Revolutionizing Olefin Metathesis: Scalable Synthesis of First Generation Grubbs Catalyst
Revolutionizing Olefin Metathesis: Scalable Synthesis of First Generation Grubbs Catalyst
The landscape of organometallic catalysis has long been defined by the tension between high performance and synthetic accessibility, a challenge acutely felt in the production of ruthenium-based olefin metathesis catalysts. Patent CN101555260B introduces a transformative methodology for synthesizing the first-generation Grubbs catalyst, RuCl2(=CHPh)(PCy3)2, by fundamentally re-engineering the precursor strategy to utilize inexpensive and readily available ruthenium trivalent halides (RuX3·nH2O). This technical breakthrough addresses the critical bottlenecks of safety, cost, and scalability that have historically constrained the widespread adoption of these powerful catalytic tools in pharmaceutical and fine chemical manufacturing. By shifting away from hazardous diazo compounds and moisture-sensitive organometallic reagents, this patented route offers a robust platform for producing high-purity catalysts suitable for demanding industrial applications. For R&D directors and procurement strategists, understanding the mechanistic elegance and economic implications of this synthesis is essential for optimizing supply chains and reducing the total cost of ownership for complex molecular synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ruthenium carbene complexes has been plagued by severe safety hazards and operational complexities that limit their commercial viability. The original method developed by the Grubbs group in 1996 relied on the reaction of RuCl2(PPh3)3 with phenyldiazomethane at cryogenic temperatures of -78°C, a process depicted in the reaction scheme below.
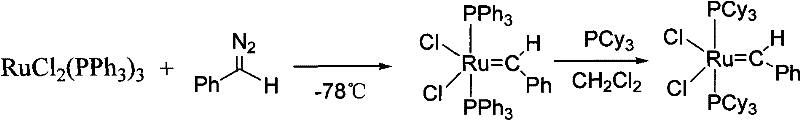
This reliance on phenyldiazomethane presents an unacceptable risk profile for large-scale manufacturing due to the compound's extreme explosiveness and thermal instability, necessitating specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, subsequent improvements involving ruthenium hydride species, while safer, introduced significant chemical inefficiencies where the product catalyst would undergo unwanted metathesis reactions with excess olefins present in the system, thereby reducing isolated yields and complicating purification workflows. Alternative approaches utilizing sulfur ylides or diethylzinc, as shown in the following scheme, offered milder conditions but introduced new vulnerabilities regarding reagent stability.
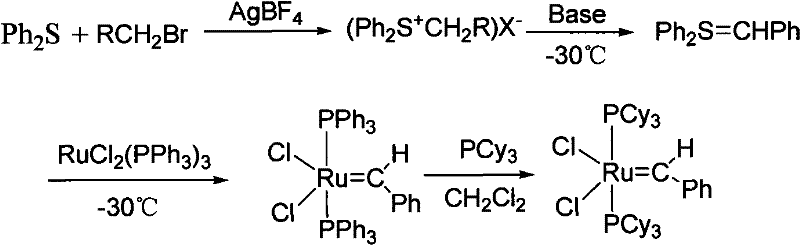
These alternative pathways require reagents that are exquisitely sensitive to moisture and oxygen, demanding strictly anhydrous and anaerobic environments that are difficult and costly to maintain on a multi-kilogram or metric-ton scale. The cumulative effect of these limitations is a supply chain characterized by high volatility, inconsistent quality, and prohibitive costs, which stifles innovation in downstream applications such as API intermediate synthesis and polymer modification.
The Novel Approach
The methodology disclosed in CN101555260B circumvents these historical impediments by establishing a streamlined three-step sequence initiated from commodity-grade ruthenium trivalent halides. This novel approach leverages the reduction of Ru(III) to Ru(II) and subsequently to Ru(0) using benign alcohols and common dienes like 1,5-cyclooctadiene (COD), effectively bypassing the need for dangerous diazo precursors or pyrophoric organozinc species. The process operates under reflux conditions in standard solvents such as ethanol and isopropanol, which are not only inexpensive but also facilitate easier solvent recovery and waste management compared to the chlorinated or ether solvents often required in legacy methods. By decoupling catalyst production from hazardous reagents, this invention enables a dramatic simplification of the operational workflow, allowing for continuous processing and significantly improved batch-to-batch consistency. For supply chain leaders, this translates to a reliable source of high-performance catalyst that is no longer subject to the whims of specialty reagent availability or extreme safety restrictions.
Mechanistic Insights into Ruthenium Reduction and Carbene Formation
The core chemical innovation of this patent lies in the controlled stepwise reduction of the ruthenium center, which ensures high fidelity in the formation of the active metal-carbon double bond. The initial step involves the coordination of a substituted cyclohexadiene to the ruthenium trivalent halide in an alcoholic medium, facilitating a redox process that stabilizes the metal in the divalent state as a p-cymene dimer. This intermediate serves as a crucial pivot point, providing a stable scaffold that protects the metal center from aggregation or oxidation before the introduction of the carbene ligand. The subsequent reaction with COD and a weak base, such as sodium carbonate, drives the reduction to the zero-valent state, generating a coordinatively unsaturated species that is primed for ligand exchange. This mechanistic pathway is superior because it avoids the violent decomposition pathways associated with diazo compounds, ensuring that the energy input is used efficiently for bond formation rather than dissipated through side reactions or thermal runaway events.
Furthermore, the final carbene formation step utilizes benzyl chloride derivatives in hydrocarbon solvents, a choice that critically impacts the impurity profile of the final product. Unlike methods where excess olefins compete for the metal center, this route employs a stoichiometrically controlled environment where the phosphine ligands (PCy3) and the carbene precursor react selectively with the zero-valent ruthenium species. This selectivity minimizes the formation of ruthenium hydride byproducts or bis-carbene species that often contaminate batches produced via hydride routes. For R&D teams focused on impurity profiling, this means a cleaner crude product that requires less aggressive chromatographic purification, preserving the integrity of the sensitive ruthenium-carbon bond. The robustness of this mechanism against trace moisture and oxygen, inherent to the stability of the intermediates, further ensures that the catalytic activity remains consistent, a vital parameter for reproducible olefin metathesis in complex molecule synthesis.
How to Synthesize First Generation Grubbs Catalyst Efficiently
Implementing this patented synthesis requires precise control over reaction parameters to maximize the yield of the zero-valent intermediate and ensure complete conversion to the final carbene complex. The process is designed to be operationally simple, utilizing standard glassware or stainless steel reactors without the need for exotic materials resistant to highly corrosive or explosive intermediates. Detailed below is the strategic overview of the synthesis workflow, highlighting the critical control points for temperature, solvent choice, and stoichiometry that define the success of the operation. For technical teams preparing for technology transfer, adhering to the specific reflux times and crystallization protocols outlined in the patent examples is essential to achieve the reported high yields and purity specifications.
- Reflux ruthenium trivalent halide (RuX3·nH2O) with substituted cyclohexadiene in alcohol to form divalent ruthenium dimer.
- React the divalent ruthenium intermediate with 1,5-cyclooctadiene (COD) and weak base in alcohol under reflux to generate zero-valent ruthenium complex.
- Treat the zero-valent complex with tricyclohexylphosphine (PCy3) and benzyl chloride derivative in hydrocarbon solvent to finalize the carbene catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this novel synthesis route offers profound economic and logistical benefits that extend far beyond the laboratory bench, directly addressing the pain points of procurement managers and supply chain directors. By replacing explosive and specialty reagents with commodity chemicals, the manufacturing cost structure is fundamentally altered, removing the premium pricing associated with hazardous material handling and disposal. This shift allows for a more predictable budgeting process and reduces the dependency on single-source suppliers of niche organometallic precursors, thereby mitigating supply risk. Moreover, the use of common solvents like ethanol and toluene simplifies the regulatory compliance landscape, as these materials are well-understood by environmental health and safety teams globally, facilitating faster permitting and smoother audits. The ability to operate at ambient or moderate reflux temperatures rather than cryogenic conditions also drastically reduces energy consumption, contributing to both cost savings and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents like phenyldiazomethane and diethylzinc removes significant cost drivers from the bill of materials, while the high yields reported in the patent examples indicate efficient atom economy. This efficiency translates to lower waste generation and reduced costs associated with solvent recovery and hazardous waste disposal, creating a leaner production model. Additionally, the simplified purification requirements mean less consumption of silica gel and eluents, further driving down the variable costs per kilogram of catalyst produced. These cumulative savings allow for a more competitive pricing strategy for the final catalyst, making olefin metathesis accessible for larger volume applications that were previously cost-prohibitive.
- Enhanced Supply Chain Reliability: Sourcing ruthenium trivalent halides and cyclohexadienes is significantly more straightforward than procuring unstable diazo compounds or sensitive sulfur ylides, which often have long lead times and strict shipping restrictions. This abundance of raw materials ensures that production schedules are not disrupted by upstream supply shortages, providing a steady flow of catalyst to downstream customers. The robustness of the process also means that manufacturing can be distributed across multiple geographic locations without requiring specialized infrastructure, enhancing resilience against regional disruptions. For supply chain heads, this reliability is paramount for maintaining continuous operations in pharmaceutical and agrochemical production lines where catalyst availability is a critical path item.
- Scalability and Environmental Compliance: The use of non-halogenated solvents in the initial steps and the avoidance of explosive intermediates make this process inherently safer and easier to scale from pilot plant to commercial production volumes. Safety incidents are minimized, reducing insurance premiums and liability risks, while the simpler waste stream facilitates easier treatment and compliance with increasingly stringent environmental regulations. The process design supports linear scale-up without the nonlinear safety risks associated with energetic materials, allowing manufacturers to confidently increase batch sizes to meet growing market demand. This scalability ensures that the supply of high-purity Grubbs catalyst can grow in tandem with the expansion of metathesis applications in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific manufacturing needs and regulatory environments.
Q: Why is the RuX3 starting material preferred over diazomethane routes?
A: The RuX3 route eliminates the use of highly explosive phenyldiazomethane required in traditional methods, significantly enhancing operational safety and allowing for larger batch sizes without cryogenic conditions.
Q: How does this method improve product purity compared to hydride routes?
A: Unlike hydride-based syntheses where excess olefins can cause unwanted metathesis side reactions, this pathway utilizes stable precursors that minimize byproduct formation, simplifying downstream purification.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the process uses common solvents like ethanol and toluene and avoids moisture-sensitive reagents like organozinc compounds, making it robust and economically viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Grubbs Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced catalytic technologies requires more than just access to a patent; it demands a partner with the technical expertise to execute complex syntheses with precision and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this ruthenium halide route are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Grubbs catalyst meets the exacting standards required for pharmaceutical intermediate synthesis and high-value fine chemical applications. Our commitment to quality assurance means that you receive a product that performs reliably in your reactions, minimizing downtime and maximizing yield in your own processes.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this safer, more scalable method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your transition to this next-generation catalyst supply is seamless and strategically sound.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
