Advanced Benzamide Compound Synthesis: A Toxic-Free Route for Commercial Scale-up
Advanced Benzamide Compound Synthesis: A Toxic-Free Route for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high efficiency with stringent environmental compliance. Patent CN102276576B introduces a groundbreaking methodology for the preparation of benzamide compounds, specifically addressing the critical safety hazards associated with traditional cyanide-based routes. This technology leverages a mild, two-step activation strategy using trifluoroacetic acid succinimide ester to generate high-purity intermediates without the need for hazardous reagents like potassium cyanide or sodium hydride. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize the supply chain for reliable pharmaceutical intermediate supplier partnerships. By shifting to this greener chemistry, manufacturers can mitigate regulatory risks while maintaining the rigorous purity standards required for downstream API synthesis. The following analysis details the technical superiority and commercial viability of this novel approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzamide derivatives often relied on multi-step sequences involving highly toxic and reactive reagents that pose severe safety and environmental challenges. As illustrated in prior art, conventional routes frequently utilize potassium cyanide and sodium hydride to facilitate carbon-nitrogen bond formation or intermediate transformations. These substances are not only acutely toxic to personnel but also generate hazardous waste streams that require complex and expensive disposal protocols. Furthermore, the use of such aggressive reagents often necessitates strict temperature controls and anhydrous conditions, increasing the energy footprint and operational complexity of the manufacturing process. The reliance on these dangerous chemicals creates a bottleneck for cost reduction in pharmaceutical intermediate manufacturing, as safety mitigation measures drive up overhead costs significantly.
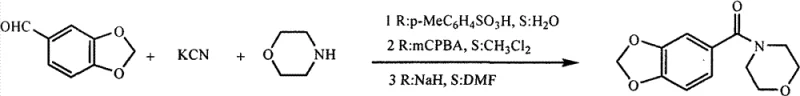
The Novel Approach
In stark contrast, the method disclosed in CN102276576B utilizes a benign activation strategy that operates under exceptionally mild conditions, fundamentally altering the risk profile of benzamide production. The process begins with the reaction of substituted benzoic acid and trifluoroacetic acid succinimide ester at room temperature to form a highly reactive succinimide ester intermediate. This activated species then undergoes nucleophilic substitution with various amines, such as morpholine, piperidine, or pyrrolidine, to yield the target benzamide compound efficiently. By eliminating the need for cyanides and strong bases, this route drastically simplifies the safety infrastructure required for production. The reaction typically completes within 5 hours at ambient temperature, offering a streamlined workflow that enhances throughput while minimizing energy consumption and environmental impact.
Mechanistic Insights into Active Ester-Mediated Amidation
The core of this technological advancement lies in the formation and utilization of the succinimide ester intermediate, which serves as a superior electrophile for amine coupling. In the first stage, the carboxylic acid group of the substituted benzoic acid is activated by the trifluoroacetic acid succinimide ester in the presence of a base like triethylamine. This generates an O-acylisourea-like transition state that rapidly converts to the stable succinimide ester, as depicted in the structural analysis of the intermediate. This activation method is particularly advantageous because it avoids the formation of racemization-prone species often seen with other coupling agents, ensuring the stereochemical integrity of chiral substrates if present. The mildness of this activation step allows for a broad scope of functional group tolerance, making it a versatile tool for synthesizing complex fine chemical intermediates.
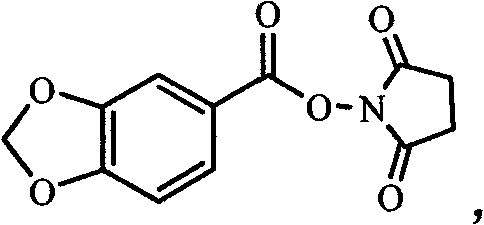
Following activation, the second stage involves the nucleophilic attack of the amine on the carbonyl carbon of the succinimide ester. This aminolysis reaction proceeds smoothly at room temperature, driven by the good leaving group ability of the succinimide moiety. The resulting tetrahedral intermediate collapses to release N-hydroxysuccinimide and form the stable amide bond of the final benzamide product. This mechanism ensures high conversion rates and minimizes the formation of side products, which is critical for achieving the high-purity OLED material or API intermediate standards demanded by global markets. The simplicity of the workup, involving only dilute hydrochloric acid washing and recrystallization, further underscores the efficiency of this mechanistic pathway in removing impurities without complex chromatography.
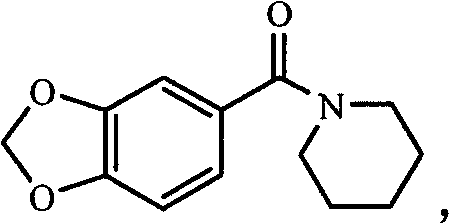
How to Synthesize Benzamide Compound Efficiently
The operational protocol for this synthesis is designed for ease of execution and scalability, making it ideal for both laboratory optimization and industrial production. The process involves dissolving the substituted benzoic acid in a solvent like dichloromethane, adding the activating ester and base, and stirring until the intermediate is formed. Subsequently, the chosen amine is introduced to complete the amidation, followed by a straightforward aqueous workup. Detailed standardized synthesis steps see the guide below for precise molar ratios and timing.
- Activate substituted benzoic acid with trifluoroacetic acid succinimide ester in dichloromethane with triethylamine at room temperature.
- React the resulting succinimide ester intermediate with amines such as morpholine or piperidine in chloroform or dichloromethane.
- Purify the final benzamide product by washing with dilute hydrochloric acid, drying, and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For Supply Chain Heads and Procurement Managers, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical efficiency. The elimination of highly toxic reagents like potassium cyanide removes the need for specialized hazardous material handling and storage facilities, leading to substantial cost savings in infrastructure and compliance. Additionally, the room temperature operation significantly reduces energy consumption compared to processes requiring reflux or cryogenic cooling, contributing to a lower carbon footprint and reduced utility costs. The simplified purification process, which avoids complex column chromatography in favor of crystallization, enhances the overall throughput and reduces the lead time for high-purity pharmaceutical intermediates. These factors combine to create a more resilient and cost-effective supply chain capable of meeting demanding delivery schedules.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents such as sodium hydride and potassium cyanide eliminates the associated costs of safety monitoring, specialized waste disposal, and neutralization processes. Furthermore, the high yield and selectivity of the reaction minimize raw material waste, ensuring that a greater proportion of input materials are converted into saleable product. This efficiency translates directly into a more competitive pricing structure for the final benzamide compounds, allowing for better margin management in volatile chemical markets.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like substituted benzoic acids and common amines, the risk of raw material shortages is significantly mitigated. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations, ensuring a steady flow of goods. This reliability is crucial for maintaining continuous manufacturing lines for downstream clients who depend on just-in-time delivery of critical intermediates for their own production schedules.
- Scalability and Environmental Compliance: The mild nature of the chemistry facilitates easy scale-up from kilogram to multi-ton scales without the need for exotic reactors or extreme safety measures. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, reducing the risk of fines or shutdowns due to non-compliance. This sustainability profile enhances the brand value of the manufacturer and appeals to eco-conscious partners seeking green chemistry solutions for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzamide synthesis technology. These answers are derived directly from the patent data and practical manufacturing experience to provide clarity on feasibility and advantages. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing production portfolios.
Q: How does this method improve environmental safety compared to conventional synthesis?
A: This method eliminates the use of highly toxic potassium cyanide and sodium hydride, significantly reducing hazardous waste generation and operator risk.
Q: What are the typical reaction conditions for this benzamide synthesis?
A: The reaction proceeds under mild conditions at room temperature, typically completing within 5 hours without the need for extreme heating or cooling.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the simplified post-treatment involving acid washing and recrystallization makes it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzamide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that deliver both quality and sustainability. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of patent CN102276576B are fully realized at an industrial level. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of benzamide compound meets the highest international standards. We are committed to providing a seamless transition from process development to commercial supply, leveraging our technical expertise to optimize yields and minimize costs for our partners.
We invite you to collaborate with us to explore how this green synthesis method can enhance your product portfolio and reduce your environmental footprint. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this approach for your projects. Let us help you secure a reliable supply of high-quality intermediates while driving efficiency and sustainability in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
