Advanced Isothermal Purification Technology for High-Purity Esomeprazole Sodium Manufacturing
The pharmaceutical industry continuously demands higher purity standards for critical active pharmaceutical ingredients (APIs) and their intermediates, particularly for chiral drugs where optical purity dictates therapeutic efficacy. Patent CN112661744A introduces a groundbreaking purification methodology for Esomeprazole Sodium, the S-isomer of omeprazole, which serves as a potent proton pump inhibitor widely used in treating gastroesophageal reflux disease. This novel technical approach addresses the persistent challenges of chiral separation and thermal degradation that have plagued traditional manufacturing routes. By leveraging a sophisticated mixed solvent system comprising both protic and aprotic components, the inventors have achieved a refined product with exceptional chemical purity exceeding 99.5% and an R-enantiomer content of less than 0.01%. This level of control is paramount for regulatory compliance and patient safety in global markets.
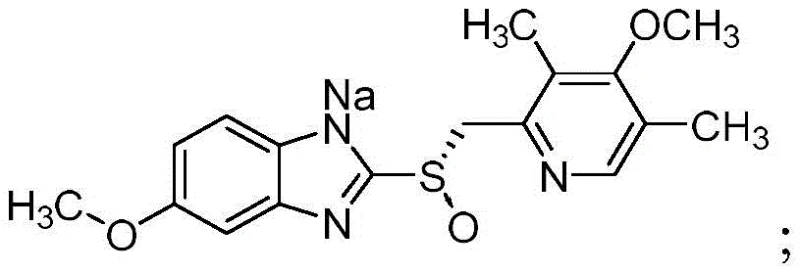
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere chemical specifications. The process eliminates the need for complex thermal cycling—heating for dissolution and cooling for crystallization—which traditionally introduces significant energy costs and operational risks. Instead, the entire purification occurs under isothermal conditions between 0°C and 30°C, drastically simplifying the equipment requirements and reducing the potential for batch-to-batch variability. This stability translates directly into a more reliable esomeprazole sodium supplier capability, ensuring consistent quality delivery even during large-scale commercial production runs where thermal gradients can be difficult to manage uniformly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Esomeprazole Sodium has been fraught with technical inefficiencies and safety concerns that hinder cost-effective manufacturing. Prior art methods, such as those disclosed in WO 94/027988, relied heavily on toluene, a solvent now recognized for its high toxicity and stringent environmental regulations, making it increasingly undesirable for modern green chemistry initiatives. Other approaches utilizing acetone or acetone-water mixtures often suffered from poor impurity removal efficiency, failing to reduce single impurity levels below the critical 0.1% threshold required for high-grade APIs. Furthermore, methods involving chiral chromatography, while effective, are prohibitively expensive and difficult to scale for multi-ton production, rendering them unsuitable for cost reduction in pharmaceutical intermediate manufacturing. Perhaps most critically, traditional recrystallization techniques requiring heating followed by cooling pose a severe risk of product degradation, leading to the formation of sulfone impurities and compromising the optical purity of the final drug substance.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the crystallization landscape by exploiting the differential solubility profiles of Esomeprazole Sodium in specific solvent blends. Rather than relying on temperature swings to drive supersaturation, this method dissolves the crude material in a precise mixture of a protic solvent, such as isopropanol, and an aprotic solvent, like butyl acetate or tetrahydrofuran. Once dissolved, the addition of further aprotic solvent acts as an anti-solvent, inducing rapid and controlled crystallization without any change in temperature. This isothermal mechanism not only preserves the structural integrity of the sensitive sulfinyl bond but also ensures that impurities remain solvated in the mother liquor. The result is a robust, scalable process that delivers high yields greater than 85% while maintaining the stringent purity specifications demanded by top-tier pharmaceutical companies.
Mechanistic Insights into Mixed Solvent Crystallization
The core scientific breakthrough of this purification technique lies in the nuanced understanding of solvation thermodynamics regarding the esomeprazole sodium salt. The inventors discovered that the target molecule exhibits high solubility in protic solvents due to hydrogen bonding interactions but demonstrates poor solubility or even insolubility in aprotic organic solvents. By creating a balanced mixed solvent environment, the process achieves a state where the crude material dissolves completely at ambient temperatures, allowing for the filtration of any insoluble particulate matter before crystallization begins. This pre-filtration step is crucial for removing mechanical impurities and seed crystals that could otherwise nucleate unwanted polymorphs or trap impurities within the crystal lattice. The subsequent addition of the aprotic anti-solvent shifts the solubility equilibrium sharply, forcing the pure S-enantiomer to precipitate out of the solution while the R-enantiomer and sulfone byproducts, which possess different solvation energies, remain dissolved.
From an impurity control perspective, this mechanism offers a dual advantage of kinetic and thermodynamic selectivity. The absence of thermal stress prevents the oxidation of the sulfide moiety to the sulfone, a common degradation pathway observed in heated processes. Additionally, the specific solvent ratios—typically ranging from 0.5 to 3.0 parts protic solvent to 1 part crude, and 4 to 10 parts aprotic solvent for crystallization—are optimized to maximize the rejection of chiral impurities. The data indicates that this precise control allows for the reduction of the R-enantiomer to undetectable levels, effectively solving the long-standing issue of optical purity in omeprazole derivatives. For R&D directors, this mechanistic clarity provides a clear path for process validation and regulatory filing, as the critical process parameters (CPPs) are well-defined and easily monitored.
How to Synthesize Esomeprazole Sodium Efficiently
Implementing this purification protocol requires careful attention to solvent selection and mass ratios to replicate the high purity outcomes described in the patent literature. The process is designed to be operationally simple, avoiding the need for specialized cryogenic equipment or high-energy heating mantles, which makes it highly attractive for facility retrofits or new plant designs. Operators simply need to maintain the reaction mixture within a mild temperature window of 0°C to 30°C throughout the dissolution and crystallization phases. The detailed standardized synthesis steps below outline the precise sequence of operations required to achieve the reported >99.5% purity and minimal solvent residue, ensuring that your production team can execute the method with confidence and reproducibility.
- Dissolve crude Esomeprazole Sodium in a mixed solvent comprising a protic solvent (e.g., isopropanol) and an aprotic solvent (e.g., butyl acetate) at ambient temperature (0-30°C).
- Filter the solution to remove insoluble impurities, then add a significant volume of additional aprotic solvent to induce crystallization while maintaining constant temperature.
- Separate the crystallized product via filtration or centrifugation and dry under reduced pressure at 30-50°C to obtain refined Esomeprazole Sodium with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this isothermal purification method represents a significant strategic advantage in the competitive landscape of generic drug manufacturing. By eliminating the heating and cooling cycles, the process inherently reduces energy consumption and minimizes the thermal load on production equipment, leading to substantial cost savings in utility expenditures. Furthermore, the simplified workflow reduces the overall cycle time per batch, allowing facilities to increase throughput without expanding their physical footprint. This efficiency gain is critical for meeting tight delivery schedules and managing inventory levels effectively in a just-in-time supply chain environment. The use of common, industrially available solvents like isopropanol and butyl acetate also mitigates supply risk, ensuring that raw material availability remains stable even during global market fluctuations.
- Cost Reduction in Manufacturing: The elimination of thermal cycling removes the need for expensive heating and chilling utilities, directly lowering the variable cost per kilogram of produced API. Additionally, the high yield of over 85% means less raw material is wasted, optimizing the overall material balance and reducing the cost of goods sold. The process also avoids the use of costly chiral columns or toxic solvents like toluene, which require expensive disposal and abatement systems, further enhancing the economic viability of the production route.
- Enhanced Supply Chain Reliability: Because the process operates under mild conditions with common solvents, it is less susceptible to disruptions caused by equipment failure or raw material shortages. The robustness of the isothermal crystallization ensures consistent batch quality, reducing the rate of failed batches and the need for re-processing. This reliability allows supply chain managers to forecast production output with greater accuracy, ensuring that downstream formulation partners receive their materials on time and specification, every time.
- Scalability and Environmental Compliance: The simplicity of the method facilitates seamless scale-up from pilot plant to commercial production, as heat transfer limitations associated with cooling large reactors are removed. Moreover, the replacement of toxic solvents with greener alternatives aligns with increasingly strict environmental regulations, reducing the regulatory burden and potential fines associated with volatile organic compound (VOC) emissions. This environmental stewardship enhances the corporate reputation of the manufacturer and ensures long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own manufacturing lines or for procurement specialists assessing the quality credentials of potential suppliers.
Q: How does this purification method control the R-enantiomer impurity?
A: The method utilizes specific solubility differences in a mixed solvent system to selectively crystallize the S-enantiomer while keeping the R-enantiomer and sulfone impurities in the mother liquor, achieving levels below 0.01%.
Q: Why is the isothermal process preferred over traditional heating and cooling cycles?
A: Esomeprazole sodium is thermally sensitive; avoiding heating prevents degradation into sulfone impurities, while eliminating the need for deep cooling reduces energy costs and simplifies industrial scale-up.
Q: What are the residual solvent levels in the final purified product?
A: The optimized solvent system allows for efficient removal of residual solvents like isopropanol and butyl acetate, ensuring the final product meets strict pharmacopoeial standards for solvent residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced purification technologies requires a partner with deep technical expertise and a proven track record of execution. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this isothermal crystallization process are fully realized in a practical manufacturing setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities down to the ppm level, guaranteeing that every batch of Esomeprazole Sodium we deliver meets the highest global pharmacopoeial standards. Our commitment to quality assurance means that you can rely on us for consistent supply without compromising on the critical optical purity required for this sensitive chiral drug.
We invite you to engage with our technical procurement team to discuss how this innovative purification method can be integrated into your supply chain to drive efficiency and quality. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your operational context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your position in the global gastrointestinal therapeutics market.
