Scalable Biomimetic Oxidation of o-Nitrotoluene for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high efficiency with stringent safety standards. A pivotal advancement in this domain is documented in patent CN100395228C, which details a novel method for preparing o-nitrobenzoic acid through biomimetic catalytic oxygen oxidation of o-nitrotoluene. This technology represents a significant departure from conventional oxidative processes by utilizing a specific class of metal phthalocyanine and metalloporphyrin catalysts within an ethanol-water solvent system. By shifting away from hazardous organic solvents like anhydrous methanol or benzene, this method addresses critical pain points regarding operator safety and environmental compliance while simultaneously enhancing reaction yields. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is essential for evaluating next-generation supply chain partners who can deliver high-purity intermediates with reduced risk profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-nitrobenzoic acid has relied heavily on oxidation reactions conducted in anhydrous methanol or methanol-benzene mixtures under alkaline conditions. While these methods were effective in solubilizing reactants, they introduced severe safety liabilities that are increasingly unacceptable in modern manufacturing environments. Anhydrous methanol, when used in the presence of pure oxygen, creates a volatile mixture with a wide explosion limit, posing a catastrophic risk during both the reaction and subsequent distillation phases. Furthermore, the inclusion of benzene as a co-solvent introduces known carcinogenic hazards, necessitating expensive containment systems and rigorous health monitoring for personnel. From a supply chain perspective, reliance on such hazardous solvents complicates logistics, increases insurance premiums, and creates potential bottlenecks due to strict regulatory controls on toxic chemical transport and storage.
The Novel Approach
The innovative methodology described in the patent data overcomes these legacy issues by employing an ethanol-water solution as the primary reaction medium. This solvent system is not only significantly less toxic but also exhibits a much higher flash point and a narrower explosion limit compared to methanol, fundamentally altering the safety profile of the oxidation process. By successfully enabling free radical oxidation in an aqueous environment—a feat previously considered difficult due to water's tendency to quench radicals—this approach achieves product yields that surpass those of traditional methods. The transition to ethanol-water eliminates the need for benzene entirely, thereby removing a major source of environmental contamination and occupational health risk. This strategic shift allows manufacturers to streamline their safety protocols and reduce the overhead costs associated with hazardous waste disposal and solvent recovery.
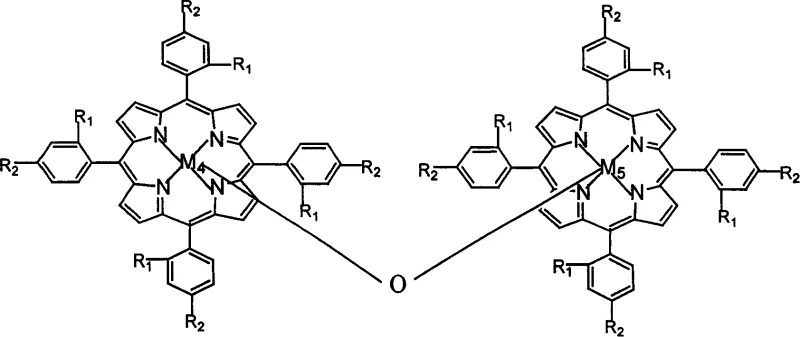
Mechanistic Insights into Biomimetic Catalytic Oxygen Oxidation
At the heart of this technological breakthrough lies the utilization of biomimetic catalysts, specifically metal phthalocyanines and metalloporphyrins, which mimic the activity of natural enzymes like cytochrome P450. These catalysts facilitate the activation of molecular oxygen under mild conditions, typically between 35°C and 55°C, allowing for the selective oxidation of the methyl group on o-nitrotoluene to a carboxyl group. The structural versatility of these catalysts, defined by various central metal atoms such as Iron, Cobalt, Manganese, and Copper, allows for fine-tuning of the electronic properties to optimize reaction kinetics. The ability of these complexes to stabilize reactive oxygen species in an aqueous alkaline medium challenges the conventional wisdom that water inhibits such radical pathways, demonstrating a sophisticated control over the reaction trajectory that minimizes side-product formation.
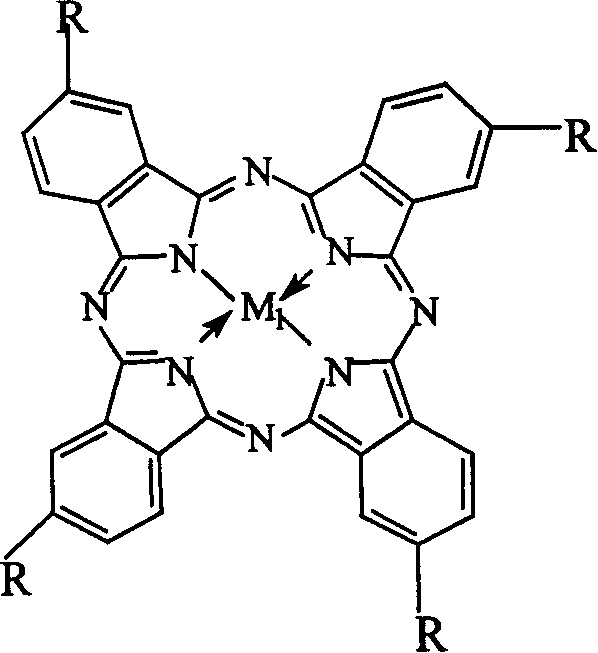
Impurity control is another critical aspect where this biomimetic system excels, particularly for applications requiring high-purity pharmaceutical intermediates. The specificity of the metalloporphyrin catalyst reduces the formation of over-oxidized byproducts or incomplete oxidation intermediates like o-nitrobenzaldehyde, which are common in less selective processes. The alkaline ethanol-water environment further aids in solubilizing the intermediate salts, preventing precipitation that could lead to localized hot spots or inconsistent reaction rates. For quality assurance teams, this means a cleaner crude product profile that requires less intensive downstream purification, directly translating to higher overall process efficiency. The robustness of the catalyst system ensures consistent performance across batches, a key requirement for maintaining the stringent impurity specifications demanded by global regulatory bodies.
How to Synthesize o-Nitrobenzoic Acid Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the biomimetic catalyst system. The process begins with the careful selection of the catalyst variant, such as iron phthalocyanine or μ-oxygen binuclear manganese porphyrin, depending on the specific kinetic requirements of the production scale. Operators must maintain the oxygen pressure within the range of 0.8 to 3.0 MPa and ensure the ethanol concentration in the solvent remains between 50% and 95% by volume to sustain the optimal reaction environment. Detailed standard operating procedures regarding catalyst loading, base addition, and temperature ramping are critical to replicating the high yields observed in the patent examples. The standardized synthesis steps outlined below provide a foundational framework for scaling this chemistry from laboratory verification to commercial production.
- Load o-nitrotoluene, biomimetic catalyst (metal phthalocyanine or porphyrin), and sodium hydroxide into a high-pressure autoclave reactor.
- Add an ethanol-water solvent mixture (50-95% ethanol volume) to create a safe, alkaline reaction medium.
- Pressurize with oxygen (0.8-3.0 MPa) and maintain temperature between 35-55°C for 2 to 14 hours to complete oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ethanol-water based oxidation technology offers substantial strategic advantages beyond mere technical performance. The elimination of benzene and the reduction of methanol usage significantly lower the regulatory burden associated with handling hazardous materials, leading to streamlined compliance audits and reduced administrative overhead. The inherent safety of the solvent system minimizes the risk of production shutdowns due to safety incidents, ensuring greater continuity of supply for downstream customers. Additionally, the use of ethanol, a commodity chemical with a stable global supply, mitigates the volatility risks associated with specialized or controlled solvents. This stability allows for more accurate long-term cost forecasting and strengthens the resilience of the supply chain against market fluctuations.
- Cost Reduction in Manufacturing: The shift to an ethanol-water solvent system eliminates the need for expensive solvent recovery processes required for benzene and reduces the energy consumption associated with distilling low-boiling methanol. By avoiding the use of toxic benzene, manufacturers also save significantly on hazardous waste treatment and disposal fees, which are often calculated based on toxicity levels. Furthermore, the higher reaction yields achieved with this method mean that less raw material is wasted per unit of product, optimizing the overall material balance. These cumulative efficiencies result in a leaner cost structure without compromising on the quality or purity of the final o-nitrobenzoic acid.
- Enhanced Supply Chain Reliability: Utilizing a safer solvent system reduces the likelihood of regulatory interventions or transportation delays that frequently plague shipments of highly flammable or toxic chemicals. Ethanol is widely available and easier to transport globally, ensuring that production schedules are not disrupted by solvent shortages. The mild reaction conditions also reduce wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. This reliability is crucial for maintaining just-in-time delivery commitments to pharmaceutical clients who cannot afford interruptions in their own manufacturing pipelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at moderate temperatures and pressures, making it suitable for large-scale reactors without requiring exotic metallurgy or extreme engineering controls. The aqueous nature of the waste stream simplifies effluent treatment, allowing for easier adherence to increasingly strict environmental discharge regulations. By adopting this green chemistry approach, companies can enhance their corporate sustainability profiles, meeting the ESG (Environmental, Social, and Governance) criteria that are becoming mandatory for suppliers to major multinational corporations. This alignment with global sustainability goals opens up new market opportunities and strengthens partnerships with environmentally conscious buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic oxidation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders assess the feasibility of integrating this method into their existing production frameworks. The focus is on clarifying the safety, yield, and scalability aspects that are most critical for decision-making in a commercial context.
Q: Why is the ethanol-water solvent system safer than traditional methanol methods?
A: Traditional methods often use anhydrous methanol or methanol-benzene mixtures, which pose significant explosion risks under pure oxygen and have high toxicity. The ethanol-water system has a much higher flash point and narrower explosion limits, drastically reducing operational hazards.
Q: What is the expected yield improvement with this biomimetic catalyst technology?
A: Experimental data indicates that yields consistently exceed 82%, often reaching up to 85.9%, which is significantly higher than the 58-78% yields typically reported in prior art using methanol-based solvents.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under mild temperatures (35-55°C) and uses readily available solvents, making it highly suitable for commercial scale-up without requiring exotic equipment or extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes like the biomimetic oxidation of o-nitrotoluene to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent technology are fully realized in a manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of o-nitrobenzoic acid meets the highest international standards. Our commitment to process safety and environmental stewardship aligns perfectly with the advantages offered by this ethanol-water solvent system, making us an ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this safer, higher-yield process impacts your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to engineer a more efficient, safe, and sustainable supply chain for your critical chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
