scalability, with reaction conditions that are easily manageable in large-scale reactors without requiring exotic high-pressure equipment. The significant reduction in hazardous byproducts and saline waste aligns perfectly with increasingly stringent global environmental regulations. This future-proofs the manufacturing site against potential regulatory crackdowns and allows for seamless expansion of capacity to meet growing market demand for electronic and aerospace materials without incurring prohibitive environmental upgrade costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalysis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this novel synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing production lines or new facility designs.
Q: What are the primary advantages of using ionic liquids over traditional base catalysts for this synthesis?
A: Unlike traditional methods that consume strong bases and generate high-salt wastewater requiring neutralization, the ionic liquid method allows for catalyst recycling. This significantly reduces three wastes and eliminates the need for consumable phase transfer catalysts, leading to higher atom utilization.
Q: How does the silica gel support improve the process efficiency?
A: Pure ionic liquids often have high viscosity, making product separation difficult. Supporting the ionic liquid on silica gel facilitates easier separation from the reaction mixture. Furthermore, the supported catalyst can be regenerated and reused multiple times while maintaining high selectivity above 96%.
Q: What is the expected purity and yield of the final 4,4'-dinitrodiphenyl ether product?
A: The patented process achieves a maximum yield of up to 98.8%. The final product purity is consistently maintained at or above 98%, with elemental analysis confirming high quality suitable for downstream polymerization into polyimides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dinitrodiphenyl Ether Supplier
As the industry moves towards more sustainable and efficient chemical manufacturing, NINGBO INNO PHARMCHEM stands at the forefront of translating patented innovations into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial output is seamless and risk-mitigated. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch of 4,4'-dinitrodiphenyl ether meets the exacting standards required for high-performance polyimide synthesis. We understand that consistency is key in the fine chemical sector, and our advanced process control systems ensure that the benefits of the ionic liquid method are fully realized in every shipment.
We invite forward-thinking partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance metrics. Together, we can drive down costs and elevate the quality standards of the global fine chemical market.
