Industrial Synthesis of Diaminomethyl Cyclobutane Oxalate for Stable Lobaplatin Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex antineoplastic agents, particularly platinum-based compounds like Lobaplatin, which is critical for treating breast cancer, small cell lung cancer, and chronic myelocytic leukemia. A pivotal breakthrough in this domain is detailed in patent CN102093226B, which outlines a novel preparation method for diaminomethyl cyclobutane oxalate. This compound serves as a vital intermediate in the synthesis of Lobaplatin, addressing significant historical challenges regarding the stability and handling of precursor materials. Traditional methods often relied on unstable liquid intermediates that complicated the supply chain and reduced overall process efficiency. By contrast, this patented methodology transforms volatile liquid amines into stable solid oxalate salts, thereby enhancing the reproducibility and purity of the final anticancer drug. The technical innovation lies not only in the chemical transformation but also in the strategic selection of catalysts and reaction conditions that favor industrial scalability while maintaining rigorous quality standards suitable for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lobaplatin relied heavily on intermediates such as trans-diaminomethyl tetramethylene in its free base form. This substance exists as a liquid at ambient conditions and possesses inherently unstable chemical properties. This instability creates a cascade of logistical and technical difficulties for pharmaceutical manufacturers. Firstly, the liquid nature of the intermediate makes long-term storage problematic, as it is prone to degradation or polymerization over time, leading to batch-to-batch variability. Secondly, transporting liquid amines requires specialized containment and safety protocols due to their volatility and potential reactivity, which inflates logistics costs and introduces supply chain risks. Furthermore, the instability of the free amine directly impacts the subsequent coupling reactions with platinum precursors. Uncontrolled side reactions can occur, resulting in lower yields of the final Lobaplatin product and the formation of difficult-to-remove impurities. These factors collectively limit the commercial viability of older synthetic routes, making them less attractive for large-scale Good Manufacturing Practice (GMP) production environments where consistency is paramount.
The Novel Approach
The methodology described in patent CN102093226B offers a transformative solution by converting the unstable liquid amine into diaminomethyl cyclobutane oxalate, a stable crystalline solid. This approach fundamentally alters the physical state of the intermediate, rendering it far more manageable for industrial operations. The process begins with the dimerization of acrylonitrile, a readily available and cost-effective bulk chemical, rather than relying on expensive or hard-to-source specialty starting materials. Through a carefully controlled catalytic sequence involving iron(III) chloride and active nickel, the synthesis achieves high selectivity for the trans-isomer, which is the required stereochemistry for biological activity. The final conversion to the oxalate salt locks the molecule into a stable lattice structure, eliminating the volatility issues associated with the free amine. This solid form allows for straightforward filtration, drying, and packaging, significantly reducing the risk of contamination during handling. Consequently, this novel route not only improves the chemical purity of the intermediate but also streamlines the entire manufacturing workflow, making it highly suitable for the demanding requirements of modern oncology drug production.
Mechanistic Insights into FeCl3-Catalyzed Dimerization and Nickel Hydrogenation
The core of this synthetic strategy relies on two distinct catalytic events that drive the transformation from simple nitriles to the complex cyclobutane diamine structure. The initial step involves the thermal dimerization of acrylonitrile. In this reaction, two molecules of acrylonitrile undergo a [2+2] cycloaddition to form a cyclobutane ring. This process is catalyzed by iron(III) chloride (FeCl3) in the presence of p-methoxyphenol, which acts as a polymerization inhibitor to prevent the formation of polyacrylonitrile chains. The reaction is conducted under nitrogen pressure at elevated temperatures ranging from 200°C to 235°C. Under these conditions, a mixture of cis- and trans-1,2-dicyanocyclobutane is generated, typically in a ratio of approximately 40:60. The presence of the Lewis acid catalyst facilitates the alignment of the double bonds, lowering the activation energy for the cycloaddition while the inhibitor ensures that the reaction stops at the dimer stage rather than proceeding to oligomers.

Following the dimerization, the crude mixture undergoes distillation and rectification. This physical separation step is crucial as it isolates the desired trans-isomer while simultaneously inducing isomerization of the cis-form, thereby maximizing the yield of the trans-dinitrile cyclobutane. The second major mechanistic phase is the catalytic hydrogenation of the nitrile groups. Using active nickel as a heterogeneous catalyst, the trans-dinitrile compound is subjected to hydrogen gas in the presence of ammonia at pressures around 20bar and temperatures between 70°C and 85°C. The ammonia serves a dual purpose: it acts as a solvent and suppresses the formation of secondary and tertiary amines by competing for the catalyst surface, ensuring the selective formation of primary amines. This reduction converts the two cyano groups (-CN) into aminomethyl groups (-CH2NH2), yielding trans-diaminomethyl cyclobutane. The precision of this hydrogenation step is vital, as over-reduction or incomplete reaction would compromise the purity of the final oxalate salt.
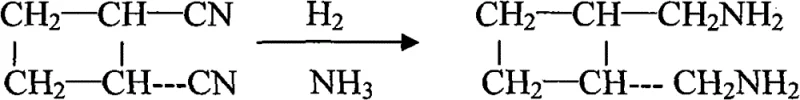
Impurity control is rigorously maintained throughout this mechanism. The use of p-methoxyphenol in the first step effectively curbs polymer formation, which is a common side reaction in acrylonitrile chemistry. In the hydrogenation step, the addition of ammonia is a classic technique to minimize the formation of dibenzylamine-type byproducts, which often plague nitrile reductions. Finally, the conversion to the oxalate salt acts as a purification checkpoint. Since the oxalate salt has specific solubility characteristics, recrystallization from a methanol-water mixture allows for the exclusion of non-basic impurities and residual catalysts. The patent data indicates that this multi-stage purification results in a final product with an oxalate content of ≥96%, demonstrating the efficacy of the mechanistic design in delivering high-purity material suitable for sensitive pharmaceutical syntheses.
How to Synthesize Diaminomethyl Cyclobutane Oxalate Efficiently
The synthesis of this critical intermediate requires precise control over reaction parameters to ensure safety and yield. The process integrates high-pressure hydrogenation and high-temperature dimerization, necessitating specialized reactor equipment capable of withstanding significant thermal and pressure loads. Operators must strictly adhere to the temperature gradients defined in the patent, particularly during the distillation phase where the separation of cis and trans isomers occurs. The subsequent salt formation step is exothermic and requires careful addition of oxalic acid to control precipitation kinetics. For a comprehensive understanding of the operational parameters, including specific molar ratios and cooling rates, please refer to the standardized synthesis guide below.
- Perform dimerization of acrylonitrile using FeCl3 catalyst and p-methoxyphenol inhibitor at 200-235°C under nitrogen pressure to obtain a cis/trans dinitrile mixture.
- Distill and rectify the mixture at 140-180°C to isolate trans-dinitrile cyclobutane through isomerization.
- Reduce the trans-dinitrile compound using active nickel catalyst with ammonia and hydrogen at 70-85°C and 20bar pressure to form trans-diaminomethyl cyclobutane.
- React the resulting amine with anhydrous oxalic acid to precipitate the crude oxalate salt, followed by recrystallization in methanol/water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from liquid amine intermediates to solid oxalate salts represents a significant optimization of the value chain. The primary advantage lies in the drastic simplification of logistics and storage protocols. Liquid amines often require inert atmosphere storage and temperature-controlled transportation to prevent degradation, which incurs substantial costs and limits the pool of eligible logistics providers. By stabilizing the molecule as a solid oxalate, the material becomes shelf-stable at ambient conditions, allowing for standard warehousing and bulk shipping methods. This transition effectively de-risks the supply chain, ensuring that production schedules are not disrupted by the spoilage of sensitive intermediates. Furthermore, the use of acrylonitrile as a starting material leverages a commodity chemical market, insulating the production cost from the volatility often seen in niche fine chemical sectors.
- Cost Reduction in Manufacturing: The elimination of complex stabilization procedures for liquid intermediates translates directly into operational expenditure savings. Traditional methods often require immediate downstream processing of unstable liquids to prevent loss, forcing a continuous, rigid production schedule. The ability to produce, store, and quality-test the solid oxalate intermediate decouples the upstream synthesis from the final drug formulation, allowing for more flexible batch planning and inventory management. Additionally, the high selectivity of the nickel-catalyzed hydrogenation reduces the burden on downstream purification units, lowering solvent consumption and waste treatment costs associated with removing complex amine byproducts.
- Enhanced Supply Chain Reliability: The robustness of the solid intermediate ensures consistent availability for downstream API manufacturers. In the event of a disruption in the final Lobaplatin synthesis line, the stable intermediate can be stockpiled without significant degradation, acting as a buffer against supply shocks. This reliability is critical for pharmaceutical companies managing just-in-time manufacturing models. Moreover, the accessibility of raw materials like acrylonitrile and common catalysts like iron and nickel ensures that the supply of the intermediate is not bottlenecked by the availability of exotic reagents, further securing the continuity of supply for global markets.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard unit operations such as autoclaves and distillation columns. The avoidance of hazardous liquid amines reduces the environmental footprint related to volatile organic compound (VOC) emissions during handling. The solid waste generated, primarily consisting of spent catalysts and oxalate mother liquors, is generally easier to treat and dispose of compared to the complex mixtures resulting from unstable liquid amine degradations. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of diaminomethyl cyclobutane oxalate. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a factual basis for evaluating the technology's potential in your supply chain.
Q: Why is the oxalate salt form preferred over the free amine for Lobaplatin synthesis?
A: The free amine (trans-diaminomethyl tetramethylene) is a liquid with unstable properties that complicate storage and transport. Converting it to the oxalate salt creates a stable solid at normal temperature and pressure, significantly improving handling safety and shelf-life for downstream pharmaceutical manufacturing.
Q: What represents the critical purity specifications for this intermediate?
A: According to the patented process, the final diaminomethyl cyclobutane oxalate crystal achieves an oxalate content of greater than or equal to 96% with a total yield exceeding 70%, ensuring high quality for the subsequent synthesis of the platinum-based antineoplastic agent.
Q: How does the catalytic system ensure process scalability?
A: The process utilizes accessible catalysts like iron(III) chloride and active nickel, along with common gases like hydrogen and ammonia. The operation conditions, such as pressures around 17-20bar and temperatures up to 235°C, are well within the capabilities of standard industrial chemical reactors, facilitating large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaminomethyl Cyclobutane Oxalate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful manufacture of life-saving oncology drugs. Our technical team has extensively analyzed the synthetic pathways for Lobaplatin precursors and possesses the expertise to replicate and optimize the patented processes described in CN102093226B. We understand that transitioning to a new intermediate source requires absolute confidence in quality and consistency. Therefore, our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of diaminomethyl cyclobutane oxalate meets the exacting standards required for GMP pharmaceutical production. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to support your long-term manufacturing needs.
We invite you to collaborate with us to optimize your supply chain for Lobaplatin production. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to a reliable source of stable, high-purity intermediates that can streamline your operations and reduce overall production costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can add value to your pharmaceutical portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
