Scalable Green Synthesis of Azelaic Acid via Dual-Catalytic Oleic Acid Oxidation
The global demand for high-purity dicarboxylic acids, particularly azelaic acid, has surged due to its critical applications in dermatological pharmaceuticals, high-performance polyamides, and plasticizers. Patent CN1746145A introduces a transformative methodology for the clean oxidation of oleic acid, addressing the longstanding inefficiencies of traditional stoichiometric oxidants. This technology leverages a sophisticated dual-catalytic system, initially employing a tungsten-based catalyst for epoxidation, followed by a cobalt-halide mediated oxidative cleavage. By shifting from hazardous reagents like potassium permanganate or ozone to a green hydrogen peroxide and oxygen system, this process not only aligns with modern environmental compliance standards but also drastically enhances economic viability. For R&D directors and procurement strategists, this patent represents a pivotal opportunity to secure a supply chain for azelaic acid that is both cost-effective and chemically robust, eliminating the bottlenecks associated with low-yield batch processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of azelaic acid has been plagued by severe technical and economic drawbacks inherent to older oxidation technologies. Traditional methods utilizing potassium permanganate as the oxidant suffer from abysmal atom economy, typically yielding only 25% to 45% of the desired product while generating massive quantities of manganese dioxide sludge that requires costly disposal. Alternatively, ozonolysis, while chemically effective, introduces significant safety hazards due to the explosive nature of ozone and the requirement for cryogenic conditions, which escalates energy consumption and capital expenditure. Furthermore, prior art involving high-pressure oxidation, such as the Sabarino Giampiero method, necessitates extreme pressures up to 70 kg/cm², demanding specialized high-grade reactor vessels that limit scalability and increase maintenance overheads. These conventional routes also struggle with selectivity, often producing a complex mixture of shorter-chain dicarboxylic acids (C5-C8) that are difficult and expensive to separate from the target C9 diacid, thereby compromising the purity required for sensitive pharmaceutical applications.
The Novel Approach
The methodology disclosed in CN1746145A fundamentally reengineers the oxidation pathway by decoupling the reaction into two distinct, optimized stages under mild conditions. Instead of relying on a single harsh oxidant, the process initiates with a controlled epoxidation and hydroxylation of the oleic acid double bond using hydrogen peroxide and a tungstic acid catalyst in a tert-butanol solvent system. This is immediately followed by an oxidative cleavage step driven by molecular oxygen and a lipophilic cobalt catalyst in the presence of a bromide promoter. This synergistic approach allows the reaction to proceed at moderate temperatures (60-95°C) and relatively low oxygen pressures (0.5-1.5 MPa), making it compatible with standard stainless steel industrial reactors. Crucially, this route tolerates crude oleic acid feedstocks with acid values between 190 and 205, removing the need for expensive feedstock purification and enabling the direct conversion of bio-based renewable resources into high-value fine chemicals with minimal waste generation.
Mechanistic Insights into Tungsten-Cobalt Dual Catalytic Oxidation
The chemical elegance of this process lies in the precise orchestration of two different catalytic cycles that maximize bond cleavage efficiency while minimizing over-oxidation. In the first stage, the tungsten catalyst activates hydrogen peroxide to form peroxotungsten species, which transfer oxygen to the olefinic bond of oleic acid, generating a mixture of epoxides and vicinal diols. This activation is critical as it primes the carbon-carbon double bond for subsequent scission without causing random chain degradation. The reaction environment, maintained by tert-butanol, ensures that the polar intermediates remain soluble, facilitating homogeneous catalysis and preventing the formation of tarry byproducts that often foul reactors in heterogeneous systems. This controlled functionalization sets the stage for the highly selective cleavage that follows, ensuring that the carbon backbone is broken specifically at the C9-C10 position.
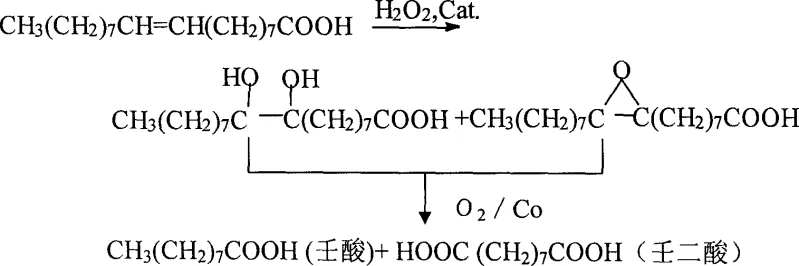
In the second stage, the introduction of the cobalt salt and ammonium bromide triggers a radical-mediated oxidative cleavage mechanism. The cobalt species, likely cycling between Co(II) and Co(III) oxidation states, decomposes hydroperoxides formed in situ to generate alkoxy and peroxy radicals. These radicals attack the activated C-C bond of the diol/epoxide intermediates, leading to a clean scission that yields nonanoic acid and azelaic acid. The presence of the bromide ion acts as a crucial co-catalyst, accelerating the radical generation rate and allowing the reaction to reach completion within 6 to 12 hours, a significant improvement over the 24 to 70 hours required by previous methods. This mechanistic precision results in a product profile dominated by the target C9 diacid, with negligible formation of suberic acid (C8) or shorter chain impurities, thereby simplifying downstream purification and ensuring high spectral purity for end-use applications.
How to Synthesize Azelaic Acid Efficiently
Implementing this synthesis route requires careful control of the transition between the epoxidation and cleavage phases to maintain catalyst activity and selectivity. The process begins with the preparation of the reaction mixture in a stirred vessel, where crude oleic acid is dissolved in tert-butanol and treated with tungstic acid. Hydrogen peroxide is added gradually to manage the exotherm, ensuring the temperature remains within the optimal 60-80°C window to form the oxygenated intermediates. Once the epoxidation is complete, the system is charged with the cobalt catalyst and bromide promoter, and oxygen pressure is applied to drive the cleavage reaction to high conversion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below for technical reference.
- Epoxidation/Hydroxylation: React crude oleic acid with hydrogen peroxide (50-80%) and tungstic acid catalyst in tert-butanol solvent at 60-80°C to form hydroxy/epoxy intermediates.
- Oxidative Cleavage: Add cobalt salt (e.g., cobalt octoate) and ammonium bromide to the mixture. Introduce oxygen pressure (0.5-1.5 MPa) and heat to 90-95°C for 6-12 hours to cleave the double bond.
- Workup and Purification: Separate and recover the catalyst, remove solvent via vacuum distillation, and purify the resulting nonanoic and azelaic acid mixture through crystallization or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers profound strategic advantages beyond mere chemical yield. The ability to utilize crude oleic acid, a widely available and price-stable bio-based commodity, insulates the manufacturing process from the volatility associated with refined chemical feedstocks. This flexibility in raw material specification allows for significant cost reduction in azelaic acid manufacturing, as the expense of pre-purifying fatty acids is entirely eliminated. Furthermore, the use of molecular oxygen and hydrogen peroxide as terminal oxidants replaces expensive stoichiometric reagents, drastically reducing the variable cost per kilogram of product. The mild reaction conditions also translate to lower energy consumption for heating and pressurization, contributing to a leaner operational cost structure that enhances competitiveness in the global fine chemicals market.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal oxidants like permanganate and the ability to recycle the tungsten and cobalt catalysts create a closed-loop system that minimizes raw material waste. By avoiding the generation of solid manganese sludge, the facility saves substantially on hazardous waste disposal fees and environmental compliance costs. Additionally, the high selectivity of the reaction reduces the burden on downstream purification units, such as distillation columns and crystallizers, leading to lower utility consumption and higher throughput capacity without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Sourcing crude oleic acid is inherently more stable than relying on specialized reagents, as it is a primary derivative of the global vegetable oil industry. This abundance ensures a continuous supply of feedstock, mitigating the risk of production stoppages due to material shortages. The robustness of the catalyst system, which tolerates impurities in the feedstock, further enhances supply continuity by reducing the frequency of batch failures caused by feedstock variability. This reliability is critical for maintaining long-term contracts with pharmaceutical and polymer clients who require consistent delivery schedules and uninterrupted material flow.
- Scalability and Environmental Compliance: The process operates at near-atmospheric to low pressures (0.5-1.5 MPa) and moderate temperatures, allowing for seamless scale-up using standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. This compatibility eliminates the need for custom-built high-pressure vessels, accelerating the timeline from pilot plant to commercial production. Moreover, the green nature of the oxidants (water and oxygen as byproducts) aligns with increasingly stringent environmental regulations, future-proofing the manufacturing site against regulatory tightening and enhancing the corporate sustainability profile for eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this oxidation technology. These answers are derived directly from the experimental data and process parameters detailed in the patent literature, providing a factual basis for feasibility assessments. Understanding these nuances is essential for engineering teams evaluating the integration of this route into existing production lines.
Q: Can crude oleic acid be used directly in this synthesis process?
A: Yes, the patent specifically highlights the ability to use crude oleic acid (acid value 190-205, iodine value 90-110) without extensive pre-purification, which significantly lowers raw material costs compared to methods requiring high-purity feedstocks.
Q: How does this method improve selectivity compared to traditional permanganate oxidation?
A: Traditional permanganate oxidation often yields only 25-45% azelaic acid with significant over-oxidation. This dual-catalytic method achieves over 88% yield and 97% conversion by controlling the radical cleavage step, minimizing the formation of shorter-chain dicarboxylic acid impurities (C5-C8).
Q: Is the catalyst system recyclable for industrial scale-up?
A: The process design allows for the separation and recovery of the tungsten and cobalt catalysts after the reaction. This recyclability reduces heavy metal waste discharge and lowers the overall operational expenditure (OPEX) for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azelaic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthesis routes for high-value intermediates like azelaic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and risk-mitigated. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of short-chain impurities and heavy metal residues. Our infrastructure is designed to support the complex catalytic requirements of modern green chemistry, positioning us as a strategic partner for companies seeking to optimize their supply chains for pharmaceutical and specialty polymer applications.
We invite you to collaborate with us to leverage this advanced oxidation technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this process can enhance your margin structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of azelaic acid for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
