Advanced Metalloporphyrin Catalytic Oxidation for Commercial o-Nitrobenzoic Acid Production
The pharmaceutical and agrochemical industries rely heavily on high-purity aromatic carboxylic acids, with o-nitrobenzoic acid serving as a critical building block for numerous active pharmaceutical ingredients and functional materials. A groundbreaking approach detailed in Chinese Patent CN103193654A introduces a highly efficient method for preparing o-nitrobenzoic acid through the catalytic oxidation of o-nitrotoluene. This innovation leverages a sophisticated metalloporphyrin-metal salt composite catalyst system that operates within a benign ethanol-water solvent medium. Unlike traditional oxidation methods that often suffer from harsh acidic conditions or toxic organic solvents, this biomimetic catalytic strategy achieves exceptional conversion rates and selectivity under mild alkaline conditions. The technical breakthrough lies in the precise tuning of redox potentials through the combination of specific metalloporphyrin structures and transition metal salts, enabling a synergistic activation of molecular oxygen. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in sustainable and cost-effective manufacturing technologies that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-nitrobenzoic acid has been plagued by significant technical and environmental challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional methods often relied on strong acidic media which caused severe corrosion to reactor equipment, necessitating expensive alloy materials and frequent maintenance schedules that disrupted production continuity. Furthermore, earlier biomimetic catalytic approaches utilizing methanol or benzene-based solvent systems presented substantial safety hazards due to high toxicity and flammability, creating rigorous regulatory compliance burdens for manufacturing facilities. Another critical bottleneck was the excessive consumption of alkali, typically ranging from 3.75mol/L to 6.0mol/L, which not only increased raw material costs but also generated massive amounts of saline wastewater during the subsequent acid neutralization steps. These legacy processes also suffered from prolonged reaction times, often extending up to 12 hours, resulting in high energy consumption and limited throughput capacity that failed to meet the dynamic demands of the global supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The novel methodology described in the patent data fundamentally reshapes the production landscape by introducing a metalloporphyrin-metal salt composite catalyst that operates in a much safer and more efficient ethanol-water solvent system. By replacing toxic organic solvents with a mixture containing 50% to 95% volume ethanol, the process eliminates major occupational health risks while maintaining excellent solubility for the reactants and catalysts. The core innovation involves the strategic combination of mononuclear or μ-oxo-binuclear metalloporphyrins with transition metal salts such as iron, cobalt, or manganese acetates and chlorides. This dual-catalyst system creates a powerful synergistic effect that dramatically accelerates the oxidation rate, reducing the required reaction time to merely 1-6 hours while operating at moderate temperatures between 35-55°C. Additionally, the alkali concentration is significantly lowered to a range of 0.5-2.5mol/L, which directly translates to reduced acid consumption for neutralization and a drastic decrease in saline waste discharge, thereby achieving true energy conservation and emission reduction goals for cost reduction in pharma manufacturing.
Mechanistic Insights into Metalloporphyrin-Metal Salt Synergistic Catalysis
The exceptional performance of this catalytic system stems from the intricate electronic interactions between the metalloporphyrin macrocycle and the added metal salt co-catalyst, which function together to activate molecular oxygen with unprecedented efficiency. The metalloporphyrin acts as a biomimetic center, mimicking natural enzymes like cytochrome P450, while the metal salt modulates the redox potential of the central metal ion within the porphyrin ring. This modulation facilitates the formation of high-valent metal-oxo species that are the active oxidants responsible for abstracting hydrogen atoms from the methyl group of o-nitrotoluene. The presence of different metal ions, such as combining iron porphyrins with manganese salts or cobalt porphyrins with zinc salts, creates a multi-electron transfer pathway that prevents the accumulation of inactive catalyst species and minimizes side reactions. This synergistic mechanism ensures that the oxidation proceeds selectively towards the carboxylic acid rather than stopping at intermediate aldehyde or alcohol stages, which is a common issue in single-component catalytic systems.
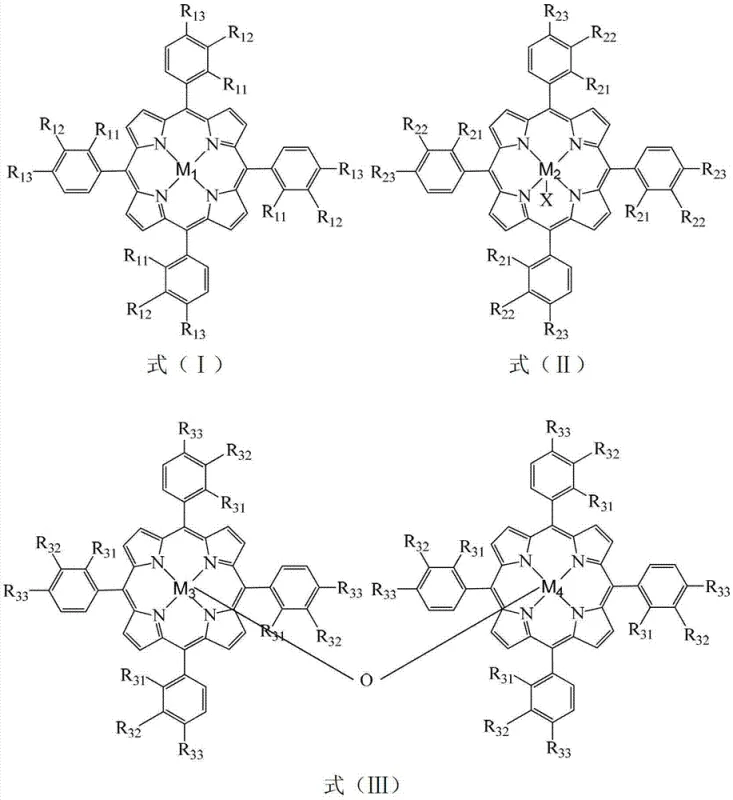
Furthermore, the structural diversity of the catalysts, as illustrated in the provided chemical diagrams, allows for fine-tuning the steric and electronic environment around the active site to match the specific substrate requirements. The patent describes various substituents on the phenyl rings of the porphyrin, such as chloro, nitro, hydroxy, or methoxy groups, which influence the electron density on the central metal and thus its reactivity towards oxygen activation. The μ-oxo-binuclear structures, in particular, offer a unique bridging oxygen atom that can participate in the oxygen transfer mechanism, potentially lowering the activation energy barrier even further compared to mononuclear analogues. Understanding these mechanistic details is crucial for R&D teams aiming to optimize the process for specific impurity profiles, as the choice of metal center and substituent pattern directly impacts the selectivity and the nature of by-products formed during the oxidation of o-nitrotoluene to o-nitrobenzoic acid.
How to Synthesize o-Nitrobenzoic Acid Efficiently
Implementing this advanced catalytic oxidation route requires precise control over reaction parameters to maximize the synergistic benefits of the composite catalyst system. The process begins with the preparation of the catalyst solution, where specific concentrations of metalloporphyrin (5-50ppm) and metal salt (5-200ppm) are dissolved in the alkaline ethanol-water medium. The substrate, o-nitrotoluene, is then introduced, and the system is pressurized with oxygen to drive the oxidation reaction forward. Maintaining the temperature within the optimal window of 35-55°C is critical to balance reaction kinetics with catalyst stability, ensuring high conversion without degrading the sensitive porphyrin structure. Detailed standard operating procedures regarding mixing speeds, oxygen flow rates, and work-up protocols are essential for reproducibility and safety in a plant environment.
- Prepare the reaction system by dissolving the metalloporphyrin catalyst (5-50ppm) and metal salt co-catalyst (5-200ppm) in an aqueous ethanol solution containing 0.5-2.5mol/L sodium hydroxide.
- Introduce o-nitrotoluene substrate into the high-pressure autoclave and seal the system to maintain an oxygen pressure between 0.5-2.5MPa.
- Heat the reaction mixture to 35-55°C and maintain stirring for 1-6 hours, followed by HPLC analysis to confirm conversion rates exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metalloporphyrin-catalyzed process offers transformative advantages that extend far beyond simple yield improvements, impacting the total cost of ownership and operational resilience. The drastic reduction in reaction time from traditional 12-hour cycles to as little as 1-6 hours effectively doubles the throughput capacity of existing reactor assets without requiring capital expenditure on new equipment. This acceleration allows manufacturers to respond more agilely to market fluctuations and urgent customer orders, significantly reducing lead time for high-purity pharmaceutical intermediates. Moreover, the shift to a low-toxicity ethanol-water solvent system simplifies solvent recovery and recycling operations, lowering the overall utility costs associated with distillation and waste treatment. These operational efficiencies combine to create a robust supply chain capable of delivering consistent quality at a competitive price point.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the substantial decrease in auxiliary chemical consumption and waste disposal costs. By lowering the alkali concentration from over 6.0mol/L to below 2.5mol/L, the process significantly reduces the amount of acid required for subsequent neutralization steps, which is a major cost driver in traditional batch processing. Additionally, the high selectivity of the composite catalyst minimizes the formation of by-products, meaning less raw material is wasted and downstream purification steps such as recrystallization become simpler and less resource-intensive. The elimination of toxic solvents like benzene also removes the need for specialized containment and hazardous waste handling fees, contributing to substantial cost savings in overall manufacturing operations without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly enhanced by the use of readily available and stable catalyst components that do not require complex storage conditions or rare earth elements. The robustness of the catalyst system under moderate oxygen pressures and temperatures reduces the risk of unplanned shutdowns due to equipment failure or safety incidents, ensuring a steady flow of goods to downstream customers. Furthermore, the simplified workflow with fewer processing steps reduces the potential for human error and batch-to-batch variability, leading to more predictable delivery schedules. This reliability is critical for long-term contracts with multinational corporations that demand strict adherence to Just-In-Time delivery models and consistent quality specifications for their API synthesis.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions which do not require extreme pressures or temperatures that pose engineering challenges at large scales. The use of oxygen as the terminal oxidant is inherently greener than stoichiometric oxidants like permanganate or dichromate, generating water as the primary by-product and aligning with increasingly stringent global environmental regulations. The reduction in saline wastewater generation alleviates the burden on effluent treatment plants, making it easier for manufacturing sites to maintain compliance with local discharge limits. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a sustainable partner in the global chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metalloporphyrin-catalyzed oxidation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Stakeholders often seek clarification on catalyst stability, solvent recovery, and impurity control, which are critical factors for technology transfer. The answers below summarize the key advantages and operational parameters that define the superiority of this method over conventional oxidation routes.
Q: How does the metalloporphyrin-metal salt composite improve yield compared to traditional methods?
A: The composite catalyst utilizes synergistic effects between different metal ions and ligands, significantly lowering activation energy and improving selectivity, raising yields from traditional 73.4%-85.9% to over 96.1%.
Q: What are the environmental benefits of this ethanol-water solvent system?
A: Replacing toxic benzene or methanol solvents with an ethanol-water mixture drastically reduces solvent toxicity and volatility, while lower alkali consumption minimizes saline wastewater generation during neutralization.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the mild reaction conditions (35-55°C, moderate oxygen pressure) and short reaction times (1-6h) make the process highly suitable for commercial scale-up with reduced energy consumption and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthesis routes for critical intermediates like o-nitrobenzoic acid in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the metalloporphyrin-catalyzed oxidation are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for API synthesis. Our commitment to technological excellence allows us to offer clients a secure and high-quality source of fine chemical intermediates that support their drug development timelines.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic technology can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits tailored to your production volume and quality requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
