Revolutionizing 7-ACA Production: Advanced Membrane Filtration and Enzymatic Hydrolysis for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for producing critical beta-lactam intermediates, and patent CN110423241B presents a transformative approach to synthesizing cephalosporin C sodium salt and its derivative, 7-aminocephalosporanic acid (7-ACA). This intellectual property details a sophisticated purification and conversion strategy that diverges significantly from legacy extraction techniques, leveraging advanced membrane technologies and precise enzymatic catalysis to achieve superior product profiles. By integrating ceramic membrane filtration with solvent extraction and immobilized enzyme hydrolysis, the disclosed process addresses long-standing inefficiencies in yield loss and environmental burden associated with traditional fermentation broth processing. For stakeholders evaluating reliable pharmaceutical intermediates supplier partnerships, understanding the mechanistic advantages of this route is essential for securing long-term supply chain resilience. The following analysis dissects the technical nuances of this innovation, highlighting its potential to redefine cost structures and quality benchmarks in the global cephalosporin market.
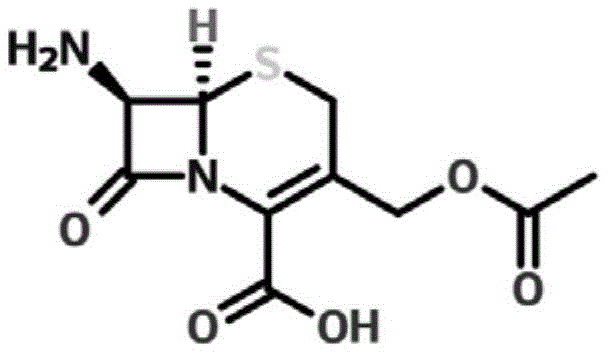
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of cephalosporin C from fermentation broths has been plagued by significant operational bottlenecks that compromise both economic efficiency and environmental compliance. Traditional protocols typically rely on acidification and coagulation followed by plate-and-frame filtration, a mechanical separation method prone to frequent blockages due to the fragile nature of the coagulated Sanskrit flower precipitates. Furthermore, the subsequent purification stages often necessitate the extensive use of macroporous adsorption resins and ion exchange resins, which generate substantial volumes of hazardous waste streams, including waste acid water and resin regeneration liquids. These effluents impose heavy financial burdens on manufacturers due to the high costs associated with wastewater treatment and solid waste disposal, while also introducing variability in product recovery rates. The reliance on these outdated separation technologies often results in inconsistent product quality, with residual impurities such as proteins, polysaccharides, and pigments persisting through to the final crystallization steps, thereby limiting the utility of the intermediate for high-end antibiotic synthesis.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined, multi-stage purification architecture that effectively circumvents the pitfalls of resin-based adsorption and mechanical filtration. By employing a dual-membrane system comprising 50-100 nanometer ceramic membranes followed by 20-100 KD ultrafiltration units, the process achieves a highly selective removal of mycelium, proteins, and colloidal impurities without the risk of pore clogging or filter cake collapse. This membrane-centric front-end purification is seamlessly coupled with a liquid-liquid extraction strategy using water-immiscible organic solvents like ethyl acetate, which concentrates the active cephalosporin C species while leaving hydrophilic contaminants behind. The subsequent salt formation with sodium isooctanoate and anti-solvent crystallization using acetone ensures the precipitation of cephalosporin C sodium salt with exceptional purity and crystal morphology. This integrated approach not only enhances the overall yield of the valuable intermediate but also drastically simplifies the downstream processing requirements, positioning it as a superior alternative for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Membrane-Assisted Extraction and Enzymatic Hydrolysis
The core innovation of this process lies in the precise orchestration of physical separation and chemical transformation steps, beginning with the rigorous clarification of the fermentation broth. The utilization of ceramic membranes at the initial stage provides a robust barrier against particulate matter, operating effectively at low temperatures (5-10°C) to preserve the stability of the thermally sensitive beta-lactam ring. Following this, the ultrafiltration step targets high-molecular-weight impurities such as pigments and residual proteins, achieving a filtrate with light transmittance values exceeding 60-80% at 425nm, which is indicative of a significantly reduced impurity load. The extraction phase leverages the differential solubility of cephalosporin C in ethyl acetate versus the aqueous phase, allowing for a concentration factor that prepares the molecule for efficient salt formation. The reaction with sodium isooctanoate is kinetically controlled, with the rapid addition of the sodium salt solution inducing a transient turbidity that quickly resolves, indicating the formation of a soluble ion-pair complex that partitions preferentially into the heavy aqueous phase. This phase separation mechanism is critical for isolating the active pharmaceutical ingredient from organic solvent residues and non-polar byproducts.
Following the isolation of the cephalosporin C sodium salt, the conversion to 7-aminocephalosporanic acid is mediated by immobilized cephalosporin C acylase, a biocatalyst that offers distinct advantages over chemical hydrolysis agents. The enzymatic cleavage of the side chain occurs under mild alkaline conditions (pH 8.00-8.50) and moderate temperatures (10-20°C), minimizing the risk of beta-lactam ring opening or epimerization that often plagues harsh chemical methods. The immobilization of the enzyme facilitates easy separation from the reaction mixture via simple screening, allowing for potential catalyst recycling and reducing the introduction of proteinaceous impurities into the final product stream. The crystallization of 7-ACA is then triggered by careful pH adjustment using hydrochloric acid, driving the supersaturation of the zwitterionic product while keeping soluble impurities in the mother liquor. This controlled crystallization regime ensures the formation of uniform crystals with low levels of specific related substances, such as deacetylated derivatives and dimers, thereby meeting the stringent purity specifications demanded by regulatory bodies for high-purity pharmaceutical intermediates.
How to Synthesize 7-Aminocephalosporanic Acid Efficiently
The practical implementation of this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. Operators must maintain precise control over temperature gradients during the membrane filtration and extraction phases, as deviations can lead to emulsion formation or incomplete impurity removal. The stoichiometry of the salt-forming reaction is equally critical, with a molar ratio of cephalosporin C to sodium isooctanoate maintained between 1:1.0 and 1:1.2 to prevent excess reagent contamination. Detailed standard operating procedures regarding the rate of anti-solvent addition and the specific activity of the immobilized enzyme are paramount for successful scale-up. For a comprehensive breakdown of the exact experimental conditions and sequential operations, please refer to the standardized guide below.
- Filter cephalosporin C fermentation broth using 50-100nm ceramic membranes followed by 20-100KD ultrafiltration to remove mycelium and pigments.
- Extract cephalosporin C into ethyl acetate, react with sodium isooctanoate to form the sodium salt, and crystallize using acetone as an anti-solvent.
- Hydrolyze the cephalosporin C sodium salt solution using immobilized cephalosporin C acylase at pH 8.0-8.5 to yield 7-aminocephalosporanic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this membrane-based and enzymatic process offers profound benefits that extend beyond mere technical superiority, directly impacting the bottom line and supply security. By eliminating the need for consumable adsorption resins and the associated regeneration chemicals, manufacturers can achieve substantial cost savings in raw material procurement and waste management overheads. The simplification of the unit operations reduces the complexity of the production line, lowering the capital expenditure required for equipment maintenance and the operational expenditure linked to labor and energy consumption. Furthermore, the enhanced purity of the intermediate reduces the burden on downstream purification steps in the synthesis of final cephalosporin antibiotics, creating a ripple effect of efficiency throughout the entire value chain. For procurement managers, this translates into a more predictable cost structure and a reduced risk of supply disruptions caused by environmental compliance issues or raw material shortages.
- Cost Reduction in Manufacturing: The elimination of macroporous and ion exchange resins removes a significant variable cost component, as these materials require frequent replacement and generate expensive hazardous waste streams that demand specialized treatment. Additionally, the high efficiency of the membrane filtration system minimizes product loss during the clarification stage, directly improving the overall mass balance and yield of the final 7-ACA product. The use of immobilized enzymes further contributes to cost optimization by enabling catalyst reuse and reducing the quantity of biocatalyst required per batch compared to free enzyme systems. These cumulative efficiencies result in a leaner manufacturing process that is less susceptible to fluctuations in the prices of auxiliary chemicals and waste disposal services.
- Enhanced Supply Chain Reliability: The robustness of the ceramic membrane technology ensures consistent throughput even when dealing with variations in fermentation broth quality, thereby stabilizing production schedules and delivery timelines. Unlike traditional filtration methods that are prone to unexpected downtime due to filter cloth blinding or resin fouling, this continuous-friendly process design supports higher availability rates for production assets. The ability to produce 7-ACA with consistently high purity reduces the likelihood of batch rejections or the need for reprocessing, which are common causes of supply delays in the pharmaceutical sector. Consequently, partners relying on this technology can expect a more dependable flow of critical intermediates, safeguarding their own downstream manufacturing operations against volatility.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as membrane modules and extraction columns can be easily paralleled or enlarged to meet increasing demand without fundamental changes to the chemistry. From an environmental standpoint, the drastic reduction in waste acid water and solid resin waste aligns with increasingly stringent global regulations on industrial effluent discharge, mitigating the risk of regulatory shutdowns or fines. The lower environmental footprint also enhances the sustainability profile of the supply chain, a factor that is becoming progressively important for multinational corporations aiming to meet their corporate social responsibility goals. This alignment with green chemistry principles future-proofs the supply source against evolving ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical evaluators. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production frameworks or for qualifying new suppliers.
Q: How does the membrane filtration process improve upon traditional resin adsorption?
A: The patented process replaces traditional plate-and-frame filtration and macroporous resin adsorption with ceramic and ultrafiltration membranes. This eliminates the generation of large volumes of waste acid water and resin regeneration waste liquid, significantly reducing environmental treatment costs and preventing filter clogging issues common in conventional coagulation methods.
Q: What represents the critical quality attributes of the 7-ACA produced by this method?
A: The process yields 7-aminocephalosporanic acid with exceptional purity, typically exceeding 99.5% content. Critical impurities such as deacetylated 7-ACA, deacetylated oxygen 7-ACA, and 7-ACA dimers are strictly controlled to levels below 0.2%, ensuring the intermediate meets the rigorous specifications required for downstream cephalosporin antibiotic synthesis.
Q: Is the enzymatic hydrolysis step scalable for industrial manufacturing?
A: Yes, the use of immobilized cephalosporin C acylase allows for efficient reuse of the biocatalyst and simplifies the separation process. The reaction operates under mild conditions (10-20°C, pH 8.0-8.5), making it highly suitable for large-scale commercial production while maintaining high conversion rates and product stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Aminocephalosporanic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain competitiveness in the global pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN110423241B can be successfully translated from the laboratory to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex beta-lactam chemistry positions us as a strategic partner capable of navigating the intricacies of cephalosporin intermediate production with precision and reliability.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and leverage these technological advancements for mutual growth. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities align with your project milestones. Let us demonstrate how our commitment to technical excellence and operational efficiency can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
