Scalable Synthesis of Diaminomethyl Cyclobutane Oxalate for High-Purity Lobaplatin Production
Scalable Synthesis of Diaminomethyl Cyclobutane Oxalate for High-Purity Lobaplatin Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical antineoplastic agents, particularly platinum-based complexes like Lobaplatin. A pivotal advancement in this domain is detailed in patent CN102093226A, which outlines a novel preparation method for diaminomethyl cyclobutane oxalate. This intermediate is essential for the synthesis of Lobaplatin, a third-generation platinum anticancer drug known for its efficacy against breast cancer, small cell lung cancer, and chronic myelocytic leukemia. The traditional reliance on unstable liquid intermediates has long plagued manufacturers with issues regarding storage, transport, and batch-to-batch consistency. This new methodology addresses these challenges by converting the reactive trans-diaminomethyl tetramethylene directly into a stable oxalate salt, thereby enhancing the overall integrity of the supply chain for oncology therapeutics.
For R&D directors and process chemists, the significance of this patent lies in its ability to transform a volatile liquid intermediate into a crystalline solid without compromising yield or purity. The process leverages accessible raw materials like acrylonitrile and utilizes standard industrial catalytic systems, making it highly attractive for commercial scale-up. By stabilizing the core cyclobutane structure early in the synthesis, manufacturers can mitigate the risks associated with the degradation of the diamine precursor. This approach not only streamlines the production workflow but also ensures that the final Lobaplatin product meets stringent pharmacopeial standards for impurity profiles and stability, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lobaplatin has relied heavily on the use of trans-diaminomethyl tetramethylene as a key building block. However, this compound exists as a liquid at room temperature and exhibits inherent instability, which poses significant logistical and chemical challenges. The liquid nature of the free base makes it prone to oxidation and degradation during storage and transportation, often requiring specialized containment and cold chain logistics that drive up operational costs. Furthermore, the instability of this liquid intermediate directly influences the reaction kinetics and purity of the subsequent platinum coordination step. Variations in the quality of the liquid diamine can lead to incomplete reactions, the formation of difficult-to-remove by-products, and ultimately, a lower yield of the final active pharmaceutical ingredient. These factors collectively create a bottleneck in cost reduction in API manufacturing, as extensive purification steps become necessary to compensate for the variability of the starting material.
The Novel Approach
The innovative strategy presented in the patent data circumvents these issues by immediately converting the synthesized trans-diaminomethyl tetramethylene into its oxalate salt form. This transformation changes the physical state of the intermediate from a volatile liquid to a stable solid at normal temperature and pressure. The solid oxalate salt is far easier to handle, store, and transport, eliminating the need for complex stabilization protocols. From a chemical perspective, the salt form acts as a protected version of the diamine, preventing unwanted side reactions until it is intentionally released during the final synthesis of Lobaplatin. This shift not only simplifies the inventory management for procurement managers but also enhances the reproducibility of the downstream synthesis. By ensuring a consistent and high-purity feedstock, the novel approach facilitates the commercial scale-up of complex pharmaceutical intermediates, offering a distinct competitive advantage in the global market for anticancer drugs.

Mechanistic Insights into FeCl3-Catalyzed Dimerization and Nickel Reduction
The core of this synthetic route begins with the dimerization of acrylonitrile, a reaction that is meticulously controlled to favor the formation of the cyclobutane ring structure. In the presence of an iron(III) chloride (FeCl3) catalyst and p-methoxyphenol as a polymerization inhibitor, acrylonitrile undergoes a [2+2] cycloaddition under elevated temperature and pressure conditions. The reaction is typically conducted at temperatures ranging from 200°C to 235°C under a nitrogen pressure of approximately 17 bar. This specific catalytic environment is crucial for suppressing the formation of linear polymers, which are common side products in acrylonitrile chemistry. The result is a dark red liquid mixture containing both cis- and trans-dinitrile cyclobutane isomers, typically in a ratio of roughly 4:6. The use of FeCl3 is particularly advantageous due to its availability and effectiveness in promoting this specific cyclization pathway without introducing expensive noble metals into the process.
Following the isolation of the trans-dinitrile cyclobutane isomer through distillation and rectification, the nitrile groups are reduced to primary amines using a heterogeneous catalytic hydrogenation process. Active nickel serves as the catalyst in this step, operating under an ammonia atmosphere and hydrogen pressure of around 20 bar at temperatures between 70°C and 85°C. The presence of ammonia is critical as it helps to suppress the formation of secondary and tertiary amines, ensuring high selectivity for the desired primary diamine. Once the trans-diaminomethyl cyclobutane is formed, it is immediately reacted with anhydrous oxalic acid. This acid-base reaction precipitates the diaminomethyl cyclobutane oxalate as a solid crude product. The final purification involves recrystallization from a methanol and water mixture, yielding a product with an oxalate content of greater than 96% and an overall yield exceeding 70%. This rigorous control over reaction parameters ensures minimal impurity generation, which is vital for reducing lead time for high-purity pharmaceutical intermediates.
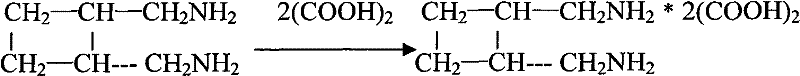
How to Synthesize Diaminomethyl Cyclobutane Oxalate Efficiently
The synthesis protocol described in the patent offers a clear pathway for manufacturing this critical intermediate with high efficiency and safety. The process is designed to be scalable, utilizing standard autoclave equipment for the high-pressure steps and conventional distillation columns for purification. The key to success lies in the precise control of temperature and pressure during the dimerization and reduction phases, as well as the careful management of the crystallization process to ensure maximum recovery of the oxalate salt. Operators must adhere strictly to the specified molar ratios of catalysts and inhibitors to prevent runaway polymerization or the formation of undesired isomers. The following guide outlines the standardized operational procedure derived from the patent examples, providing a foundational framework for process engineers looking to implement this technology.
- Perform dimerization of acrylonitrile using FeCl3 catalyst and p-methoxyphenol inhibitor at 200-235°C under nitrogen pressure to form a cis/trans-dinitrile cyclobutane mixture.
- Distill and rectify the mixture at 140-180°C to isolate trans-dinitrile cyclobutane through counter-rotating isomerization.
- Reduce the trans-dinitrile compound using active nickel catalyst with ammonia and hydrogen at 20bar pressure to obtain trans-diaminomethyl cyclobutane.
- React the resulting diamine with anhydrous oxalic acid to precipitate the crude oxalate salt, followed by recrystallization in methanol and water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this oxalate-based intermediate represents a significant opportunity to optimize operational expenditures and mitigate risk. The primary advantage stems from the physical stability of the product; unlike its liquid predecessor, the solid oxalate salt does not require specialized cold chain logistics or inert atmosphere storage during transit. This simplification of logistics translates directly into substantial cost savings in transportation and warehousing. Furthermore, the extended shelf life of the solid intermediate reduces the risk of inventory write-offs due to degradation, allowing for more flexible purchasing strategies and larger batch orders that leverage economies of scale. The robustness of the synthesis also means that supply continuity is less likely to be disrupted by batch failures, ensuring a steady flow of materials for downstream API production.
- Cost Reduction in Manufacturing: The elimination of complex stabilization procedures for the liquid diamine significantly lowers processing costs. By avoiding the need for immediate conversion or specialized storage of the unstable free base, manufacturers can streamline their facility usage and reduce energy consumption associated with climate-controlled storage. Additionally, the high selectivity of the active nickel reduction step minimizes the formation of hard-to-separate impurities, which reduces the burden on downstream purification units and lowers solvent consumption. These cumulative efficiencies contribute to a leaner manufacturing process that enhances overall profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like acrylonitrile and oxalic acid as starting materials ensures that the supply chain is not dependent on exotic or scarce reagents. This accessibility reduces the risk of raw material shortages and price volatility. Moreover, the solid nature of the final intermediate allows for safer and more straightforward handling during loading and unloading operations, reducing the potential for accidents and delays. This reliability is crucial for maintaining just-in-time delivery schedules and meeting the rigorous demands of global pharmaceutical clients who require consistent quality and timely shipments.
- Scalability and Environmental Compliance: The process conditions, such as the use of nitrogen and hydrogen pressure, are well within the capabilities of standard industrial chemical reactors, facilitating easy scale-up from pilot plant to commercial production volumes. The absence of heavy metal catalysts in the final product, thanks to the use of iron and nickel which are easier to remove or are less toxic than precious metals, simplifies waste treatment and regulatory compliance. The crystallization step using methanol and water allows for efficient solvent recovery and recycling, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of diaminomethyl cyclobutane oxalate. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains or development pipelines.
Q: Why is the oxalate salt form preferred over the free base for Lobaplatin synthesis?
A: The free base trans-diaminomethyl tetramethylene is a liquid with unstable properties that complicate storage and transport. Converting it immediately into the diaminomethyl cyclobutane oxalate salt creates a stable solid at normal temperature and pressure, significantly improving handling safety and ensuring consistent purity for downstream platinum coordination.
Q: What catalysts are utilized in the dimerization and reduction steps?
A: The process employs iron(III) chloride (FeCl3) combined with p-methoxyphenol as a polymerization inhibitor for the initial acrylonitrile dimerization. Subsequently, active nickel is used as the heterogeneous catalyst for the high-pressure hydrogenation step to reduce the nitrile groups to amines.
Q: How does this method impact the purity of the final Lobaplatin API?
A: By stabilizing the intermediate as a crystalline oxalate salt, the method minimizes degradation and impurity formation during storage. This results in a more consistent feedstock for the platinum complexation reaction, leading to Lobaplatin with lower total impurities and improved stability profiles compared to methods using the unstable liquid intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaminomethyl Cyclobutane Oxalate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology treatments. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial materials and full-scale commercial manufacturing. We are committed to delivering diaminomethyl cyclobutane oxalate with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to replicate the advanced FeCl3-catalyzed dimerization and nickel reduction processes allows us to offer a product that consistently supports the synthesis of stable and effective Lobaplatin.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project goals. Let us be your partner in bringing high-quality anticancer therapies to the market faster and more efficiently.
